Abstract
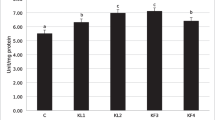
This study was carried out to investigate and compare the effect of water-soluble polysaccharides extract of the green alga, Ulva rigida (WPU), as a dietary supplement on growth performance, antioxidant enzyme activity, non-specific immune responses, and disease resistance of juvenile grey mullet, Mugil cephalus, subjected to bacterial infection with Photobacterium damselae. Three replicate groups of fish (initial mean weight, 15 ± 0.1 g) were fed with four isonitrogenous and isolipidic diets containing four levels, 0 or control, 5, 10, and 15 mg kg−1 of WPU (WPU0 or control, WPU5, WPU10, and WPU15) for 8 weeks over the growth trial. Thereafter, ten fish from each dietary treatment were infected with bacteria P. damselae to evaluate disease resistance of infected fish. Feeding grey mullet at 10 mg kg−1 WPU level (WPU10 diets) produced higher weight gain (211.36 ± 6%), specific growth rate (1.88 ± 0.03%) and protein efficiency ratio (4.26 ± 0.1%) compare to the control group. The diets supplemented with three levels of WPU-stimulated lysozyme, phagocytic, and respiratory burst activity of fish, but, the stimulatory capacities of the extracts varied greatly, depending on the concentrations used and diet; WPU10 was higher than WPU 5 and WPU15 diets. The best responses in antioxidant enzyme activities of superoxide dismutase (4.8 U mL−1), glutathione (3.6 U mL−1), and malondialdehyde (25.0 U mL−1) were induced by the extracts obtained from WPU10 diet. In addition, using WPU extract in all diets decreased mortality in M. cephalus in a dose-dependent manner after challenge with P. damselae. These results suggest that the use of water-soluble polysaccharides from U. rigida at 10 mg kg−1 dose improve growth, immune responses, and antioxidant activity in M. cephalus.

Similar content being viewed by others
References
Abbassa A-H (2006) The impact of striped mullet, Mugil cephalus on natural food and phytoplankton selectivity at different feeding regimes in earthen fishponds. J Fish Aquat Sci 1:87–96
Alderman D, Hastings T (1998) Antibiotic use in aquaculture: development of antibiotic resistance–potential for consumer health risks. Int J Food Sci Technol 33:139–155
Ardó L, Yin G, Xu P, Váradi L, Szigeti G, Jeney Z, Jeney G (2008) Chinese herbs (Astragalus membranaceus and Lonicera japonica) and boron enhance the non-specific immune response of Nile tilapia (Oreochromis niloticus) and resistance against Aeromonas hydrophila. Aquaculture 275:26–33
Austin B, Austin D (2007) Bacterial fish pathogens: diseases of farmed and wild fish. Springer, Cham, pp 15–46
Bikker P, van Krimpen MM, van Wikselaar P, Houweling-Tan B, Scaccia N, van Hal JW, Huijgen WJ, Cone JW, López-Contreras AM (2016) Biorefinery of the green seaweed Ulva lactuca to produce animal feed, chemicals and biofuels. J Appl Phycol 28:3511–3525
Boyd CE, Massaut L (1999) Risks associated with the use of chemicals in pond aquaculture. Aquac Eng 20:113–132
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bricknell I, Dalmo RA (2005) The use of immunostimulants in fish larval aquaculture. Fish Shellfish Immunol 19:457–472
Cabello FC (2006) Heavy use of prophylactic antibiotics in aquaculture: a growing problem for human and animal health and for the environment. Environ Microbiol 8:1137–1144
Castro R, Piazzon M, Zarra I, Leiro J, Noya M, Lamas J (2006) Stimulation of turbot phagocytes by Ulva rigida C. Agardh polysaccharides. Aquaculture 254:9–20
Castro R, Zarra I, Lamas J (2004) Water-soluble seaweed extracts modulate the respiratory burst activity of turbot phagocytes. Aquaculture 229:67–78
Crosetti D (2016) Current state of grey mullet fisheries and culture. In: Biology, Ecology and Culture of Grey Mullets (Mugilidae). CRC Press, Boca Raton pp 398–450
Quezada-Rodríguez P, Fajer-Ávila EJ (2017) The dietary effect of ulvan from Ulva clathrata on hematological-immunological parameters and growth of tilapia (Oreochromis niloticus). J Appl Phycol 29:423–431
Dügenci SK, Arda N, Candan A (2003) Some medicinal plants as immunostimulant for fish. J Ethnopharmacol 88:99–106
El Zarka S, Fahmy FK (1968) Experiments in the culture of the grey mullet Mugil cephalus in brackishwater ponds in the UAR. FAO Fish Rep 44(5):255–266
Ellis AE (1990) Lysozyme assays. Techn Fish Immunol 1:101–103
Ergün S, Soyutürk M, Güroy B, Güroy D, Merrifield D (2009) Influence of Ulva meal on growth, feed utilization, and body composition of juvenile Nile tilapia (Oreochromis niloticus) at two levels of dietary lipid. Aquac Int 17:355
Kakoolaki S, Akbary P, Zorriehzahra MJ, Salehi H, Sepahdari A, Afsharnasab M, Mehrabi MR, Jadgal S (2016) Camellia sinensis supplemented diet enhances the innate non-specific responses, haematological parameters and growth performance in Mugil cephalus against Photobacterium damselae. Fish Shellfish Immunol 57:379–385
Kim D-H, Austin B (2006) Innate immune responses in rainbow trout (Oncorhynchus mykiss, Walbaum) induced by probiotics. Fish Shellfish Immunol 21:513–524
Kokabi M, Yousefzadi M (2015) Checklist of the marine macroalgae of Iran. Bot Mar 58:307–320
Lahaye M, Robic A (2007) Structure and functional properties of ulvan, a polysaccharide from green seaweeds. Biomacromolecules 8:1765–1774
Liao IC, Chao NH, Tseng CC (2016) Capture and culture of Mugilidae in Taiwan. In: Biology, Ecology and Culture of Grey Mullets (Mugilidae). CRC Press, Boca Raton pp 487–500
Martinez JL (2009) Environmental pollution by antibiotics and by antibiotic resistance determinants. Environ Pollut 157:2893–2902
Moffitt CM, Cajas-Cano L (2014) Blue growth: the 2014 FAO state of world fisheries and aquaculture. Fisheries 39:552–553
Parry RM Jr, Chandan RC, Shahani KM (1965) A rapid and sensitive assay of muramidase. Proc Soc Exp Biol Med 119:384–386
Peixoto MJ, Svendsen JC, Malte H, Pereira LF, Carvalho P, Pereira R, Gonçalves JF, Ozório RO (2016) Diets supplemented with seaweed affect metabolic rate, innate immune, and antioxidant responses, but not individual growth rate in European seabass (Dicentrarchus labrax). J Appl Phycol 28:2061–2071
Pérez MJ, Falqué E, Domínguez H (2016) Antimicrobial action of compounds from marine seaweed. Mar Drugs 14(3):52
Qi H, Zhang Q, Zhao T, Hu R, Zhang K, Li Z (2006) In vitro antioxidant activity of acetylated and benzoylated derivatives of polysaccharide extracted from Ulva pertusa (Chlorophyta). Bioorg Med Chem Lett 16:2441–2445
Qi H, Zhao T, Zhang Q, Li Z, Zhao Z, Xing R (2005) Antioxidant activity of different molecular weight sulfated polysaccharides from Ulva pertusa Kjellm (Chlorophyta). J Appl Phycol 17:527–534
Reverter M, Bontemps N, Lecchini D, Banaigs B, Sasal P (2014) Use of plant extracts in fish aquaculture as an alternative to chemotherapy: current status and future perspectives. Aquaculture 433:50–61
Sakai M (1999) Current research status of fish immunostimulants. Aquaculture 172(1):63–92
Secombes CJ (1990) Isolation of salmonid macrophages and analysis of their killing activity. Techn Fish Immunol 1:137–154
Tabarsa M, Han JH, Kim CY, You SG (2012) Molecular characteristics and immunomodulatory activities of water-soluble sulfated polysaccharides from Ulva pertusa. J Med Food 15:135–144
Thirunavukkarasu R, Pandiyan P, Balaraman D, Jayaraman I, Subaramaniyan K, George EGJ (2015) Enhancements of non-specific immune response in Mugil cephlus by using seaweed extract protection against Vibrio alginolyticus (BRTR07). J Coast Life Med 3:776–786
Trevisani M, Mancusi R, Cecchini M, Costanza C, Prearo M (2017) Detection and characterization of histamine-producing strains of Photobacterium damselae sub sp. damselae isolated from mullets. Vet Sci 4(2):31
Vatsos IN, Rebours C (2015) Seaweed extracts as antimicrobial agents in aquaculture. J Appl Phycol 27:2017–2035
Wassef EA, El-Sayed A-FM, Sakr EM (2013) Pterocladia (Rhodophyta) and Ulva (Chlorophyta) as feed supplements for European seabass, Dicentrarchus labrax L., fry. J Appl Phycol 25:1369–1376
Wells ML, Potin P, Craigie JS, Raven JA, Merchant SS, Helliwell KE, Smith AG, Camire ME, Brawley SH (2017) Algae as nutritional and functional food sources: revisiting our understanding. J Appl Phycol 29:949–982
Yaich H, Garna H, Besbes S, Paquot M, Blecker C, Attia H (2013) Effect of extraction conditions on the yield and purity of ulvan extracted from Ulva lactuca. Food Hydrocoll 31:375–382
Yangthong M, Hutadilok-Towatana N, Thawonsuwan J, Phromkunthong W (2016) An aqueous extract from Sargassum sp. enhances the immune response and resistance against Streptococcus iniae in the Asian sea bass (Lates calcarifer Bloch). J Appl Phycol 28:3587–3598
Zhu D, Wen X, Xuan X, Li S, Li Y (2016) The green alga Ulva lactuca as a potential ingredient in diets for juvenile white spotted snapper Lutjanus stellatus Akazaki. J Appl Phycol 28:703–711
Acknowledgements
The authors gratefully acknowledge the generous support of Iranian Fisheries Sciences Research Institute (IFSRI) for the project no: 0-12-12-9353.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akbary, P., Aminikhoei, Z. Effect of water-soluble polysaccharide extract from the green alga Ulva rigida on growth performance, antioxidant enzyme activity, and immune stimulation of grey mullet Mugil cephalus . J Appl Phycol 30, 1345–1353 (2018). https://doi.org/10.1007/s10811-017-1299-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1299-8