Abstract
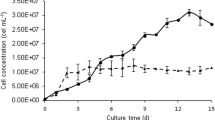
The photoautotrophic co-culture of Chlorella vulgaris and the yeast Rhodotorula glutinis (MP culture) from industrial wastewater was investigated. Cell numbers, biomass, lipid production, and fatty acid content were measured. Owing to co-culture interaction with yeast and microalgae, the MP culture resulted in the highest number of cells (27.73 × 105 cells mL-1) and biomass (0.808 g L−1). Lipid production in the MP culture (117.73 mg L−1) was fourfold higher than that in the photoautotrophic pure culture (23.1 mg L−1). The content of palmitic acid (C16:0) was 24.65%, whereas that of oleic acid (C18:1) was 56.34% in the MP culture, which was higher than in other cultures. The results of this study indicate that MP cultures can be used to effectively support the growth of microorganisms and as an approach for biodiesel production.


Similar content being viewed by others
References
Borowitzka MA (1999) Commercial production of microalgae: ponds, tanks, tubes and fermenters. J Biotechnol 70:313–321
Borowitzka MA (2013) Energy from microalgae: a short history. In: Borowitzka MA, Moheimani NR (eds) Algae for biofuels and energy. Springer, Dordrecht, pp 1–15
Cai S, Hu C, Du S (2007) Comparisons of growth and biochemical composition between mixed culture of alga and yeast and monocultures. J Biosci Bioeng 104(5):391–397
Cheirsilp B, Kitcha S, Torpee S (2012) Co-culture of an oleaginous yeast Rhodotorula glutinis and a microalga Chlorella vulgaris for biomass and lipid production using pure and crude glycerol as a sole carbon source. Ann Microbiol 62:987–993
Chi Z, Zheng Y, Jiang A, Chen S (2011) Lipid production by culturing oleaginous yeast and algae with food waste and municipal wastewater in an integrated process. Appl Biochem Biotech 165:442–453
Cooksey KE, Guckert JB, Williams SA, Collis PR (1987) Fluorometric determination of the neutral lipid content of microalgal cells using Nile Red. J Microbiol Meth 6:333–345
Crabbe E, Nolasco-Hipolito C, Kobayashi G, Sonomoto K, Ishizaki A (2001) Biodiesel production from crude palm oil and evaluation of butanol extraction and fuel properties. Process Biochem 37:65–71
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Fulton L (2004) Biomass and agriculture sustainability markets and policies. International Energy Agency (IEA) biofuels study–interim report: result and key messages so far. IEA France OECD Publication Service; pp.105–112
Jham N, Teles F, Campos G (1982) Use of aqueous HCl/MeOH as esterification reagent for analysis of fatty acids derived from soybean lipids. J Am Oil Chem Soc 59(3):132–133
Kavadia A, Komaitis M, Chevalot I, Blanchard F, Marc I, Aggelis G (2001) Lipids and γ-linolenic acid accumulation in strains of Zygomycetes growing on glucose. J Am Oil Chem Soc 78:341–346
Konar S, Boocock D, Maom V, Liu J (1994) Fuels and chemicals from sewage sludge: 3. hydrocarbon liquids from the catalytic pyrolysis of sewage sludge lipids over activated alumina. Fuel 73(5):642–646
Krawczyk T (1996) Biodiesel—alternative fuel makes in roads but hurdles remain. Inform 7:801–829
Li Q, Wang M-Y (1997) Use food industry waste to produce microbial oil. Sci Technol Food Ind 6:65–69
Liu Z-Y, Wang G-C, Zhou B-C (2008) Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresour Technol 99:4717–4722
Metting B, Pyne J-W (1996) Biologically active compounds from microalgae. Enzym Microb Technol 8:386–394
Ördög V, Stirk WA, Bálint P, Lovász C, Pulz O, van Staden J (2013) Lipid productivity and fatty acid composition in Chlorella and Scenedesmus strains grown in nitrogen-stressed conditions. J Appl Phycol 25:233–243
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Vello V, Phang S-M, Chu W-L, Abdul Majid N, Lim P-E, Loh S-K (2014) Lipid productivity and fatty acid composition-guided selection of Chlorella strains isolated from Malaysia for biodiesel production. J Appl Phycol 26:1399–1413
Xue F, Miao J, Zhang X, Tan T (2010) A new strategy for lipid production by mix cultivation of Spirulina platensis and Rhodotorula glutinis. Appl Biochem Biotechnol 160:498–503
Zhang K, Sun B, She X, Zhao F, Cao Y, Ren D, Lu J (2014) Lipid production and composition of fatty acids in Chlorella vulgaris cultured using different methods: photoautotrophic heterotrophic and pure and mixed conditions. Ann Microbiol 64:1239–1246
Zhu L-Y, Zong M-H, Wu H (2008) Efficient lipids production with Trichosporon fermentans and its use for biodiesel preparation. Bioresour Technol 99:7881–7885
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (Nos. 31571772, 31671963, and 31201339), the National High Technology Research and Development Program of China (863Program, No. 2013AA102205), and the Major State Research Development Program of China (No. 2016YFD0400604).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, K., Zheng, J., Xue, D. et al. Effect of photoautotrophic and heteroautotrophic conditions on growth and lipid production in Chlorella vulgaris cultured in industrial wastewater with the yeast Rhodotorula glutinis . J Appl Phycol 29, 2783–2788 (2017). https://doi.org/10.1007/s10811-017-1168-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1168-5