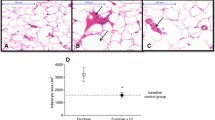
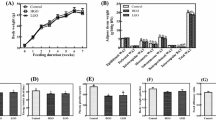
Abstract
Several species of the alga Dunaliella contain high levels of β-carotene. Low dietary β-carotene intake is associated with type 2 diabetes. Dunaliella contains high levels of all-trans and 9-cis isomers of β-carotene and is the best known naturally occurring nutritional source of 9-cis β-carotene. Since vitamin A has been suggested to play a role in glucose and lipid metabolism, we aimed to study the effect of Dunaliella supplementation on diabetes in mice. Ten diabetic db/db mice were fed chow diet fortified with 8 % 9-cis-rich Dunaliella powder. Ten db/db and heterozygous db/+ mice each served as controls and were fed chow diet alone. The control db/db mice developed severe hyperglycemia with fasting glucose levels reaching 400 mg dL−1. Dunaliella significantly inhibited the elevation of plasma glucose (p = 0.007). The area under the curve of the glucose tolerance test was 24 % lower in Dunaliella-treated mice than in the control db/db mice. Triglyceride elevation was significantly lower in the Dunaliella group than in the db/db group (p = 0.007). The mRNA levels of several pro-inflammatory genes in adipose tissue, including monocyte chemotactic protein-1, intercellular adhesion molecule, vascular adhesion molecule-1, receptor-associated protein factor 6, and p-selectin, were elevated in the db/db group as compared to the db/+, whereas their levels were significantly lower in the Dunaliella-treated group. These results suggest that 9-cis β-carotene-rich Dunaliella may inhibit diabetes in db/db mice by reducing inflammation in adipose tissue. This study also emphasizes the importance of β-carotene isomers in our diet.





Similar content being viewed by others
References
Amengual J, Gouranton E, van Helden YG, Hessel S, Ribot J, Kramer E, Kiec-Wilk B, Razny U, Lietz G, Wyss A, Dembinska-Kiec A, Palou A, Keijer J, Landrier JF, Bonet ML, von Lintig J (2011) Beta-carotene reduces body adiposity of mice via BCMO1. PLoS One 6:e20644. doi:10.1371/journal.pone.0020644
Arnlov J, Zethelius B, Riserus U, Basu S, Berne C, Vessby B, Alfthan G, Helmersson J (2009) Serum and dietary beta-carotene and alpha-tocopherol and incidence of type 2 diabetes mellitus in a community-based study of Swedish men: report from the Uppsala Longitudinal Study of Adult Men (ULSAM) study. Diabetologia 52:97–105
Ben-Amotz A, Fishler R (1998) Analysis of carotenoids with emphasis on 9-cis β-carotene in vegetables and fruits commonly consumed in Israel. Food Chem 62:515–520
Berglund O, Frankel BJ, Hellman B (1978) Development of the insulin secretory defect in genetically diabetic (db/db) mouse. Acta Endocrinol (Copenh) 87:543–551
Beydoun MA, Shroff MR, Chen X, Beydoun HA, Wang Y, Zonderman AB (2011) Serum antioxidant status is associated with metabolic syndrome among U.S. adults in recent national surveys. J Nutr 141:903–913
Blumentrath J, Neye H, Verspohl EJ (2001) Effects of retinoids and thiazolidinediones on proliferation, insulin release, insulin mRNA, GLUT 2 transporter protein and mRNA of INS-1 cells. Cell Biochem Funct 19:159–169
Campo-Paysaa F, Marletaz F, Laudet V, Schubert M (2008) Retinoic acid signaling in development: tissue-specific functions and evolutionary origins. Genesis 46:640–656
Davies PJ, Berry SA, Shipley GL, Eckel RH, Hennuyer N, Crombie DL, Ogilvie KM, Peinado-Onsurbe J, Fievet C, Leibowitz MD, Heyman RA, Auwerx J (2001) Metabolic effects of rexinoids: tissue-specific regulation of lipoprotein lipase activity. Mol Pharmacol 59:170–176
Fabregas J, Herrero C, Parafita M, Paz JM, Cabezas B, Abalde J (1988) Decrease in plasma cholesterol, triglycerides and CPK levels in rats fed on the marine microalga Dunaliella tertiolecta. Planta Med 54:109–111
Felipe F, Bonet ML, Ribot J, Palou A (2004) Modulation of resistin expression by retinoic acid and vitamin A status. Diabetes 53:882–889
Freemantle SJ, Spinella MJ, Dmitrovsky E (2003) Retinoids in cancer therapy and chemoprevention: promise meets resistance. Oncogene 22:7305–7315
Frey SK, Vogel S (2011) Vitamin A metabolism and adipose tissue biology. Nutrients 3:27–39
Gouranton E, Thabuis C, Riollet C, Malezet-Desmoulins C, El Yazidi C, Amiot MJ, Borel P, Landrier JF (2011) Lycopene inhibits proinflammatory cytokine and chemokine expression in adipose tissue. J Nutr Biochem 22:642–648
Gustafson B (2010) Adipose tissue, inflammation and atherosclerosis. J Atheroscler Thromb 17:332–341
Harari A, Harats D, Marko D, Cohen H, Barshack I, Kamari Y, Gonen A, Gerber Y, Ben-Amotz A, Shaish A (2008) A 9-cis β-carotene-enriched diet inhibits atherogenesis and fatty liver formation in LDL receptor knockout mice. J Nutr 138:1923–1930
Hebuterne X, Wang XD, Johnson EJ, Krinsky NI, Russell RM (1995) Intestinal absorption and metabolism of 9-cis-β-carotene in vivo: biosynthesis of 9-cis-retinoic acid. J Lipid Res 36:1264–1273
Hessel S, Eichinger A, Isken A, Amengual J, Hunzelmann S, Hoeller U, Elste V, Hunziker W, Goralczyk R, Oberhauser V, von Lintig J, Wyss A (2007) CMO1 deficiency abolishes vitamin A production from β-carotene and alters lipid metabolism in mice. J Biol Chem 282:33553–33561
Kanda H, Tateya S, Tamori Y, Kotani K, Hiasa K, Kitazawa R, Kitazawa S, Miyachi H, Maeda S, Egashira K, Kasuga M (2006) MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J Clin Invest 116:1494–1505
Kane MA, Folias AE, Pingitore A, Perri M, Obrochta KM, Krois CR, Cione E, Ryu JY, Napoli JL (2010) Identification of 9-cis-retinoic acid as a pancreas-specific autacoid that attenuates glucose-stimulated insulin secretion. Proc Natl Acad Sci U S A 107:21884–21889
Koistinen HA, Remitz A, Gylling H, Miettinen TA, Koivisto VA, Ebeling P (2001) Dyslipidemia and a reversible decrease in insulin sensitivity induced by therapy with 13-cis-retinoic acid. Diabetes Metab Res Rev 17:391–395
Kolseth IB, Agren J, Sundvold-Gjerstad V, Lyngstadaas SP, Wang JE, Dahle MK (2012) 9-cis retinoic acid inhibits inflammatory responses of adherent monocytes and increases their ability to induce classical monocyte migration. J Innate Immun 4:176–186
Lalloyer F, Pedersen TA, Gross B, Lestavel S, Yous S, Vallez E, Gustafsson JA, Mandrup S, Fievet C, Staels B, Tailleux A (2009) Rexinoid bexarotene modulates triglyceride but not cholesterol metabolism via gene-specific permissivity of the RXR/LXR heterodimer in the liver. Arterioscler Thromb Vasc Biol 29:1488–1495
Levin AA, Sturzenbecker LJ, Kazmer S, Bosakowski T, Huselton C, Allenby G, Speck J, Kratzeisen C, Rosenberger M, Lovey A et al (1992) 9-cis retinoic acid stereoisomer binds and activates the nuclear receptor RXR alpha. Nature 355:359–361
Lobo GP, Amengual J, Li HN, Golczak M, Bonet ML, Palczewski K, von Lintig J (2010) β,β-carotene decreases peroxisome proliferator receptor gamma activity and reduces lipid storage capacity of adipocytes in a β,β-carotene oxygenase 1-dependent manner. J Biol Chem 285:27891–27899
Manolescu DC, Sima A, Bhat PV (2010) All-trans retinoic acid lowers serum retinol-binding protein 4 concentrations and increases insulin sensitivity in diabetic mice. J Nutr 140:311–316
Mokady S, Avron M, Ben-Amotz A (1990) Accumulation in chick livers of 9-cis versus all-trans β-carotene. J Nutr 120:889–892
Mukherjee R, Strasser J, Jow L, Hoener P, Paterniti JR Jr, Heyman RA (1998) RXR agonists activate PPARalpha-inducible genes, lower triglycerides, and raise HDL levels in vivo. Arterioscler Thromb Vasc Biol 18:272–276
Napoli JL (1999) Retinoic acid: its biosynthesis and metabolism. Prog Nucleic Acid Res Mol Biol 63:139–188
Perry JR, Ferrucci L, Bandinelli S, Guralnik J, Semba RD, Rice N, Melzer D, DIAGRAM Consortium, Saxena R, Scott LJ, McCarthy MI, Hattersley AT, Zeggini E, Weedon MN, Frayling TM (2009) Circulating β-carotene levels and type 2 diabetes—cause or effect? Diabetologia 52:2117–2121
Shaish A, Harari A, Hananshvili L, Cohen H, Bitzur R, Luvish T, Ulman E, Golan M, Ben-Amotz A, Gavish D, Rotstein Z, Harats D (2006) 9-cis β-carotene-rich powder of the alga Dunaliella bardawil increases plasma HDL-cholesterol in fibrate-treated patients. Atherosclerosis 189:215–221
Shulman AI, Mangelsdorf DJ (2005) Retinoid X receptor heterodimers in the metabolic syndrome. N Engl J Med 353:604–615
Stephensen CB, Rasooly R, Jiang X, Ceddia MA, Weaver CT, Chandraratna RA, Bucy RP (2002) Vitamin A enhances in vitro Th2 development via retinoid X receptor pathway. J Immunol 168:4495–4503
Takayanagi K (2011) Prevention of adiposity by the oral administration of β-cryptoxanthin. Front Neurol 2:67. doi:10.3389/fneur.2011.00067
Tourniaire F, Gouranton E, von Lintig J, Keijer J, Bonet ML, Amengual J, Lietz G, Landrier JF (2009) β-Carotene conversion products and their effects on adipose tissue. Genes Nutr 4:179–187
Wang XD, Krinsky NI, Benotti PN, Russell RM (1994) Biosynthesis of 9-cis-retinoic acid from 9-cis-β-carotene in human intestinal mucosa in vitro. Arch Biochem Biophys 313:150–155
Woo MN, Jeon SM, Kim HJ, Lee MK, Shin SK, Shin YC, Park YB, Choi MS (2010) Fucoxanthin supplementation improves plasma and hepatic lipid metabolism and blood glucose concentration in high-fat fed C57BL/6N mice. Chem Biol Interact 186:316–322
Yen TT, Allan JA, Yu PL, Acton MA, Pearson DV (1976) Triacylglycerol contents and in vivo lipogenesis of ob/ob, db/db and Avy/a mice. Biochim Biophys Acta 441:213–220
Ziouzenkova O, Orasanu G, Sharlach M, Akiyama TE, Berger JP, Viereck J, Hamilton JA, Tang G, Dolnikowski GG, Vogel S, Duester G, Plutzky J (2007a) Retinaldehyde represses adipogenesis and diet-induced obesity. Nat Med 13:695–702
Ziouzenkova O, Orasanu G, Sukhova G, Lau E, Berger JP, Tang G, Krinsky NI, Dolnikowski GG, Plutzky J (2007b) Asymmetric cleavage of β-carotene yields a transcriptional repressor of retinoid X receptor and peroxisome proliferator-activated receptor responses. Mol Endocrinol 21:77–88
Acknowledgments
We thank Dr. Esfir Ulman for technical help and to Dr. Dorit Samocha-Bonnet for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial interest
Aviv Shaish, Dror Harats, and Ayelet Harari have received a grant from Nikken Sohonsha. Ami Ben-Amotz is a consultant of Nikken Sohonsha.
Rights and permissions
About this article
Cite this article
Harari, A., Harats, D., Marko, D. et al. Supplementation with 9-cis β-carotene-rich alga Dunaliella improves hyperglycemia and adipose tissue inflammation in diabetic mice. J Appl Phycol 25, 687–693 (2013). https://doi.org/10.1007/s10811-012-9903-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-012-9903-4