Abstract
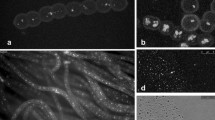
We investigated the formation of lipid bodies in the microalga Chlorella vulgaris CCALA 256 under lipid-induction conditions in autotrophically grown cultures. We found that cell division ceased after depletion of nitrates from the growth medium within the first days of cultivation. The growth of non-dividing cells subsequently led to the rapid accumulation of lipids. We describe in detail both biogenesis and mobilization of lipid bodies using fluorescence and transmission electron microscopy. Small lipid bodies fused very soon after their creation in the cytosol, forming, eventually, one huge lipid body. In stationary growth phase, lipids were present in the form of a large lipid body occupying most of the cell volume. After replenishment of nitrogen, lipid content decreased rapidly and, within 24 hours, the large lipid body was fragmented into smaller ones. This mobilization of the cellular lipid store occurred independently of light.






Similar content being viewed by others
References
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Campbell PK, Beer T, Batten D (2011) Life cycle assessment of biodiesel production from microalgae in ponds. Bioresour Technol 102:50–56
Davidi L, Katz A, Pick U (2012) Characterization of major lipid droplet proteins from Dunaliella. Planta 236:19–33
Eastmond PJ (2006) SUGAR-DEPENDENT1 encodes a patatin domain triacylglycerol lipase that initiates storage oil breakdown in germinating Arabidopsis seeds. Plant Cell 18:665–675
Eltgroth ML, Watwood RL, Wolfe GV (2005) Production and cellular localization of neutral long-chain lipids in the haptophyte algae Isochrysis galbana and Emiliania huxleyi. J Phycol 41:1000–1009
Gallagher BJ (2011) The economics of producing biodiesel from algae. Renew Energ 36:158–162
Goodson C, Roth R, Wang ZT, Goodenough U (2011) Structural correlates of cytoplasmic and chloroplast lipid body synthesis in Chlamydomonas reinhardtii and stimulation of lipid body production with acetate boost. Eukaryot Cell 10:1592–1606
Graham IA, Denby KJ, Leaver CJ (1994) Carbon catabolite repression regulates glyoxylate cycle gene expression in higher plants. Plant Cell 6:761–772
Halim R, Gladman B, Danquah MK, Webley PA (2011) Oil extraction from microalgae for biodiesel production. Bioresour Technol 102:178–185
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54:621–639
Illman AM, Scragg AH, Shales SW (2000) Increase in Chlorella strains calorific values when grown in low nitrogen medium. Enzym Microb Technol 27:631–635
Khozin-Goldberg I, Shrestha P, Cohen Z (2005) Mobilization of arachidonyl moieties from triacylglycerols into chloroplastic lipids following recovery from nitrogen starvation of the microalga Parietochloris incisa. Biochim Biophys Acta 1738:63–71
Krohn BJ, McNeff CV, Yan B, Nowlan D (2011) Production of algae-based biodiesel using the continuous catalytic Mcgyan® process. Bioresour Technol 102:94–100
Larson TR, Harrison PJ (1997) Storage lipid metabolism during nitrogen assimilation in a marine diatom. In: Williams JP (ed) Physiology, biochemistry, and molecular biology of plant lipids. Kluwer Academic, Dordrecht, pp 256–258
Lee DH (2011) Algal biodiesel economy and competition among bio-fuels. Bioresour Technol 102:43–49
Li Y, Lian S, Tong D, Song R, Yang W, Fan Y, Qing R, Hua C (2011a) One-step production of biodiesel from Nannochloropsis sp. on solid base Mg–Zr catalyst. Appl Energy 88:3313–3317
Li Y, Han D, Sommerfeld M, Hu Q (2011b) Photosynthetic carbon partitioning and lipid production in the oleaginous microalga Pseudochlorococcum sp. (Chlorophyceae) under nitrogen-limited conditions. Bioresour Technol 102:123–129
Liu CP, Lin LP (2001) Ultrastructural study and lipid formation of Isochrysis sp. CCMP 1324. Bot Bull Acad Sin 42:207–214
Lv J-M, Cheng L-H, Xu X-H, Zhang L, Chen H-L (2010) Enhanced lipid production of Chlorella vulgaris by adjustment of cultivation conditions. Bioresour Technol 101:6797–6804
Makri A, Bellou S, Birkou M, Papatrehas K, Dolapsakis NP, Bokas D, Papanikolaou S, Aggelis G (2011) Lipid synthesized by micro-algae grown in laboratory- and industrial-scale bioreactors. Eng Life Sci 11:52–58
Martin T, Oswald O, Graham IA (2002) Arabidopsis seedling growth, storage lipid mobilization, and photosynthetic gene expression are regulated by carbon:nitrogen availability. Plant Physiol 128:472–481
Merchant SS, Kropat J, Liu B, Shaw J, Warakanont J (2011) TAG, You're it! Chlamydomonas as a reference organism for understanding algal triacylglycerol accumulation. Curr Opin Biotechnol 23:1–12
Merzlyak MN, Chivkunova OB, Gorelova OA, Reshetnikova IV, Solovchenko AE, Khozin-Goldberg I, Cohen Z (2007) Effect of nitrogen starvation on optical properties, pigments, and arachidonic acid content of the unicellular green alga Parietochloris incisa (Trebouxiophyceae, Chlorophyta). J Phycol 43:833–843
Mock T, Kroon BMA (2002) Photosynthetic energy conversion under extreme conditions—I: important role of lipids as structural modulators and energy sink under N-limited growth in Antarctic sea ice diatoms. Phytochemistry 61:41–51
Moellering ER, Benning C (2010) RNAi silencing of a major lipid droplet protein affects lipid droplet size in Chlamydomonas reinhardtii. Eukaryot Cell 9:97–106
Mutlu YB, Işık O, Uslu L, Koç K, Durmaz Y (2011) The effects of nitrogen and phosphorus deficiencies and nitrite addition on the lipid content of Chlorella vulgaris (Chlorophyceae). Afr J Biotechnol 10:453–456
Parthibane V, Rajakumari S, Venkateshwari V, Iyappan R, Rajasekharan R (2012) Oleosin is bifunctional enzyme that has both monoacylglycerol acyltransferase and phospholipase activities. J Biol Chem 287:1946–1954
Patil PD, Gude VG, Mannarswamy A, Deng S, Cooke P, Munson-McGee S, Rhodes I, Lammers P, Nirmalakhandan N (2011) Optimization of direct conversion of wet algae to biodiesel under supercritical methanol conditions. Bioresour Technol 102:118–122
Peled E, Leu S, Zarka A, Weiss M, Pick U, Khozin-Goldberg I, Boussiba S (2011) Isolation of a novel oil globule protein from the green alga Haematococcus pluvialis (Chlorophyceae). Lipids 46:851–861
Pruvost J, Van Vooren G, Le Gouic B, Couzinet-Mossion A, Legrand J (2011) Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour Technol 102:150–158
Přibyl P, Cepák V, Zachleder V (2012a) Production of lipids in 10 strains of Chlorella and Parachlorella, and enhanced lipid productivity in Chlorella vulgaris. Appl Microbiol Biotechnol 94:549–561
Přibyl P, Eliáš M, Cepák V, Lukavský J, Kaštánek P (2012b) Zoosporogenesis, morphology, ultrastructure, pigment composition and phylogenetic position of Trachydiscus minutus (Eustigmatophyceae, Heterokontophyta). J Phycol 48:231–242
Ramazanov A, Ramazanov Z (2006) Isolation and characterization of a starchless mutant of Chlorella pyrenoidosa STL-PI with a high growth rate, and high protein and polyunsaturated fatty acid content. Phycol Res 54:255–259
Ratledge C (2004) Fatty acid biosynthesis in microorganisms being used for single cell oil production. Biochimie 86:807–815
Rodolfi L, Chini Zitelli G, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112
Roessler PG (1990) Environmental control of glycerolipid metabolism in microalgae: commercial implications and future research directions. J Phycol 26:393–399
Siaut M, Cuine S, Cagnon C, Fessler B, Nguyen M, Carrier P, Beyly A, Beisson F, Triantaphylides C, Li-Beisson YH, Peltier G (2011) Oil accumulation in the model green alga Chlamydomonas reinhardtii: characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnol 11:7–21
Singh A, Olsen SI (2011) A critical review of biochemical conversion, sustainability and life cycle assessment of algal biofuels. Appl Energy 88:3548–3555
Solovchenko AE, Chivkunova OB, Maslova IP (2011) Pigment composition, optical properties, and resistance to photodamage of the microalga Haematococcus pluvialis cultivated under high light. Russ J Plant Physiol 58:9–17
Solovchenko AE (2012) Physiological role of neutral lipid accumulation in eukaryotic microalgae under stresses. Russ J Plant Physiol 59:167–176
Spoehr HA, Milner HW (1949) The chemical composition of Chlorella; effect of environmental conditions. Plant Physiol 24:120–149
Subhadra BG (2010) Sustainability of algal biofuel production using integrated renewable energy park (IREP) and algal biorefinery approach. Energ Pol 38:5892–5901
Wang ZT, Ullrich N, Joo S, Waffenschmidt S, Goodenough U (2009) Algal lipid bodies: stress induction, purification, and biochemical characterization in wild-type and starchless Chlamydomonas reinhardtii. Eukaryot Cell 8:1856–1868
Work VH, Radakovits R, Jinkerson RE, Meuser JE, Elliott LG, Vinyard DJ, Laurens LML, Dismukes GC, Posewitz MC (2010) Increased lipid accumulation in the Chlamydomonas reinhardtii sta7–10 starchless isoamylase mutant and increased carbohydrate synthesis in complemented strains. Eukaryot Cell 9:1251–1261
Xiong W, Liu L, Wu C, Yang C, Wu Q (2010) 13C-tracer and gas chromatography–mass spectrometry analyses reveal metabolic flux distribution in the oleaginous microalga Chlorella protothecoides. Plant Physiol 154:1001–1011
Yu Y, Chen BL, You WL (2007) Identification of the alga known as Nannochloropsis Z-1 isolated from a prawn farm in Hainan, China as Chlorella. World J Microbiol Biotechnol 23:207–210
Zachleder V, Šetlík I (1982) Effect of irradiance on the course of RNA synthesis in the cell cycle of Scenedesmus quadricauda. Biol Plant 24:341–353
Zheng H, Yin J, Gao Z, Huang H, Ji X, Dou C (2011) Disruption of Chlorella vulgaris cells for the release of biodiesel-producing lipids: a comparison of grinding, ultrasonication, bead milling, enzymatic lysis, and microwaves. Appl Biochem Biotechnol 164:1215–1224
Acknowledgments
We thank Prof. John Brooker for editing the English. This work was supported by the Ministry of Education and Youth of the Czech Republic, project no: 1M0571, a long-term research development project no. RVO 67985939, and by the Japan Science and Technology Agency, project CRST.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Přibyl, P., Cepák, V. & Zachleder, V. Production of lipids and formation and mobilization of lipid bodies in Chlorella vulgaris . J Appl Phycol 25, 545–553 (2013). https://doi.org/10.1007/s10811-012-9889-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-012-9889-y