Abstract
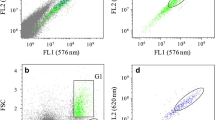
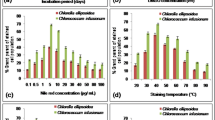
High-throughput screening of microalgae for use as a potential feedstock for biodiesel requires a reliable method for the rapid detection of intracellular neutral lipid content. In this study, we report a modified and improved Nile Red (NR) fluorescence staining procedure for use as a rapid and sensitive screening tool to estimate levels of intracellular neutral lipid in the picopleustonic microalgae, Nannochloropsis sp. Addition of either glycerol or dimethyl sulfoxide (DMSO) into microalgae cultures greatly enhances lipid staining efficiency and increases the fluorescence intensity of stained cells. The optimized procedure requires glycerol and DMSO at the concentration of 0.1 and 0.165 g mL−1, respectively, for peak fluorescence in a live culture of Nannochloropsis sp. Incubation for 5 min for glycerol-NR staining and 10 min for DMSO-NR staining at room temperature, in darkness, is used for the NR concentration of 0.3 and 0.7 μg mL−1 for glycerol and DMSO, respectively. For the selection of lipid-rich cells of Nannochloropsis sp. using flow cytometric cell sorting, the glycerol-NR procedure is recommended as glycerol, unlike DMSO, does not inhibit subsequent growth of sorted cells.






Similar content being viewed by others
References
Chen W, Zhang CW, Song LR, Sommerfeld M, Hu Q (2009) A high throughput Nile Red method for quantitative measurement of neutral lipids in microalgae. J Microbiol Meth 77:41–47
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306
Chiu S-Y, Kao C-Y, Tsai M-T, Ong S-C, Chen C-H, Lin C-S (2009) Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresour Technol 100:833–838
Cooksey KE, Guckert JB, Williams SA, Callis PR (1987) Fluorometric-determination of the neutral lipid-content of microalgal cells using Nile Red. J Microbiol Meth 6:333–345
de la Jara A, Mendoza H, Martel A, Molina C, Nordstron L, de la Rosa V et al (2003) Flow cytometric determination of lipid content in a marine dinoflagellate, Crypthecodinium cohnii. J Appl Phycol 15:433–438
Elsey D, Jameson D, Raleigh B, Cooney MJ (2007) Fluorescent measurement of microalgal neutral lipids. J Microbiol Meth 68:639–642
Gouveia L, Oliveira AC (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274
Greenspan P, Fowler SD (1985) Spectrofluorometric studies of the lipid probe, Nile Red. J Lipid Res 26:781–789
Guillard RRL (2005) Purification methods for microalgae. In: Andersen RA (ed) Algal culturing techniques. Elsevier, Amsterdam, pp 117–132
Lee SJ, Yoon BD, Oh HM (1998) Rapid method for the determination of lipid from the green alga Botryococcus braunii. Biotechnol Tech 12:553–556
Miao X, Wu Q (2006) Biodiesel production from heterotrophic microalgal oil. Bioresour Technol 97:841–846
Pienkos PT, Darzins A (2009) The promise and challenges of microalgal-derived biofuels. Biofuels, Bioprod Biorefin 3:431–440
Poncet JM, Veron B (2003) Cryopreservation of the unicellular marine alga, Nannochloropsis oculata. Biotechnol Lett 25:2017–2022
Priscu JC, Priscu LR, Palmisano AC, Sullivan CW (1990) Estimation of neutral lipid-level in Antartic sea ice microalgae by Nile Red fluorescence. Antarct Sci 2:149–155
Rodolfi L, Zittelli GC, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112
Rosenberg JN, Oyler GA, Wilkinson L, Betenbaugh MJ (2008) A green light for engineered algae: redirecting metabolism to fuel a biotechnology revolution. Curr Opin Biotechnol 19:430–436
Sensen C, Heimann K, Melkonian M (1993) The production of clonal and axenic cultures of microalgae using fluorescence-activated cell sorting. Eur J Phycol 28:93–97
Sheehan J, Dunahay T, Benemann J, Roessler P (1998) Look back at the U.S. Department of Energy’s Aquatic Species Program: Biodiesel from Algae. NREL/TP-580-24190, National Renewable Energy Lab., Golden, CO. Department of Energy, Washington, USA
Simonetti O, Hoogstraate AJ, Bialik W, Kempenaar JA, Schrijvers A, Bodde HE et al (1995) Visualization of diffusion pathways across the stratum corneum of native and in-vitro reconstructed epidermis by confocal laser scanning microscopy. Arch Dermatol Res 287:465–473
Solomon J, Hand R Jr, Mann R (1986) Ultrastructural and flow cytometric analyses of lipid accumulation in microalgae. SERI/STR-231-3089, Oak Ridge National Lab., Solar Energy Research Inst., Golden, CO, USA
Suen Y, Hubbard JS, Holzer G, Tornabene TG (1987) Total lipid production of the green-alga Nannochloropsis sp. QII under different nitrogen regimes. J Phycol 23:289–296
Tzovenis I, Triantaphyllidis G, Naihong X, Chatzinikolaou E, Papadopoulou K, Xouri G et al (2004) Cryopreservation of marine microalgae and potential toxicity of cryoprotectants to the primary steps of the aquacultural food chain. Aquaculture 230:457–473
Veldhuis MJW, Cucci TL, Sieracki ME (1997) Cellular DNA content of marine phytoplankton using two new fluorochromes: taxonomic and ecological implications. J Phycol 33:527–541
Zou N, Zhang CW, Cohen Z, Richmond A (2000) Production of cell mass and eicosapentaenoic acid (EPA) in ultrahigh cell density cultures of Nannochloropsis sp. (Eustigmatophyceae). Eur J Phycol 35:127–133
Acknowledgments
Funding of this research project was provided by Agency for Science, Technology and Research of Singapore (A* STAR). We also thank Toh Kok Tee (NUMI), Yeo Yin Sheng Wilson (TMSI), and Dr. Balasubramanian Sivaloganathan for technical supports.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Doan, TT.Y., Obbard, J.P. Improved Nile Red staining of Nannochloropsis sp.. J Appl Phycol 23, 895–901 (2011). https://doi.org/10.1007/s10811-010-9608-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-010-9608-5