Abstract
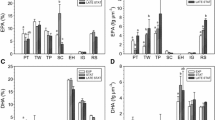
Porphyridium cruentum was grown in 10 L batch culture at 18°C, pH 8.0 and 28‰ salinity. The cells were harvested in the stationary phase and the fatty acid composition analysed by GC and tocopherol content by HPLC. A total of 14 fatty acids were identified including saturated fatty acids (13:0, 14:0, 14:0 iso, 15:0, 16:0, 16:0iso) and monounsaturated fatty acids (MUFAs; 16:1(n-7), 18:1(n-7), 18:1(n-9). Polyunsaturated fatty acids (PUFAs) were the predominant fatty acids detected, reaching 43.7% of total fatty acids in the stationary phase of culture. Among the PUFAs, eicosapentaenoic acid (EPA, 20:5(n-3)) was dominant (25.4%), followed by 12.8% arachidonic acid (AA, 20:4(n-6)). α-Tocopherol and γ-tocopherol contents were 55.2 μg g−1 dry weight and 51.3 μg g−1 dry weight respectively.

Similar content being viewed by others
References
Batista I, Martins MFG (1991) Estudos de floculação/coagulação de Chlorella sp. usando quitosana e vários catiões polivalentes. Seminário sobre aquacultura mediterrânica Portugal, vol 19. INIP, Lisbon pp 53–62
Becker EW (1994) Microalgae biotechnology and microbiology. Cambridge University Press, Cambridge
Borowitzka MA, Borowitzka LJ (eds) (1988) Micro-algal biotechnology. Cambridge University Press, Cambridge, p 477
Brown MR, Mular M, Miller I, Farmer C, Trenerry C (1999) The vitamin content of microalgae used in aquaculture. J Appl Phycol 11:247–255
Carballo-Cárdenas EC, Tuan PM, Jansen M, Wijffels RH (2003) Vitamin E (α-tocopherol) production by the marine microalgae Dunaliella tertiolecta and Tetraselmis suecica in batch cultivation. Biomolec Eng 20:139–147
Chen JY, Latshaw JD, Lee HO, Min DB (1998) α-Tocopherol content and oxidative stability of egg yolk as related to dietary α-tocopherol. J Food Sci 63:919–922
Chiu A, Kimball AB (2003) Topical vitamins, minerals and botanical ingredients as modulators of environmental and chronological skin damage. Br J Dermatol 149:681–691
Cohen Z (1991) Preparation of eicosapentanoienoic acid (EPA) concentrate from Porphyridium cruentum. J Am Oil Chem Soc 68:16–19
Cohen Z (1999) Porphyridium cruentum. In: Cohen Z (ed) Chemicals from microalgae. Taylor & Francis, London, pp 41–56
Cohen Z, Vonshak A, Richmond A (1988) Effect of environmental conditions on fatty acid composition of the red alga Porphyridium cruentum: correlation to growth rate. J Phycol 24:328–332
Fuentes MM, Fernandez GG, Perez JA, Guerrero JJ (2000) Biomass nutrient profiles of the microalga Porphyridium cruentum. Food Chem 70:345–353
Gimenez A, Gonzalez MJI, Medina AR, Grima EM, Salas SG, Cerdan LE (1998) Downstream processing and purification of eicosapentaenoic (20:5n-3) and arachidonic (20:4n-6) acids from the microalgae Porphyridium cruentum. Bioseparation 7:89–99
Ginzberg A, Cohen M, Sod-Moriah UA, Shany S, Rosenstrauch A, Arad SM (2000) Chickens fed with biomass of the red microalga Porphyridium sp. have reduced blood cholesterol level and modified fatty acid composition in egg yolk. J Appl Phycol 12:325–330
Gómez-Coronado DJM, Ibãnez E, Rupérez FJ, Barbas C (2004) Tocopherol measurement in edible products of vegetable origin. J Chromatogr A 1054:227–233
Guil-Guerrero JL, El-Hassan B, Fuentes MMR (2001) Eicosapentaenoic and arachidonic acids purification from the red microalga Porphyridium cruentum. Bioseparation 9:299–306
Huo J, Nelis HJ, Lavens P, Sorgeloos P, De Leenheer AP (1997) Determination of vitamin E in microalgae using HPLC with fluorescence detection. J Chromatogr A 782:63–68
Iwamoto H, Sato S (1986) EPA production by freshwater algae. J Am Oil Chem Soc 63:434
Lepage G, Roy CC (1986) Direct transesterification of all classes of lipids in a one-step reaction. J Lipid Res 27:114–119
Renaud SM, Zhou HC, Parry DL, Thinh LV, Woo KC (1995) Effect of temperature on the growth, total lipid content and fatty acid composition of recently isolated tropical microalgae Isochrysis sp., Nitzschia closterium, Nitzschia paleacea, and commercial species Isochrysis sp. (clone T. ISO). J Appl Phycol 7:595–602
Richmond A (1986) Cell response to environmental factors. In: Richmond A (ed) Handbook of microalgal mass culture. CRC Press, Boca Raton, pp 69–99
Satu N, Murata N (1980) Temperature shift-induced responses in lipids in the blue-green alga, Anabaena variabilis. The central role of diacylmonogalactosylglycerol in thermo adaption. Biochim Biophys Acta 619:353–365
Schlegel HG (1985) Allgemeine Microbiologie. Thieme, Stuttgart, p 571
Sing S, Arad S, Richmond A (2000) Extracellular polysaccharide production in outdoor mass cultures of Porpyridium sp. in flat plate glass reactors. J Appl Phycol 12:269–275
Singh G, Chandra RK (1988) Biochemical and cellular effects of fish and fish oils. Prog Food Nutr 12:371–419
Sukenik A, Carmeli Y, Berner T (1989) Regulation of fatty acid composition by irradiance level in the eustigmatophyte Nannochloropsis sp. J Phycol 25:686–692
Thompson PA, Guo M, Harrison PJ, Whyte JNC (1992) Effects of variation in temperature. I. On the fatty acid composition of eight species of marine phytoplankton. J Phycol 28:488–497
Vismara R, Vestri S, Kusmic C, Brarsanti L, Gualtieri P (2003) Natural vitamin E enrichment of Artemia salina fed freshwater and marine microalgae. J Appl Phycol 15:75–80
Vonshak A (1988) Porphyridium. In: Borowitzka MA, Borowitzka LJ (eds) Micro-algal biotechnology. Cambridge University Press, Cambridge, pp 122–134
Acknowledgements
The authors would like to thank Prof. Amos Richmond for his valuable advice and Prof. Michael Borowitzka for the time he spent in revising and correcting some aspects of the article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Durmaz, Y., Monteiro, M., Bandarra, N. et al. The effect of low temperature on fatty acid composition and tocopherols of the red microalga, Porphyridium cruentum . J Appl Phycol 19, 223–227 (2007). https://doi.org/10.1007/s10811-006-9127-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-006-9127-6