Abstract
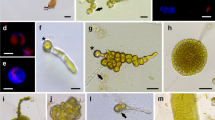
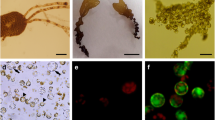
Protoplasts were isolated from tissue fragments (<1 mm2) of three Philippine cultivars of Kappaphycus alvarezii: the giant cultivar, cultivar L and Bohol wild type, by enzymatic dissolution of cell walls. Yields of viable protoplasts from young and old thalli (apical, middle, basal segments) were compared at various temperatures, duration of treatment and pH using eight combinations of commercial enzymes (abalone acetone powder and cellulase), and prepared extracts from fresh viscera of abalone (Haliotis asinina) and a terrestrial garden snail. Isolated protoplasts were grown in various culture media, temperatures, photoperiods and irradiance values to determine the conditions that favor germination and growth.
Protoplast yields in tissues treated with commercial enzymes and the garden snail extract were lower than those obtained in tissues treated with fresh abalone extracts. Generally, the number of viable protoplasts increased with duration of enzyme treatment at 25 °C with a maximum yield of 8.2 × 103 g−1 tissue at 48 h. Yields were consistently higher in all cultivars at pH 6.1. The yields were also high from the middle segments of the giant cultivar (3.7 × 103 g−1 tissue) and Bohol wild type (4.5 × 103 g−1 tissue) treated with fresh abalone extract, and from basal segments of cultivar L and tissues treated with garden snail extract. The germination rate of protoplasts was highest (39.8%) at 25 °C and 20 μmol photon m−2 s−1, using a 12:12 light dark photoperiod. The filament was 3.7 mm long by Day 5. These findings are relevant to developing cultures from protoplasts for genetic or strain improvement of K. alvarezii cultivars.
Similar content being viewed by others
References
Chen LCM (1987) Protoplast morphogenesis of Porphyra leucosticta in culture. Bot. mar. 30: 399–403.
Chen LCM, Craigie JS, Xie ZK (1994) Protoplast production from Porphyra linearis using a simplified agarase procedure capable of commercial application. J. appl. Phycol. 6: 35–39.
Chen YC, Chiang YM (1994) Development of protoplasts from Grateloupia sparsa and G. filicina (Halymeniaceae, Rhodophyta). Bot. mar. 37: 361–366.
Cheney DP (1999) Strain improvement of seaweeds through genetic manipulation: Current status. World Aquaculture 30: 55–56; 66–67.
Cheney DP, Luistro AH, Bradley PM (1987) Carrageenan analysis of tissue cultures and whole plants of Agardhiella subulata. Hydrobiologia 151/152: 161–166.
Dawes CJ, Lluisma AO, Trono GC (1994) Laboratory and field growth studies of commercial strains of Eucheuma denticulatum and Kappaphycus alvarezii in the Philippines. J. appl. Phycol. 6: 21–24.
Fleurence J (1999) The enzymatic degradation of algal cell walls: A useful approach for improving protein accessibility? J. appl. Phycol. 11: 313–314.
Hurtado AQ, Cheney DP (2003) Propagule production of Eucheuma denticulatum (Burman) Collins et Harvey by tissue culture. Bot. mar. 46: 338–341.
Lechat H, Amat M, Mazoyer J, Gallant DJ, Buleon A, Lahaye M (1997) Cell wall composition of the carrageenophyte Kappaphycus alvarezii (Gigartinales, Rhodophyta) partitioned by wet sieving. J. appl. Phycol. 9: 565–572.
Liu QY, Chen LC-M, Taylor ARA (1992) Ultrastructure of cell wall regeneration by isolated protoplasts of Palmaria palmata (Rhodophyta). Bot. mar. 35: 21–33.
Liu XW, Gordon ME (1987) Tissue and cell culture of New Zealand Pterocladiaand Porphyra species. Hydrobiologia 151/152: 147–154.
Packer MA (1994) Protoplast formation from single cells and small tissue fragments of wild Porphyra fronds (Rhodophyta). Bot. mar. 37: 101–108.
Polne-Fuller M, Gibor A (1984) Development studies in Porphyra. I. Blade differentiation in Porphyra perforata as expressed by morphology, enzymatic digestion and protoplast regeneration. J. Phycol. 20: 609–619.
Polne-Fuller M, Gibor A (1987) Calluses and callus-like growth in seaweeds: Induction and culture. Hydrobiologia 151/152: 131–138.
Polne-Fuller M, Gibor A (1988) Tissue culture of seaweeds. In Bird KT, Benson PH (eds). Seaweed Cultivation for Renewable Resources. Elsevier Publication, Netherlands. pp. 219–239.
Trono GC, Lluisma AO, Montaño MNE (2000) Primer on Farming and Strain Selection of Kappaphycus and Eucheumain the Philippines. PCAMRD, UP-MSI and UNDP.
Vreeland V, Kloareg B (2000) Cell wall biology in red algae: Divide and conquer. J. Phycol. 36: 793–797.
Vreeland V, Zablackis E, Laetsch WM (1992) Monoclonal antibodies as molecular markers for the intercellular and cell wall distribution of carrageenan epitopes in Kappaphycus (Rhodophyta) during tissue development. J. Phycol. 28: 328– 342.
Zablackis E, Vreeland V, Doboszewski B, Laetsch WM (1991) Differential localization of carrageenan gelling sequences in Kappaphycus alvarezii var. tambalang (Rhodophyta) with FITC-conjugated carrageenan oligosaccharides. J. Phycol. 27: 241–248.
Zablackis E, Vreeland V, Kloareg B (1993) Isolation of protoplasts from Kappaphycus alvarezii var. tambalang (Rhodophyta) and secretion of iota-carrageenan fragment by cultured cells. J. Exp. Bot. 44 (266): 1515–1522.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salvador, R.C., Serrano, A.E. Isolation of protoplasts from tissue fragments of Philippine cultivars of Kappaphycus alvarezii (Solieriaceae, Rhodophyta). J Appl Phycol 17, 15–22 (2005). https://doi.org/10.1007/s10811-005-5516-5
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10811-005-5516-5