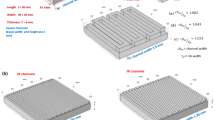
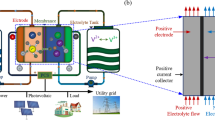
Abstract
In this study, we have investigated the effects of varying flow channel depths and addition of various channel obstructions on the electrochemical performance and pumping power requirements of vanadium redox flow batteries (VRFBs). Specifically, 3D-printed ramps and prismatic obstructions were inserted into the channels of interdigitated flow field (IDFF) and parallel flow field (PFF) designs to observe the effect of non-uniform channel depth on the mass transport properties of open- and closed-ended flow channels. Results were compared with conventional flow field geometries. Integration of ramps into the closed-ended (i.e., IDFF) flow channels resulted in 15% improvement in peak power density (PPD) at a flow rate of 50 mL min−1. Addition of ramps to IDFF has also resulted in a significant 40% drop in required pumping pressure due to guided and gradual delivery of the electrolyte to the electrode plane. In addition, the effects of varying channel depths in open-ended (i.e., PFF) channels were found to be much more drastic with improvements in PPD up to 150%. Overall, findings of this study highlight the significance of varying channel depths on improving the mass transport characteristics of VRFBs and offer an alternative approach for design of high-performance flow cells.
Graphical Abstract







Similar content being viewed by others
References
Weber AZ, Mench MM, Meyers JP, Ross PN, Gostick JT, Liu Q (2011) Redox flow batteries: a review. J Appl Electrochem 41(10):1137. https://doi.org/10.1007/s10800-011-0348-2
Soloveichik GL (2015) Flow batteries: current status and trends. Chem Rev 115(20):11533–11558. https://doi.org/10.1021/cr500720t
Arenas LF, Ponce de León C, Walsh FC (2017) Engineering aspects of the design, construction and performance of modular redox flow batteries for energy storage. J Energy Storage 11:119–153. https://doi.org/10.1016/j.est.2017.02.007
Boettcher PA, Agar E, Dennison CR, Kumbur EC (2016) Modeling of ion crossover in vanadium redox flow batteries: a computationally-efficient lumped parameter approach for extended cycling. J Electrochem Soc 163(1):A5244–A5252. https://doi.org/10.1149/2.0311601jes
Knehr KW, Agar E, Dennison CR, Kalidindi AR, Kumbur EC (2012) A transient vanadium flow battery model incorporating vanadium crossover and water transport through the membrane. J Electrochem Soc 159(9):A1446–A1459. https://doi.org/10.1149/2.017209jes
Darling RM, Gallagher KG, Kowalski JA, Ha S, Brushett FR (2014) Pathways to low-cost electrochemical energy storage: a comparison of aqueous and nonaqueous flow batteries. Energy Environ Sci 7(11):3459–3477. https://doi.org/10.1039/c4ee02158d
Viswanathan V, Crawford A, Stephenson D, Kim S, Wang W, Li B, Coffey G, Thomsen E, Graff G, Balducci P, Kintner-Meyer M, Sprenkle V (2014) Cost and performance model for redox flow batteries. J Power Sources 247:1040–1051. https://doi.org/10.1016/j.jpowsour.2012.12.023
Ke X, Prahl JM, Alexander JID, Wainright JS, Zawodzinski TA, Savinell RF (2018) Rechargeable redox flow batteries: flow fields, stacks and design considerations. Chem Soc Rev 47(23):8721–8743. https://doi.org/10.1039/c8cs00072g
Dennison CR, Agar E, Akuzum B, Kumbur EC (2016) Enhancing mass transport in redox flow batteries by tailoring flow field and electrode design. J Electrochem Soc 163(1):A5163–A5169. https://doi.org/10.1149/2.0231601jes
Aaron DS, Liu Q, Tang Z, Grim GM, Papandrew AB, Turhan A, Zawodzinski TA, Mench MM (2012) Dramatic performance gains in vanadium redox flow batteries through modified cell architecture. J Power Sources 206:450–453. https://doi.org/10.1016/j.jpowsour.2011.12.026
Pezeshki AM, Clement JT, Veith GM, Zawodzinski TA, Mench MM (2015) High performance electrodes in vanadium redox flow batteries through oxygen-enriched thermal activation. J Power Sources 294:333–338. https://doi.org/10.1016/j.jpowsour.2015.05.118
Aaron D, Yeom S, Kihm KD, Ashraf Gandomi Y, Ertugrul T, Mench MM (2017) Kinetic enhancement via passive deposition of carbon-based nanomaterials in vanadium redox flow batteries. J Power Sources 366:241–248. https://doi.org/10.1016/j.jpowsour.2017.08.108
Kim KJ, Park M-S, Kim Y-J, Kim JH, Dou SX, Skyllas-Kazacos M (2015) A technology review of electrodes and reaction mechanisms in vanadium redox flow batteries. J Mater Chem A 3(33):16913–16933. https://doi.org/10.1039/c5ta02613j
Wang W, Luo Q, Li B, Wei X, Li L, Yang Z (2013) Recent progress in redox flow battery research and development. Adv Funct Mater 23(8):970–986. https://doi.org/10.1002/adfm.201200694
Agar E, Dennison CR, Knehr KW, Kumbur EC (2013) Identification of performance limiting electrode using asymmetric cell configuration in vanadium redox flow batteries. J Power Sources 225:89–94. https://doi.org/10.1016/j.jpowsour.2012.10.016
Milshtein JD, Tenny KM, Barton JL, Drake J, Darling RM, Brushett FR (2017) Quantifying mass transfer rates in redox flow batteries. J Electrochem Soc 164(11):E3265–E3275. https://doi.org/10.1149/2.0201711jes
Zhou XL, Zhao TS, An L, Zeng YK, Wei L (2017) Critical transport issues for improving the performance of aqueous redox flow batteries. J Power Sources 339:1–12. https://doi.org/10.1016/j.jpowsour.2016.11.040
Darling RM, Perry ML (2014) The influence of electrode and channel configurations on flow battery performance. J Electrochem Soc 161(9):A1381–A1387. https://doi.org/10.1149/2.0941409jes
Rudolph S, Schröder U, Bayanov RI, Blenke K, Bayanov IM (2015) Optimal electrolyte flow distribution in hydrodynamic circuit of vanadium redox flow battery. J Electroanal Chem 736:117–126. https://doi.org/10.1016/j.jelechem.2014.11.004
Kumar S, Jayanti S (2017) Effect of electrode intrusion on pressure drop and electrochemical performance of an all-vanadium redox flow battery. J Power Sources 360:548–558. https://doi.org/10.1016/j.jpowsour.2017.06.045
Houser J, Pezeshki A, Clement JT, Aaron D, Mench MM (2017) Architecture for improved mass transport and system performance in redox flow batteries. J Power Sources 351:96–105. https://doi.org/10.1016/j.jpowsour.2017.03.083
Lisboa KM, Marschewski J, Ebejer N, Ruch P, Cotta RM, Michel B, Poulikakos D (2017) Mass transport enhancement in redox flow batteries with corrugated fluidic networks. J Power Sources 359:322–331. https://doi.org/10.1016/j.jpowsour.2017.05.038
Cervantes-Alcalá R, Miranda-Hernández M (2018) Flow distribution and mass transport analysis in cell geometries for redox flow batteries through computational fluid dynamics. J Appl Electrochem 48(11):1243–1254. https://doi.org/10.1007/s10800-018-1246-7
Mögelin H, Barascu A, Krenkel S, Enke D, Turek T, Kunz U (2018) Effect of the pore size and surface modification of porous glass membranes on vanadium redox-flow battery performance. J Appl Electrochem 48(6):651–662. https://doi.org/10.1007/s10800-018-1201-7
Schafner K, Becker M, Turek T (2018) Capacity balancing for vanadium redox flow batteries through electrolyte overflow. J Appl Electrochem 48(6):639–649. https://doi.org/10.1007/s10800-018-1187-1
Kim Y, Choi YY, Yun N, Yang M, Jeon Y, Kim KJ, Choi J-I (2018) Activity gradient carbon felt electrodes for vanadium redox flow batteries. J Power Sources 408:128–135. https://doi.org/10.1016/j.jpowsour.2018.09.066
Xu Q, Zhao TS, Leung PK (2013) Numerical investigations of flow field designs for vanadium redox flow batteries. Appl Energy 105:47–56. https://doi.org/10.1016/j.apenergy.2012.12.041
Jyothi Latha T, Jayanti S (2014) Hydrodynamic analysis of flow fields for redox flow battery applications. J Appl Electrochem 44(9):995–1006. https://doi.org/10.1007/s10800-014-0720-0
Zeng Y, Li F, Lu F, Zhou X, Yuan Y, Cao X, Xiang B (2019) A hierarchical interdigitated flow field design for scale-up of high-performance redox flow batteries. Appl Energy 238:435–441. https://doi.org/10.1016/j.apenergy.2019.01.107
Ke X, Prahl JM, Alexander JID, Savinell RF (2018) Redox flow batteries with serpentine flow fields: distributions of electrolyte flow reactant penetration into the porous carbon electrodes and effects on performance. J Power Sources 384:295–302. https://doi.org/10.1016/j.jpowsour.2018.03.001
MacDonald M, Darling RM (2018) Modeling flow distribution and pressure drop in redox flow batteries. AIChE J 64(10):3746–3755. https://doi.org/10.1002/aic.16330
Mayrhuber I, Dennison CR, Kalra V, Kumbur EC (2014) Laser-perforated carbon paper electrodes for improved mass-transport in high power density vanadium redox flow batteries. J Power Sources 260:251–258. https://doi.org/10.1016/j.jpowsour.2014.03.007
Al-Yasiri M, Park J (2017) Study on channel geometry of all-vanadium redox flow batteries. J Electrochem Soc 164(9):A1970–A1982. https://doi.org/10.1149/2.0861709jes
Agar E, Benjamin A, Dennison CR, Chen D, Hickner MA, Kumbur EC (2014) Reducing capacity fade in vanadium redox flow batteries by altering charging and discharging currents. J Power Sources 246:767–774. https://doi.org/10.1016/j.jpowsour.2013.08.023
Acknowledgements
The authors would like to thank the National Science Foundation (Grant #1351161) for supporting this work. The authors would also like to acknowledge AvCarb Material Solutions for supplying the electrode materials.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akuzum, B., Alparslan, Y.C., Robinson, N.C. et al. Obstructed flow field designs for improved performance in vanadium redox flow batteries. J Appl Electrochem 49, 551–561 (2019). https://doi.org/10.1007/s10800-019-01306-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-019-01306-1