Abstract
Purpose
To validate the hypothesis that BAK induces low-grade inflammation in the anterior chamber, we designed a study to investigate whether switching from BAK-preserved to preservative-free latanoprost in patients with primary open-angle glaucoma (POAG) would reduce the flare levels.
Patients
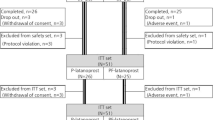
Forty-one eyes of twenty-two patients with primary open-angle glaucoma treated with BAK-preserved latanoprost for at least 6 months as monotherapy were included. Exclusion criteria included any use of topical eye drops other than latanoprost, pseudoexfoliation and pigment dispersion glaucoma, wearing of contact lenses and intraocular surgery in the past year.
Methods
At the start of the study, we measured baseline flare values. We then switched all patients to preservative-free latanoprost. After 1, 2, and 3 months, a routine ophthalmological examination was performed and flare measurement repeated.
Results
Thirty-three eyes were followed up throughout the entire 3-month period. One month after the switch to preservative-free latanoprost, a statistically significant mean drop in flare of − 0.96 ph/ms (P = 0.025) was observed. Mean flare decreased further by − 1.31 ph/ms (P = 0.0027) after 2 months and by − 1.25 ph/ms (P = 0.0041) after 3 months.
Conclusion
The switch from BAK-preserved to preservative-free latanoprost induced a statistically significant reduction in mean flare value. Whereas our previous study showed an increase in flare when initiating treatment with BAK-preserved eye drops, this study shows a decrease in flare upon cessation of BAK-preserved drugs. The combined evidence from the two studies strongly suggests that in humans BAK exerts its effects not only on the ocular surface, but also at the level of the anterior chamber.

Similar content being viewed by others
References
Baudouin C, Labbé A, Lian H et al (2010) Preservatives in eyedrops: the good, the bad and the ugly. Prog Retin Eye Res 19:312–334
Jaenen N, Baudouin C, Pouliquen P et al (2007) Ocular symptoms and signs with preserved and preservative-free glaucoma medications. Eur J Ophthalmol 17:341–349
Leung EW, Medeiros FA, Weinreb RN (2008) Prevalence of ocular surface disease in glaucoma patients. J Glaucoma 17:350–355
Rossi GC, Tinelli C, Pasinetti GM et al (2009) Drye eye syndrome-related quality of life in glaucoma patients. Eur J Ophthalmol 19:572–579
Fechtner RD, Godfrey DG, Budenz D et al (2010) Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea 29:618–621
Pisella PJ, Pouliquen P, Baudouin C (2002) Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication. Br J Ophthalmol 86:418–423
Goto Y, Ibaraki N, Miyake K et al (2003) Human lens epithelial cell damage and stimulation of their secretion of chemical mediators by benzalkonium chloride rather tha latanoprost and timolol. Arch Ophthalmol 121:835–839
Hamard P, Blondin C, Debbasch C, Warnet J-M, Baudouin C, Brignole F (2003) In vitro effects of preserved and unpreserved antiglaucoma drugs on apoptotic marker expression by human trabecular cells. Graefe’s Arch Clin Exp Ophthalmol 241:1037–1043
Samples JR, Binder PS, Nayak S (1989) The effect of epinephrine and benzalkonium chloride on cultured corneal endothelial and trabecular meshwork cells. Exp Eye Res 49:1–12
Miyake K, Ota I, Ibaraki N et al (2001) Enhanced disruption of the blood-aqueous barrier and the incidence of angiographic cystoid macular edema by topical timolol and its preservative in early postoperative pseudophakia. Arch Ophthalmol 119:387–394
Abe RY, Zacchia RS, Santana PR, Costa VP (2014) Effects of benzalkonium chloride on the blood-aqueous and blood-retinal barriers of pseudophakic eyes. J Ocul Pharmacol Ther 30:413–418
Stevens AM, Kestelyn PA, De Bacquer D, Kestelyn PG (2012) Benzalkonium chloride induces anterior chamber inflammation in previously untreated patients with ocular hypertension as measured by flare meter: a randomized clinical trial. Acta Ophthalmol 90:e221–e224
Shah SM, Spalton DJ, Smith SE (1991) Measurement of aqueous cells and flare in normal eyes. Br J Ophthalmol 75:348–352
Onodera T, Gimbel HV, Debroff BM (1993) Aqueous flare and cell number in healthy eyes of Caucasians. Jpn J Ophthalmol 37:445–451
Guillen-Monterrubio OM, Hartikainen J, Taskinen K et al (1997) Quantitative determination of aqueous flare and cells in healthy eyes. Acta Ophthalmol Scand 75:58–62
Bigar F, Herbort CP, Pittet N (1991) Tyndallométrie de la chambre antérieure avec le laser Flare-Cell meter Kowa FC-1000. Klin Mbl Augenheilk 198:396–398
Oshika T, Kato S, Sawa M et al (1989) Aqueous flare intensity and age. Jpn J Ophthalmol 33:237–242
El-Harazi SM, Ruiz RS, Feldman RM et al (2002) Quantitative assessment of aqueous flare: the effect of age and pupillary dilation. Ophthalmic Surg Lasers 33:379–382
Baleriola B, Garcia-Feijoo J, Martinez-de-la-Casa J et al (2008) Apoptosis in the trabecular meshwork of glaucomatous patients. Mol Vis 14:1513–1516
Schmier JK, Coveret DW, Robin AL (2010) First-year treatment costs among new initiators of topical prostaglandin analog identified from November 2007 through April 2008. Curr Med Res Opin Published Online First: 2 November 2010. https://doi.org/10.1185/03007995.2010.531254
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kestelyn, P.A., Kestelyn, P.G., De Bacquer, D. et al. Switch from BAK-preserved to preservative-free latanoprost decreases anterior chamber flare in POAG patients. Int Ophthalmol 39, 105–109 (2019). https://doi.org/10.1007/s10792-017-0792-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-017-0792-z