Abstract
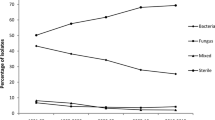
To review the susceptibility of bacterial isolates to ceftazidime and vancomycin isolated from patients with endophthalmitis. Microbiology records of patients with endophthalmitis between June 2010 and May 2013 were reviewed. Vitreous and AC fluids obtained from patients with endophthalmitis were subjected to direct microscopy examination and culture. Antibiotic susceptibility of the isolates was performed by Kirby Bauer disk diffusion method. Resistant to ceftazidime in Gram negative bacteria (GNB) by disk diffusion method is confirmed by minimum inhibitory concentration using E test. Culture was positive for bacteria/Fungi in 224/356 patients (62.9 %). Out of 224 patients, 191 (85.2 %) patients showed bacterial growth and 33 (14.0 %) showed fungal growth. Mixed bacterial infection was seen in five patients. Among the GNB, 23/123 (18 %) of the isolates were resistant to ceftazidime, and all the Gram positive bacteria 73/73 (100 %) were susceptible to vancomycin. Sixteen of 123 (13 %) GNB were resistant to amikacin. Although there is an increase in resistance to ceftazidime compared to amikacin in GNB, amikacin intravitreal injection is associated with macular toxicity and no single antibiotic has full coverage for all GNB. Combination of vancomycin and ceftazidime empiric therapy can be continued in patients with suspected endophthalmitis and treatment is modified based on clinical response and susceptibility results.
Similar content being viewed by others
References
Callegen MC, Engelbert M, Parke DW, Jett BD, Gilmore MS (2002) Bacterial endophthalmitis: epidemiology, therapeutics and bacterium-host interactions. Clin Microbiol Rev 15:111–124
Kwok AKH, Hui M, Pang CP et al (2002) An in vitro study of ceftazidime and vancomycin concentrations in various fluid media: implications for use in treating endophthalmitis. Invest Ophthalmol Vis Sci 43:1182–1188
Morfin-Otero R, Tinoco-Fevila JC, Sader HS et al (2012) Resistance trends in Gram negative bacteria: surveillance results from two Mexican hospitals 2005–2010. BMC Res Notes 5:277
Zhanel GG, Adam HJ, Loe DE et al (2011) Antimicrobial susceptibility of 15,644 pathogens from Canadian hospitals: results of the CANWARD 2007–2009 study. Diagn Microbial Infect Dis 69:291–306
Pathengay A, Morekar M, Puthussery R et al (2011) Clinical and microbiologic review of culture proven endophthalmitis caused by multi drug resistant bacteria in patients seen at a tertiary eye care center south India. Retina 31:806–811
Hillier RJ, Arjmand P, Rebick G, Ostrowski M, Muni RH (2013) Post traumatic vancomycin resistant enterococcal endophthalmitis. J Ophthalmic Inflamm Infect 3:42
Campochiaro PA, Lim JI (1994) Aminoglycoside toxicity in the treatment of endophthalmitis. The aminoglycoside toxicity study group. Arch Ophthalmol 112:48–53
Melo GB, Bispo PJM, Yu MCZ, Pignatari ACC, Hoffling-Lima AL (2011) Microbial profile and antibiotic susceptibility of culture positive bacterial endophthalmitis. Eye 25:382–388
Schimel AM, Flynn HW, Flynn HW Jr (2013) Endophthalmitis isolates and antibiotic susceptibilities: a 10 year review of culture proven cases. Am J Ophthalmol 156:50–52
Sharma S, Padhi TR, Basu S, Kar S, Roy A, Das T (2014) Endophthalmitis seen patients at a tertiary eye care centre in Odisha. A clinico-microbiological analysis. Indian J Med Res 139:91–98
Falavarjani JG, Nekoozadesh S, Modarres M et al (2012) Isolation and antibiotic resistance of culture proven endophthalmitis cases presented to a referral center in Tehran. Middle East Afr J Ophthalmol 19:361–363
Bains HS, Weinberg DV, Feder RS, Noskin GA (2007) Post operative vancomycin resistant Enterococcus faecium endophthalmitis. Arch Ophthalmol 125:1292–1293
Esmaeli B, Holz HR, Ahmadi MA, Krathen RA, Raad II (2003) Endogenous endophthalmitis secondary to vancomycin resistant enterococci infection. Retina 23:118–119
Khera M, Pathengay A, Jindal A (2013) Vancomycin-resistant Gram positive bacterial endophthalmitis: epidemiology, treatment options and outcomes. J Ophthalmic Inflamm Infect 3:46
Jindal A, Pathengay A, Khera M et al (2013) Combined ceftazidime and amikacin resistant among Gram negative isolates in acute onset post operative endophthalmitis: prevalence, antimicrobial susceptibilities and visual acuity outcome. J Ophthalmic Inflamm Infect 3:62
Jindal A, Pathengay A, Mithal K et al (2014) Endophthalmitis after open globe injuries: change in microbial spectrum and isolate susceptibility pattern over 14 years. J Ophthalmic Inflamm Infect 4:5
Anand AR, Therese KL, Madhavan HN (2000) Spectrum of etiological agents of post operative endophthalmitis and antibiotic susceptibilities of bacterial isolates. Indian J Ophthalmol 48:123–128
Seawright AA, Bourke RD, Cooling RJ (1996) Macular toxicity after intravitreal amikacin. Aust N Z J Ophthalmol 24:143–146
Kumar A, Dada T (1999) Periretinal haemorrhages: an unusual manifestation of intravitreal amikacin toxicity. Aust N Z J Ophthalmol 27:435–436
Jackson TL, Williamson TH (1999) Amikacin retinal toxicity. Br J Ophthalmol 83:1199–1200
Hariprasad SM, Shah GK, Mieler WF et al (2006) Vitreous and aqueous penetration of orally administered moxifloxacin in humans. Arch Ophthalmol 124:178–182
Thompson AM (2007) Ocular toxicities of fluoroquinolones. Clin Exp Ophthalmol 35:566–577
Lewis RJ, Mohr JF 3rd (2008) Disglycaemias and fluoroquinolones. Drug Saf 31:283–292
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reddy, A.K., Reddy, R.R., Paruvelli, M.R. et al. Susceptibility of bacterial isolates to vancomycin and ceftazidime from patients with endophthalmitis: Is there a need to change the empirical therapy in suspected bacterial endophthalmitis?. Int Ophthalmol 35, 37–42 (2015). https://doi.org/10.1007/s10792-014-0015-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-014-0015-9