Abstract
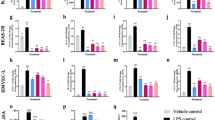
Nowadays, macrophages are recognized as key cells involved in chronic inflammatory conditions, and play central roles in all inflammatory diseases and cancer. Due to their extensive involvement in the pathogenesis of inflammatory diseases, they are now considered a relevant therapeutic target in the development of new therapeutic strategies. 2-Iminothiazolidines are associated with important anti-inflammatory activity and represent a rich source for the development of new drugs and treatments. Our research focuses on evaluating the anti-inflammatory capacity of these compounds and their relationship with M1/M2 macrophage polarization. The results demonstrate that 2-iminothiazolidines have the capacity to decrease the levels of anti-inflammatory biomarkers, such as cytokines (IL-1β, TNF-α, and IL-6), nitric oxide synthase (with impact on NOx production), and COX-2, following a significant decline in NF-kB activation. We also observed an increase in levels of anti-inflammatory cytokines (IL-4 and IL-13) in the in vitro model of RAW 264.7 macrophages induced by LPS. Moreover, this is the first report, suggesting that the anti-inflammatory activity of 2-iminothiazolidines is associated with the ability to enhance phagocytosis, increase Arginase-1 and CD206 expression, and increase the secretion of IL-10. Furthermore, an in vivo study using the acute lung injury model induced by LPS proved the anti-inflammatory activity of a selected 2-iminothiazolidine, named methyl 2-(benzoylimino)-3-methyl-4-(4-nitrobenzyl)-1,3-thiazolidine-4-carboxylate. All these results, taken together, lead us to hypothesize that the mechanism of anti-inflammatory effect observed with this compound is closely related to the ability of this compound to produce macrophage repolarization, from the M1 to the M2 phenotype.



Similar content being viewed by others
References
Aghasafari P, George U, Pidaparti R (2019) A review of inflammatory mechanism in airway diseases. Inflamm Res 68(1):59–74. https://doi.org/10.1007/s00011-018-1191-2
Ali Y, Alam MS, Hamid H, Husain A, Dhulap A, Bano S, Kharbanda C (2017) Novel 2,4-dichlorophenoxy acetic acid substituted thiazolidin-4-ones as anti-inflammatory agents: design, synthesis and biological screening. Bioorg Med Chem Lett 27(4):1017–1025. https://doi.org/10.1016/j.bmcl.2016.12.069
Bhalgat CM, Darda PV, Bothara KG, Bhandari SV, Gandhi J, Ramesh B (2014) Synthesis and pharmacological screening of some novel anti-hypertensive agents possessing 5-benzylidene-2-(phenylimino)-thiazolidin-4-one ring. Eur J Med Chem 76:580–588. https://doi.org/10.1016/j.ejmech.2014.02.048
Bhandari S, Sakla AP, Shankaraiah N (2020) FeCl3-Catalyzed [3+2] cycloaddition reaction: a mild synthetic approach to spirooxindolo-2-iminothiazolidine Scaffolds. Chem Select 5(10):2886–2891. https://doi.org/10.1002/slct.201904397
Bhattacharyya A (2020) Synthetic routes to 2-iminothiazolidines: state-of-the-art 2006–2020. Curr Org Chem 24:2823–2844. https://doi.org/10.2174/1385272824999201019162400
Bodhak C, Mandal S, Dey P, Mukherjee SK, Pramanik A (2022) Efficient synthesis of functionalized 2-iminothiazolines by ultrasonication under solvent-free conditions and access to 5-aryl-2-iminothiazolines. Results Chem 4:100301. https://doi.org/10.1016/j.rechem.2022.100301
Bosco MC (2019) Macrophage polarization: reaching across the aisle? J Allergy Clin Immunol 143(4):1348–1350. https://doi.org/10.1016/j.jaci.2018.12.995
Chan C, Martin P, Liptrott NJ, Siccardi M, Almond L, Owen A (2017) Incompatibility of chemical protein synthesis inhibitors with accurate measurement of extended protein degradation rates. Pharmacol Res Perspect 5(5):e00359. https://doi.org/10.1002/prp2.359
Csaba G (2017) Is there a hormonal regulation of phagocytosis at unicellular and multicellular levels? A critical review. Acta Microbiol Immunol Hung 64(4):357–372. https://doi.org/10.1556/030.64.2017.024
Di Benedetto P, Ruscitti P, Vadasz Z, Toubi E, Giacomelli R (2019) Macrophages with regulatory functions, a possible new therapeutic perspective in autoimmune diseases. Autoimmun Rev 18(10):102369. https://doi.org/10.1016/j.autrev.2019.102369
Elkamhawy A, Kim NY, Hassan AHE, Park J-E, Yang J-E, Oh K-S, Roh EJ (2018) Design, synthesis and biological evaluation of novel thiazolidinedione derivatives as irreversible allosteric IKK-β modulators. Eur J Med Chem 157:691–704. https://doi.org/10.1016/j.ejmech.2018.08.020
Elkamhawy A, Kim NY, Hassan AHE, Park J-E, Paik S, Yang J-E, Roh EJ (2020) Thiazolidine-2,4-dione-based irreversible allosteric IKK-β kinase inhibitors: optimization into in vivo active anti-inflammatory agents. Eur J Med Chem 188:111955. https://doi.org/10.1016/j.ejmech.2019.111955
Feehan KT, Gilroy DW (2019) Is resolution the end of inflammation? Trends Mol Med 25(3):198–214. https://doi.org/10.1016/j.molmed.2019.01.006
Ferreira M, Sá MM (2015) Formal [3+2] Annulation Involving Allylic Bromides and Thioureas. Synthesis of 2-iminothiazolidines through a base-catalyzed intramolecular anti-michael addition. Adv Synth Catal 357(4):829–833. https://doi.org/10.1002/adsc.201401026
Ferreira M, Munaretto LS, Gouvêa DP, Bortoluzzi AJ, Sá MM (2022) Diversity-oriented synthesis of 2-iminothiazolidines: pushing the boundaries of the domino nucleophilic displacement/intramolecular anti-michael addition process. Eur J Org Chem. https://doi.org/10.1002/ejoc.202101562
Fields RD, Lancaster MV (1993) Dual-attribute continuous monitoring of cell proliferation/cytotoxicity. Am Biotechnol Lab 11(4):48–50
Fraternale A, Brundu S, Magnani M (2015) Polarization and repolarization of macrophages. J Clin Cell Immunol 6(319):2. https://doi.org/10.4172/2155-9899.1000319
Fullerton JN, Gilroy DW (2016) Resolution of inflammation: a new therapeutic frontier. Nat Rev Drug Discov 15(8):551–567. https://doi.org/10.1038/nrd.2016.39
Gaber T, Strehl C, Buttgereit F (2017) Metabolic regulation of inflammation. Nat Rev Rheumatol 13(5):267–279. https://doi.org/10.1038/nrrheum.2017.37
Ghafoori H, Rezaei M, Mohammadi A (2017) Anti-inflammatory effects of novel thiazolidinone derivatives as bioactive heterocycles on RAW 264.7 cells. Iran J Allergy Asthma Immunol 28–38
Gharib SA et al (2019) Transcriptional and functional diversity of human macrophage repolarization. J Allergy Clin Immun 143(4):1536–1548. https://doi.org/10.1016/j.jaci.2018.10.046
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126(1):131–138. https://doi.org/10.1016/0003-2697(82)90118-X
Gyertyán I, Petöcz L, Gacsályi I, Fekete MIK, Tekes K, Kápolnai L (1991) Psychopharmacological effects of an imino-thiazolidine derivative antidepressant candidate, EGYT-4201. Drug Dev Res 22(4):385–399. https://doi.org/10.1002/ddr.430220410
He L, Jhong J-H, Chen Q, Huang K-Y, Strittmatter K, Kreuzer J, Marneros AG (2021) Global characterization of macrophage polarization mechanisms and identification of M2-type polarization inhibitors. Cell Rep 37(5):109955. https://doi.org/10.1016/j.celrep.2021.109955
Hu J, Wang Y, Wei X, Wu X, Chen G, Cao G, Shen X, Zhang X, Tang Q, Liang G, Li X (2013) Synthesis and biological evaluation of novel thiazolidinone derivatives as potential anti-inflammatory agents. Eur J Med Chem 64:292–301. https://doi.org/10.1016/j.ejmech.2013.04.010
Kieler M, Hofmann M, Schabbauer G (2021) More than just protein building blocks: How amino acids and related metabolic pathways fuel macrophage polarization. FEBS J 288(12):3694–3714. https://doi.org/10.1111/febs.15715
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Mokarram N, Bellamkonda RV (2014) A perspective on immunomodulation and tissue repair. Ann Biomed Eng 42(2):338–351. https://doi.org/10.1007/s10439-013-0941-0
Murray PJ (2017) Macrophage polarization. Annu Rev Physiol 79:541–566. https://doi.org/10.1146/annurev-physiol-022516-034339
Nascimento MVPS, Munhoz ACM, Facchin BMDC, Fratoni E, Rossa TA, Sá MM, Campa CC, Ciraolo E, Hirsch E, Dalmarco EM (2019) New pre-clinical evidence of anti-inflammatory effect and safety of a substituted fluorophenyl imidazole. Biomed Pharmacother 111:1399–1407. https://doi.org/10.1016/j.biopha.2019.01.052
Nathan C, Ding A (2010) Nonresolving inflammation. Cell 140(6):871–882. https://doi.org/10.1016/j.cell.2010.02.029
O’Brien J, Wilson I, Orton T, Pognan F (2000) Investigation of the Alamar Blue (resazurin) fluorescent dye for the assessment of mammalian cell cytotoxicity. Eur J Mol Biol Biochem 267(17):5421–5426. https://doi.org/10.1046/j.1432-1327.2000.01606.x
Orecchioni M, Ghosheh Y, Pramod AB, Ley K (2019) Macrophage polarization: different gene signatures in M1 (LPS+) vs. classically and M2 (LPS–) vs. alternatively activated macrophages. Front Immunol. https://doi.org/10.3389/fimmu.2019.01084
Ottanà R, Maccari R, Barreca ML, Bruno G, Rotondo A, Rossi A, Chiricosta G, Di Paola R, Sautebin L, Cuzzocrea S, Vigorita MG (2005) 5-Arylidene-2-imino-4-thiazolidinones: design and synthesis of novel anti-inflammatory agents. Bioorg Med Chem 13(13):4243–4252. https://doi.org/10.1016/j.bmc.2005.04.058
Panigrahy D, Gilligan MM, Serhan CN, Kashfi K (2021) Resolution of inflammation: an organizing principle in biology and medicine. Pharmacol Ther 227:107879. https://doi.org/10.1016/j.pharmthera.2021.107879
Ratner S, Clarke HT (1937) The action of formaldehyde upon cysteine. J Am Chem Soc 59(1):200–206. https://doi.org/10.1021/ja01280a050
Saeed A, Zaman S, Jamil M, Mirza B (2008) Synthesis and antifungal activity of some novel N-(4-phenyl-3-aroylthiazol-2(3H)-ylidene) substituted benzamides. Turk J Chem 32(5):585–592. https://doi.org/10.3906/che-0802-17
Shankaran K, Donnelly KL, Shah SK, Guthikonda RN, MacCoss M, Humes JL, Pacholok SG, Grant SK, Kelly TM, Wong KK (2004) Evaluation of pyrrolidin-2-imines and 1,3-thiazolidin-2-imines as inhibitors of nitric oxide synthase. Bioorg Med Chem Lett 14:4539–4544. https://doi.org/10.1016/j.bmcl.2004.06.033
Shapouri-Moghaddam A, Mohammadian S, Vazini H, Taghadosi M, Esmaeili S-A, Mardani F, Seifi B, Mohammadi A, Afshari JT, Sahebkar A (2018) Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol 233(9):6425–6440. https://doi.org/10.1002/jcp.26429
Shukla UK, Singh R, Khanna JM, Saxena AK, Singh HK, Sur RN, Dhawan BN, Anand N (1992) Synthesis of trans-2-[N-(2-Hydroxy-1,2,3,4-tetrahydronaphthalene-1-yl)]iminothiazolidine and related compounds—a new class of antidepressants. Collect Czech Chem Commun 57(2):415–424. https://doi.org/10.1135/cccc19920415
Sugimoto MA, Sousa LP, Pinho V, Perretti M, Teixeira MM (2016) Resolution of inflammation: what controls its onset? Front Immunol. https://doi.org/10.3389/fimmu.2016.00160
Takiguchi H, Yang CX, Yang CWT, Sahin B, Whalen BA, Milne S, Sin DD (2021) Macrophages with reduced expressions of classical M1 and M2 surface markers in human bronchoalveolar lavage fluid exhibit pro-inflammatory gene signatures. Sci Rep 11:8282. https://doi.org/10.1038/s41598-021-87720-y
Ueda S, Terauchi H, Yano A, Matsumoto M, Kubo T, Kyoya Y, Suzuki K, Ido M, Kawasaki M (2004) 4,5-Dialkylsubstituted 2-imino-1,3-thiazolidine derivatives as potent inducible nitric oxide synthase inhibitors. Bioorg Med Chem 12(15):4101–4116. https://doi.org/10.1016/j.bmc.2004.05.031
van der Goes A, Hoekstra K, van den Berg TK, Dijkstra CD (2000) Dexamethasone promotes phagocytosis and bacterial killing by human monocytes/macrophages in vitro. J Leukoc Biol 67(6):801–807. https://doi.org/10.1002/jlb.67.6.801
Varela ML, Mogildea M, Moreno I, Lopes A (2018) Acute inflammation and metabolism. Inflammation 41(4):1115–1127. https://doi.org/10.1007/s10753-018-0739-1
Wang LX, Zhang SX, Wu HJ, Rong XL, Guo J (2019) M2b macrophage polarization and its roles in diseases. J Leukoc Biol 106(2):345–358. https://doi.org/10.1002/JLB.3RU1018-378RR
Wculek SK, Dunphy G, Heras-Murillo I, Mastrangelo A, Sancho D (2021) Metabolism of tissue macrophages in homeostasis and pathology. Cell Mol Immunol 8:1–25. https://doi.org/10.1038/s41423-021-00791-9
Zhao M, Yuan X, Pei Y-H, Ye H-Y, Peng A-H, Tang M-H, Guo D-L, Deng Y, Chen L-J (2019) Anti-inflammatory ellagitannins from Cleidion brevipetiolatum for the treatment of rheumatoid arthritis. J Nat Prod 82(9):2409–2418. https://doi.org/10.1021/acs.jnatprod.8b00984
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohr, E.T.B., Lubschinski, T.L., da Rosa, J.S. et al. The anti-inflammatory activity of 2-iminothiazolidines: evidence for macrophage repolarization. Inflammopharmacol 30, 2427–2439 (2022). https://doi.org/10.1007/s10787-022-01084-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-022-01084-x