Abstract
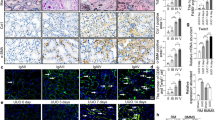
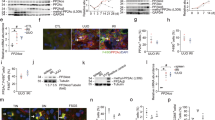
Renal fibrosis is the fundamental pathway leading to end-stage renal disease, while its exact molecular basis remains incompletely elucidated. Previous studies have demonstrated that transforming growth factor beta 1 (TGFβ1) is an inducer of the epithelial-to-mesenchymal transition (EMT) of renal tubular epithelial cells, while bone morphogenic protein 7 (BMP7) counteracts TGFβ1-induced EMT and reverses chronic renal injury. Although macrophage recruitment is believed to play an important role during the whole pathogenesis, the mechanism underlying their activate involvement in the formation of renal fibrosis besides phagocytosizing extracellular matrix and apoptotic cells is largely unknown. Here, in a mouse unilateral ureteral obstruction (UUO) model, we show that the recruited macrophages are mainly M1 macrophages at early stage. However, these F4/80-positive and CD301-negative M1 macrophages were shortly polarized into F4/80-positive and CD301-positive M2 macrophages, respectively, which released high levels TGFβ1, to contradict the local expression of BMP7 to facilitate EMT-induced renal fibrosis. M2 macrophages depletion specifically inhibited EMT, and subsequently the renal fibrosis. Adoptive transplantation of M2 macrophages increased the features of renal fibrosis. Our study thus identified double-edged effects of macrophages in the formation of renal fibrosis, which suggest that modulation of macrophage polarization may substantially improve the treatment of renal fibrosis.




Similar content being viewed by others
Abbreviations
- UUO:
-
Unilateral ureteral obstruction
- EMT:
-
Epithelial-to-mesenchymal transition
- TGFβ1:
-
Transforming growth factor beta 1
- BMP7:
-
Bone morphogenic protein 7
- α-SMA:
-
Alpha-smooth muscle actin
Reference
Ma, L.J., H. Yang, A. Gaspert, G. Carlesso, M.M. Barty, J.M. Davidson, et al. 2003. Transforming growth factor-β-dependent and -independent pathways of induction of tubulointerstitial fibrosis in β6(-/-) mice. American Journal of Pathology 163: 1261–1273.
Sato, M., Y. Muragaki, S. Saika, A.B. Roberts, and A. Ooshima. 2003. Targeted disruption of TGF-β1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. Journal of Clinical Investigation 112: 1486–1494.
Goto, Y., N. Manabe, K. Uchio-Yamada, M. Yamaguchi-Yamada, N. Inoue, Y. Yamamoto, et al. 2004. Augmented cytoplasmic Smad4 induces acceleration of TGF-β1 signaling in renal tubulointerstitial cells of hereditary nephrotic ICGN mice with chronic renal fibrosis; possible role for myofibroblastic differentiation. Cell and Tissue Research 315: 209–221.
Hwang, M., H.J. Kim, H.J. Noh, Y.C. Chang, Y.M. Chae, K.H. Kim, et al. 2006. TGF-β1 siRNA suppresses the tubulointerstitial fibrosis in the kidney of ureteral obstruction. Experimental and Molecular Pathology 81: 48–54.
Liu, F.Y., X.Z. Li, Y.M. Peng, H. Liu, and Y.H. Liu. 2007. Arkadia-Smad7-mediated positive regulation of TGF-β signaling in a rat model of tubulointerstitial fibrosis. American Journal of Nephrology 27: 176–183.
Wang, Y., Z. Zhang, H. Shen, Y. Lu, H. Li, X. Ren, et al. 2008. TGF-β1/Smad7 signaling stimulates renal tubulointerstitial fibrosis induced by AAI. Journal of Receptor and Signal Transduction Research 28: 413–428.
Tamura, M., R. Aizawa, M. Hori, and H. Ozaki. 2009. Progressive renal dysfunction and macrophage infiltration in interstitial fibrosis in an adenine-induced tubulointerstitial nephritis mouse model. Histochemistry and Cell Biology 131: 483–490.
Yeh, Y.C., W.C. Wei, Y.K. Wang, S.C. Lin, J.M. Sung, and M.J. Tang. 2010. Transforming growth factor-β1 induces Smad3-dependent β1 integrin gene expression in epithelial-to-mesenchymal transition during chronic tubulointerstitial fibrosis. American Journal of Pathology 177: 1743–1754.
Sugimoto, H., C. Yang, V.S. LeBleu, M.A. Soubasakos, M. Giraldo, M. Zeisberg, et al. 2007. BMP-7 functions as a novel hormone to facilitate liver regeneration. FASEB Journal 21: 256–264.
Zeisberg, M. 2006. Bone morphogenic protein-7 and the kidney: Current concepts and open questions. Nephrology, Dialysis, Transplantation 21: 568–573.
Zeisberg, M., J. Hanai, H. Sugimoto, T. Mammoto, D. Charytan, F. Strutz, et al. 2003. BMP-7 counteracts TGF-β1-induced epithelial-to-mesenchymal transition and reverses chronic renal injury. Nature Medicine 9: 964–968.
Manucha, W. 2007. Biochemical-molecular markers in unilateral ureteral obstruction. Biocell: Official Journal of the Sociedades Latinoamericanas de Microscopia Electronica et al. 31: 1–12.
Wen, J.G. 2002. Partial unilateral ureteral obstruction in rats. Neurourology and Urodynamics 21: 231–250.
Belmiro, C.L., R.G. Goncalves, E.O. Kozlowski, A.F. Werneck, C.M. Takyia, M. Leite-Jr, et al. 2011. Dermatan sulfate reduces monocyte chemoattractant protein 1 and TGF-β production, as well as macrophage recruitment and myofibroblast accumulation in mice with unilateral ureteral obstruction. Brazilian Journal of Medical and Biological Research 44: 624–633.
Sung, S.A., S.K. Jo, W.Y. Cho, N.H. Won, and H.K. Kim. 2007. Reduction of renal fibrosis as a result of liposome encapsulated clodronate induced macrophage depletion after unilateral ureteral obstruction in rats. Nephron Experimental Nephrology 105: e1–e9.
Naruse, T., Y. Yuzawa, T. Akahori, M. Mizuno, S. Maruyama, R. Kannagi, et al. 2002. P-selectin-dependent macrophage migration into the tubulointerstitium in unilateral ureteral obstruction. Kidney International 62: 94–105.
Takeda, A., A. Fukuzaki, H. Kaneto, S. Ishidoya, Y. Ogata, T. Sasaki, et al. 2000. Role of leukocyte adhesion molecules in monocyte/macrophage infiltration in weanling rats with unilateral ureteral obstruction. International Journal of Urology: Official Journal of the Japanese Urological Association 7: 415–420.
Gordon, S. 2003. Alternative activation of macrophages. Nature Reviews Immunology 3: 23–35.
Gordon, S., and F.O. Martinez. 2010. Alternative activation of macrophages: Mechanism and functions. Immunity 32: 593–604.
Geissmann, F., M.G. Manz, S. Jung, M.H. Sieweke, M. Merad, and K. Ley. 2010. Development of monocytes, macrophages, and dendritic cells. Science 327: 656–661.
Ricardo, S.D., H. van Goor, and A.A. Eddy. 2008. Macrophage diversity in renal injury and repair. Journal of Clinical Investigation 118: 3522–3530.
Xiao, X., I. Gaffar, P. Guo, J. Wiersch, S. Fischbach, L. Peirish, et al. 2014. M2 macrophages promote beta-cell proliferation by up-regulation of SMAD7. Proceedings of the National Academy of Sciences of the United States of America 111: E1211–E1220.
van Rooijen, N., J. Bakker, and A. Sanders. 1997. Transient suppression of macrophage functions by liposome-encapsulated drugs. Trends in Biotechnology 15: 178–185.
van Rooijen, N., and R. van Nieuwmegen. 1984. Elimination of phagocytic cells in the spleen after intravenous injection of liposome-encapsulated dichloromethylene diphosphonate. An enzyme-histochemical study. Cell and Tissue Research 238: 355–358.
Plosker, G.L., and K.L. Goa. 1994. Clodronate. A review of its pharmacological properties and therapeutic efficacy in resorptive bone disease. Drugs 47: 945–982.
Kanis, J.A., and E.V. McCloskey. 1997. Clodronate. Cancer 80: 1691–1695.
Gewin, L., and R. Zent. 2012. How does TGF-β mediate tubulointerstitial fibrosis? Seminars in Nephrology 32: 228–235.
Wells, R.G. 2004. BMP-7: A new TGF-β family member takes the stage in colitis treatment. Inflammatory Bowel Diseases 10: 169.
Acknowledgments
This work was supported by the internal funding of Renmin Hospital of Wuhan University, China.
Conflict of Interest
The authors have declared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, B., Liu, X., Fan, Y. et al. Macrophages Regulate Renal Fibrosis Through Modulating TGFβ Superfamily Signaling. Inflammation 37, 2076–2084 (2014). https://doi.org/10.1007/s10753-014-9941-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-014-9941-y