Abstract
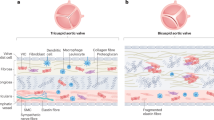
Calcific aortic valve disease (CAVD) is the most frequent heart valve disorder. Studies indicate that mineralization of the aortic valve may be related to the inflammatory process. However, no clear evidence has been given regarding clinical evolution of aortic stenosis and the inflammatory process within the aortic valve. Aortic valves excised from 285 patients with CAVD undergoing aortic valve replacement were analyzed for the presence of chronic inflammatory infiltrates, and those findings were related to the hemodynamic severity of aortic stenosis. In a subset of 57 patients, in whom additional valvular tissue and the clinical progression rate of aortic stenosis were available, the density of leukocytes was determined as well as the number of TNF-α transcripts. Histological analyses revealed that in 81 (28.4 %) patients, the presence of chronic inflammatory infiltrates was documented within CAVD tissue, which was characterized by the existence of a cluster of cells as well as the presence of neovascularisation and osseous metaplasia. The presence of an inflammatory process within the CAVD tissue was independently related to the remodeling process and the peak transaortic gradient. In addition, the density of leukocytes within CAVD tended to correlate (r = 0.25, p = 0.05) with the progression rate of aortic stenosis. Dense inflammatory infiltrate within CAVD is associated with an active remodeling process, the severity of aortic stenosis, and the hemodynamic progression rate.





Similar content being viewed by others
Reference
Cote, C., P. Pibarot, J.P. Despres, D. Mohty, A. Cartier, B.J. Arsenault, C. Couture, and P. Mathieu. 2008. Association between circulating oxidised low-density lipoprotein and fibrocalcific remodelling of the aortic valve in aortic stenosis. Heart 94: 1175–1180.
Messika-Zeitoun, D., M.C. Aubry, D. Detaint, L.F. Bielak, P.A. Peyser, P.F. Sheedy, S.T. Turner, J.F. Breen, C. Scott, A.J. Tajik, and M. Enriquez-Sarano. 2004. Evaluation and clinical implications of aortic valve calcification measured by electron-beam computed tomography. Circulation 110: 356–362.
Mohty, D., P. Pibarot, J.P. Despres, C. Cote, B. Arsenault, A. Cartier, P. Cosnay, C. Couture, and P. Mathieu. 2008. Association between plasma LDL particle size, valvular accumulation of oxidized LDL, and inflammation in patients with aortic stenosis. Arteriosclerosis, Thrombosis, and Vascular Biology 28: 187–193.
Rajamannan, N.M., F.J. Evans, E. Aikawa, K.J. Grande-Allen, L.L. Demer, D.D. Heistad, C.A. Simmons, K.S. Masters, P. Mathieu, K.D. O'Brien, F.J. Schoen, D.A. Towler, A.P. Yoganathan, and C.M. Otto. 2011. Calcific aortic valve disease: not simply a degenerative process: a review and agenda for research from the National Heart and Lung and Blood Institute Aortic Stenosis Working Group. Executive summary: calcific aortic valve disease—2011 update. Circulation 124: 1783–1791.
Cowell, S.J., D.E. Newby, R.J. Prescott, P. Bloomfield, J. Reid, D.B. Northridge, and N.A. Boon. 2005. A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. The New England Journal of Medicine 352: 2389–2397.
Rossebo, A.B., T.R. Pedersen, K. Boman, P. Brudi, J.B. Chambers, K. Egstrup, E. Gerdts, C. Gohlke-Barwolf, I. Holme, Y.A. Kesaniemi, W. Malbecq, C.A. Nienaber, S. Ray, T. Skjaerpe, K. Wachtell, and R. Willenheimer. 2008. Intensive lipid lowering with simvastatin and ezetimibe in aortic stenosis. The New England Journal of Medicine 359: 1343–1356.
Chan, K.L., K. Teo, J.G. Dumesnil, A. Ni, and J. Tam. 2010. Effect of lipid lowering with rosuvastatin on progression of aortic stenosis: results of the Aortic Stenosis Progression Observation: Measuring Effects of Rosuvastatin (ASTRONOMER) trial. Circulation 121: 306–314.
Otto, C.M. 2008. Calcific aortic stenosis—time to look more closely at the valve. The New England Journal of Medicine 359: 1395–1398.
Warren, B.A., and J.L. Yong. 1997. Calcification of the aortic valve: its progression and grading. Pathology 29: 360–368.
Wypasek, E., J. Natorska, G. Grudzien, G. Filip, J. Sadowski, and A. Undas. 2012. Mast cells in human stenotic aortic valves are associated with the severity of stenosis. Inflammation. doi:10.1007/s10753-012-9565-z.
Dweck, M.R., C. Jones, N.V. Joshi, A.M. Fletcher, H. Richardson, A. White, M. Marsden, R. Pessotto, J.C. Clark, W.A. Wallace, D.M. Salter, G. McKillop, E.J. van Beek, N.A. Boon, J.H. Rudd, and D.E. Newby. 2012. Assessment of valvular calcification and inflammation by positron emission tomography in patients with aortic stenosis. Circulation 125: 76–86.
Yip, C.Y., and C.A. Simmons. 2011. The aortic valve microenvironment and its role in calcific aortic valve disease. Cardiovascular Pathology 20: 177–182.
Charest, A., A. Pepin, R. Shetty, C. Cote, P. Voisine, F. Dagenais, P. Pibarot, and P. Mathieu. 2006. Distribution of SPARC during neovascularisation of degenerative aortic stenosis. Heart 92: 1844–1849.
Mohler III, E.R., F. Gannon, C. Reynolds, R. Zimmerman, M.G. Keane, and F.S. Kaplan. 2001. Bone formation and inflammation in cardiac valves. Circulation 103: 1522–1528.
Briand, M., I. Lemieux, J.G. Dumesnil, P. Mathieu, A. Cartier, J.P. Despres, M. Arsenault, J. Couet, and P. Pibarot. 2006. Metabolic syndrome negatively influences disease progression and prognosis in aortic stenosis. Journal of the American College of Cardiology 47: 2229–2236.
Briand, M., P. Pibarot, J.P. Despres, P. Voisine, J.G. Dumesnil, F. Dagenais, and P. Mathieu. 2006. Metabolic syndrome is associated with faster degeneration of bioprosthetic valves. Circulation 114: I512–I517.
Katz, R., N.D. Wong, R. Kronmal, J. Takasu, D.M. Shavelle, J.L. Probstfield, A.G. Bertoni, M.J. Budoff, and K.D. O'Brien. 2006. Features of the metabolic syndrome and diabetes mellitus as predictors of aortic valve calcification in the Multi-Ethnic Study of Atherosclerosis. Circulation 113: 2113–2119.
Mathieu, P., I. Lemieux, and J.P. Despres. 2010. Obesity, inflammation, and cardiovascular risk. Clinical Pharmacology and Therapeutics 87: 407–416.
Mathieu, P., P. Poirier, P. Pibarot, I. Lemieux, and J.P. Despres. 2009. Visceral obesity: the link among inflammation, hypertension, and cardiovascular disease. Hypertension 53: 577–584.
Kaden, J.J., R. Kilic, A. Sarikoc, S. Hagl, S. Lang, U. Hoffmann, M. Brueckmann, and M. Borggrefe. 2005. Tumor necrosis factor alpha promotes an osteoblast-like phenotype in human aortic valve myofibroblasts: a potential regulatory mechanism of valvular calcification. International Journal of Molecular Medicine 16: 869–872.
Bosse, Y., A. Miqdad, D. Fournier, A. Pepin, P. Pibarot, and P. Mathieu. 2009. Refining molecular pathways leading to calcific aortic valve stenosis by studying gene expression profile of normal and calcified stenotic human aortic valves. Circulation. Cardiovascular Genetics 2: 489–498.
Derbali, H., Y. Bosse, N. Cote, P. Pibarot, A. Audet, A. Pepin, B. Arsenault, C. Couture, J.P. Despres, and P. Mathieu. 2010. Increased biglycan in aortic valve stenosis leads to the overexpression of phospholipid transfer protein via Toll-like receptor 2. American Journal of Pathology 176: 2638–2645.
Acknowledgments
This study was supported by CIHR grants MOP114893 and MOP79342 and the Heart and Stroke Foundation of Canada (HSFC). N. Coté is a recipient of a doctoral research award from the Fonds de Recherche du Québec–Santé (FRQS). Y. Bossé is a research scholar from the HSFC. P. Pibarot holds the Canada Research Chair in Valvular Heart Diseases. P. Mathieu is a research scholar from the FRQS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coté, N., Mahmut, A., Bosse, Y. et al. Inflammation Is Associated with the Remodeling of Calcific Aortic Valve Disease. Inflammation 36, 573–581 (2013). https://doi.org/10.1007/s10753-012-9579-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-012-9579-6