Abstract
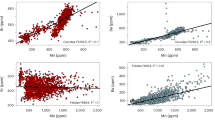
We assessed the feeding habits of the Amazonian manatee inhabiting blackwater (Negro River) and clearwater (Tapajós River) igapós (floodplains) using δ13C and δ15N of dentin and bone collagen from different ontogenetic classes (nursing calves, juveniles, and adults). Within an individual, the dentine δ13C and δ15N values did not vary with tooth position. Bones were more depleted in 13C and 15N compared to teeth, and the δ13C and δ15N in bone differed among classes. Food sources had δ13C values typical of algae, plants of C3 or C4 photosynthetic pathways. Mixing models showed that lactating females (inferred by isotopic values from nursing calves) had higher proportional consumption of C4 plants, while, for other adults, C3 plants were more frequent in the diet of manatees from both rivers. Juveniles had a mixed diet of C3 pants and C4 plants. We hypothesize the C4 plants signal of calves results from female movements to the floodplains of nearby whitewater rivers (várzeas), where C4 plants are abundant. Individuals from Tapajós may be more resident, as C3 plants and C4 plants are available during the flood pulse. Results demonstrate that manatee diets vary with ontogenetic classes and water typology. Preservation of habitats (igapó/várzea) is therefore required for the survival of Amazonian manatees.




Similar content being viewed by others
References
Abend, A. G. & T. D. Smith, 1995. Differences in ratios of stable isotopes of nitrogen in long-finned pilot whales (Globicephala melas) in the western and eastern North Atlantic Ices. Journal of Marine Science 52: 837–841.
Albernaz, T. L., E. R. Secchi, L. R. de Oliveira & S. Botta, 2016. Ontogenetic and gender-related variation in the isotopic niche within and between three species of fur seals (genus Arctocephalus). Hydrobiologia 787: 123–139.
Alves-Stanley, C. D. & G. A. J. Worthy, 2009. Carbon and nitrogen stable isotope turnover rates and diet–tissue discrimination in Florida manatees (Trichechus manatus latirostris). The Journal of Experimental Biology 212: 2349–2355.
Ames, A. L., E. S. Vleet & W. M. Sackett, 1996. The use of stable carbon isotope analysis for determining the dietary habits of the Florida manatee, Trichechus manatus latirostris. Marine Mammal Science 12: 555–563.
Arraut, E. M., M. Marmontel, J. E. Mantovani, E. M. L. Novo, D. W. Macdonald & R. E. Kenward, 2010. The lesser of two evils: seasonal migrations of Amazonian manatees in the Western Amazon. Journal of Zoology 208: 246–256.
Ayres, J. M, 1986. Uakaris and Amazonian Flooded forest. Thesis, University of Cambridge, Cambridge.
Barbour, T., 1973. Birth of manatee. Journal of Mammalogy 18: 106–107.
Beatty, B. L., T. Vitkovski, O. Lambert & T. E. Macrini, 2012. Osteological associations with unique tooth development in manatees (Trichechidae, Sirenia): a detailed look at modern Trichechus and a review of the fossil record. The Anatomical Record 295: 1504–1512.
Best, R. C., 1981. Food and feeding habits of wild and captive Sirenia. Mammal Review 11: 3–29.
Best, R. C., 1982. Seasonal breeding in the Amazonian manatee, Trichechus inuguis Mammalia: Sirenia. Biotropica Notes 14: 76–78.
Best, R. C., 1983. Apparent dry-season fasting in Amazonian Manatees (Mammalia: Sirenia). Biotropica 15: 61–64.
Best, R. C., 1984. The aquatic mammals and reptiles on the Amazon. In Sioli, H. (ed.), The Amazon. Limnology and landscape ecology of a mighty tropical river and its basin. Springer, Dordrecht: 370–412.
Bini, L. M. & S. M. Thomaz, 2005. Prediction of Egeria najas and Egeria densa occurrence in a large subtropical reservoir (Itaipu Reservoir, Brazil-Paraguay). Aquatic Botany 83: 227–238.
Bocherens, H., G. Pacaud, P. A. Lazarev & A. Mariotti, 1996. Stable isotope abundances (13C, 15N) in collagen and soft tissues from Pleistocene mammals from Yakutia: implications for the palaeobiology of the Mammoth Steppe. Palaeogeography, Palaeoclimatology, Palaeoecology 126: 31–44.
Bond, A. L. & A. W. Diamond, 2011. Recent Bayesian stable-isotope mixing models are highly sensitive to variation in discrimination factors. Ecological Applications 21: 1017–1023.
Boon, P. I. & S. E. Bunn, 1994. Variations in the stable isotope composition of aquatic plants and their implications for food web analysis. Aquatic Botany 48: 99–108.
Boskey, A. L., 2007. Mineralization of bones and teeth. Elements 3: 385–391.
Bowen, W. D. & S. J. Iverson, 2013. Methods of estimating marine mammal diets: a review of validation experiments and sources of bias and uncertainty. Marine Mammal Science 29: 719–754.
Castelblanco-Martínez, D. N., A. L. Bermúdez-Romero, I. Gómez Camelo, F. C. W. Rosas, F. Trujillo & E. Zerda-Ordóñez, 2009. Seasonality of habitat use, mortality and reproduction of the vulnerable Antillean manatee Trichechus manatus manatus in the Orinoco River, Colombia: implications for conservation. Oryx 43: 235–242.
Cherel, Y., K. A. Hobson & C. Guinet, 2015. Milk isotopic values demonstrate that nursing fur seal pups are a full trophic level higher than their mothers. Rapid Communications in Mass Spectrometry 30(29): 1485–1490.
Ciotti, L. L., F. O. Luna & E. R. Secchi, 2014. Intra-and interindividual variation in δ13C and δ15N composition in the Antillean manatee Trichechus manatus manatus from northeastern Brazil. Marine Mammal Science 30(3): 1238–1247.
Clementz, M. T., A. Goswami, P. D. Gingerich & P. L. Koch, 2006. Isotopic records from early whales and sea cows: contrasting patterns of ecological transition. Journal of Vertebrate Paleontology 26: 355–370.
Clementz, M. T., P. L. Koch & C. A. Beck, 2007. Diet induced differences in carbon isotope fractionation between sirenians and terrestrial ungulates. Marine Biology 151: 1773–1784.
Colares, I. G., 1991. Hábitos alimentares do peixe-boi da Amazônia (Trichechus inunguis, Mammalia: Sirenia). Dissertation, Instituto Nacional de Pesquisas da Amazônia.
Colares, I. G. & E. P. Colares, 2002. Food plants eaten by Amazonian Manatees (Trichechus inunguis, Mammalia: Sirenia). Brazilian Archives of Biology and Technology 45: 67–72.
Crema, L. C. 2017. Characterization of clear and blackwaters igapós and their food availabilities for the Amazon manatee (Trichechus inunguis). Thesis, Nacional Institute of Amazonian Researches.
da Silva, V. M. F., F. C. W. Rosas & A. M. Cantanhede, 2008. Peixe-boi da Amazônia, Trichechus inunguis (Natterer, 1883). In Machado, A. B. M.; G. M. Drummond & A. P. Paglia (eds) Livro Vermelho da Fauna Brasileira Ameaçada de Extinção. Ministério do Meio Ambiente e Fundação Biodiversitas, Belo Horizonte: 816–818.
da Silveira, E. K. P., 1998. O manejo dos manatis do caribe, Trichechus m. manatus Linné, 1758, e da Amazônia T. inunguis (Natterer, 1883) em cativeiro e alguns aspectos de sua história natural. Boletim da Fundação Brasileira de Conservação da Natureza 23: 82–103.
Deniro, M. J. & M. J. Schieninger, 1983. Stable carbon and nitrogen isotope ratios of bone collagem: variations within individuals, between sexes, and within populations raised on monotonous diets. Journal of Archaeological Science 10: 199–203.
Deutsch, C. J., J. P. Reid, R. K. Bonde, D. E. Easton, H. I Kochman & T. J. O’Shea, 2003. Seasonal movements, migratory behavior, and site fidelity of West Indian manatees along the Atlantic coast of the United States. Wildlife Monographs: 1–77.
Domning, D. P., 1981. Distribution and status of manatees Trichechus spp. near the mouth of the Amazon river, Brazil. Biological Conservation 19: 85–97.
Domning, D. P., 1983. Marching teeth of the manatee. Natural History 92: 8–11.
Domning, D. P. & D. M. Magor, 1978. Taxa horizontal de substituição de dentes de peixe-boi. Acta Amazonica 7: 435–438.
Domning, D. P. & L. C. Hayek, 1984. Horizontal tooth replacement in the Amazonian manatee (Trichechus inunguis). Mammalia 48: 105–127.
Doyle, R. D., 1991. Primary production and nitrogen cycling within the periphyton community associated with emergent aquatic macrophytes in an Amazon floodplain lake. Thesis, University of Maryland.
Drago, M., E. A. Crespo, A. Aguilar, L. Cardona, N. García, S. L. Dans & N. Goodal, 2009. Historic diet change of the South American sea lion in Patagonia as revealed by isotopic analysis. Marine Ecology Progress Series 384: 273–286.
Drago, M., V. Franco-Trecu, L. Cardona & P. Inchausti, 2015. Diet-to-female and female-to-pup isotopic discrimination in South American sea lions. Rapid Communications in Mass Spectrometry 29: 1513–1520.
Duncan, W. P. & M. N. Fernandes, 2010. Physicochemical characterization of the white, black, and Clearwater rivers of the Amazon Basin and its implications on the distribution of freshwater stingrays (Chondrichthyes, Potamotrygonidae). Panajamjas 5: 454–464.
Ferreira, L. V., 2000. Effect of flooding duration on species richness, floristic composition and forest structure in river margin habitats in Amazonian blackwater floodplain forests: implications for future design of protected areas. Biodiversity and Conservation 9: 1–14.
Ferreira C. S., M. T. F. Piedade, A. O. Wittmann & A. C. Franco, 2010. Plant reproduction in the Central Amazonian floodplains: challenges and adaptations. AoB PLANTS.
Fittkau, E. J., U. Irmler, W. J. Junk, F. Reiss & G. W. Schmidt, 2012. Productivity, biomass and population dynamics in Amazonian water bodies. In Golley, F. B. & D. Medina (eds), Tropical ecological systems: trends in terrestrial and aquatic research, Vol. 11. Springer, New York: 289–311.
Franzini, A., N. Castelblanco-Martinez, F. C. W. Rosas & V. M. F. da Silva, 2013. What do local people know about Amazonian manatees? Traditional ecological knowledge of Trichechus inunguis in the oil province of Urucu, AM, Brazil. Brazilian Journal of Nature Conservation 11: 75–80.
Furch, K. & W. J. Junk, 1992. Nutrient dynamics of submersed decomposing Amazonian herbaceous plant species Paspalum fasciculatum and Echinochloa polystachya. Revue d’Hydrobiolgie Tropicale 25: 75–85.
Furch, K. & W. J. Junk, 1997. Chemical composition, food value and decomposition of herbaceous plants leaves and leaf litter of the floodplain forest. In Junk, W. J. (ed.), The Central Amazon Floodplain: Ecological Studies. Springer, Berlin: 187–203.
Goulding, M., M. L. Carvalho & E. G. Ferreira, 1988. Rio Negro: rich life in poor water. SBP Academic Publishing, The Hague.
Guterres-Pazin, M., M. Marmontel, F. C. W. Rosas, V. F. V. Pazin & E. M. Venticinque, 2014. Feeding ecology of the Amazonian Manatee (Trichechus inunguis) in the Mamirauá and Amanã sustainable development reserves, Brazil. Aquatic Mammals 40: 139–149.
Han, H., W. Wei, Y. Nie, W. Zhou, Y. Hu, Q. Wu & F. Wei, 2016. Distinctive diet-tissue isotopic discrimination factors derived from the exclusive bamboo-eating giant panda. Integrative Zoology 11: 447–456.
Hanson, N. N., C. M. Wurster, M. I. Bird, K. Reid & I. L. Boyd, 2009. Intrinsic and extrinsic forcing in life histories: patterns of growth and stable isotopes in male Antarctic fur seal teeth. Marine Ecology Progress Series 388: 263–272.
Hartman, D. S. 1971. Behavior and Ecology of Florida manatee, Trichechus manatus latirostris (Harlan), bay Crystal River, Citrus Country. Thesis, Cornell University.
Hobson, K. A. & R. G. Clark, 1992. Assessing avian diets using stable isotopes I: turnover of 13C in tissues. Condor 94: 181–188.
Hobson, K. A., R. T. Alisauskas & R. G. Clark, 1993. Stable nitrogen isotope enrichment in avian tissues due to fasting and nutritional stress: implications for isotopic analyses of diet. Condor 95: 388–394.
Hobson, K. A., B. N. McLellan & J. G. Woods, 2000. Using stable carbon (δ13C) and nitrogen (δ15N) isotopes to infer trophic relation-ships among black and grizzly bears in the upper Columbia River basin, British Columbia. Canadian Journal of Zoology 78: 1332–1339.
Hobson, K. A. & T. W. Quirk, 2014. Effect of age and ration on diet tissue isotopic (δ13C, δ15N) discrimination in striped skunks (Mephitis mephitis). Isotopes in Environmental and Health Studies 50: 300–306.
Hoson, O., S. Kawada & S. Oda, 2012. Ossification patterns of basicranial sutures in manatees, genus Trichechus. Mammal Study 37: 213–225.
IUCN, 2016. The International Union for Conservation of Nature Red List of Threatened Species. Version 2016-2. http://www.iucnredlist.org. Accessed 21 October 2016.
Jenkins, S. G., S. T. Partridge, T. R. Stephenson, S. D. Farley & C. T. Robbins, 2001. Nitrogen and carbon isotope fractionation between mothers, neonates, and nursing offspring. Oecologia 129: 336–341.
Jim, S. 2000. The development of bone cholesterol 13C values as a new source of palaeodietary information: qualitative and quantitative models of its use in conjunction with bone collagen and apatite 13C values. Thesis, University of Bristol.
Junk, W. J., 1983. Ecology of swamps in the Middle Amazon. In Gore, A. J. P. (org) Mires: Swamp, Bog, Fen and Moor, Regional Studies: Ecosystems of the World, Part B. 1 edn. Elsevier Science Ltd, Amsterdam: 269–294.
Junk, W. J. & M. T. F. Piedade, 1993. Herbaceous plants of the Amazon floodplains near Manaus: species diversity and adaptations to the flood pulse. Amazoniana 12: 467–484.
Junk, W. J. & M. T. F. Piedade, 1997. Plant life in the floodplain with special reference to herbaceous plants. In Junk, W. J. (ed.), The Central Amazon Floodplain: Ecology of a Pulsing System. Ecological Studies. Springer, New York: 147–185.
Junk, W. J. & V. M. F. da Silva, 1997. Mammals, reptiles and amphibians. In Junk, W. J. (ed.) The central Amazon floodplain: ecology of a pulsing system. Ecological Studies, Springer, Berlin: 409–417.
Junk, W. J. & C. Howard-Williams, 1984. Ecology of aquatic macrophytes in Amazonia. In Sioli, H. (org) The Amazon: limnology and landscape ecology of a mighty tropical river and its basin, 1edn. Dordrecht: 269–293.
Junk, W. J., P. B. Bayley & R. E. Sparks, 1989. In Dodge, D. (ed.) Proceedings of the International Large River Symposium. Canadian special publication of fisheries and aquatic sciences 106(1): 110–127.
Kirk, J. T. O., 1994. Light and photosynthesis in aquatic ecosystems, 2nd ed. Cambridge University Press, Cambridge.
Klinge, H., K. Furch, E. Harms & J. Revilla, 1983. Foliar nutrient levels of native tree species from Central Amazonia. Inundation forests. Amazoniana 8: 19–45.
Koch, P. L., 2007. Isotopic study of the biology of modern & fossil vertebrates. In Michener, R. & K. Lajtha (eds), Stable Isotopes in Ecology and Environmental Science, 2nd ed. EUA, Blackwell Publishing, Hoboken: 99–154.
Lefebvre, L. W., J. P. Reid, W. J. Kenworthy & J. A. Powell, 2000. Characterizing manatee habitat use and seagrass grazing in Florida and Puerto Rico: implications for conservation and management. Pacific Conservation Biology 1: 289–298.
Lopes, A., F. Wittmann, J. Schöngart & M. T. F. Piedade, 2014. Herbáceas aquáticas em seis igapós na Amazônia Central: Composição e diversidade de gêneros. Revista Geográfica Acadêmica 8: 5–17.
MacFadden, B. J., P. Higgins, M. T. Clementz & D. S. Jones, 2004. Diets, habitat preferences, and niche differentiation of Cenozoic sirenians from Florida: evidence from stable isotopes. Paleobiology 30: 297–324.
Marmontel, M., T. J. O’Shea, H. I. Kochman & S. R. Humphrey, 1996. Age determination in manatees using growth-layer-group counts in bone. Marine Mammal Science 12: 54–88.
Marshall, B. G., B. R. Forsberg & M. J. F. Thomé-Souza, 2008. Autotrophic energy sources for Paracheirodon axelrodi (Osteichthyes, Characidae) in the middle rio Negro, Central Amazon, Brasil. Hydrobiologia 596: 95–103.
Michener, R. H. & D. M. Schell, 1994. Stable isotope ratios as tracers in marine aquatic food webs. In Lajtha, K. & R. H. Michener (eds), Stable Isotopes in Ecology and Environmental Science. Blackwell Scientific, London: 138–157.
MMA, 2014. Portaria Nº 444 de 17 de dezembro de 2014. Ministério do Meio Ambiente, Brasília.
Nelson, D. E., A. Angerbjörn, K. Lidén & I. Turk, 1998. Stable isotopes and the metabolism of the European cave bear. Oecologia 4: 177–181.
Newsome, S. D., P. L. Koch, M. A. Etnier & D. Aurioles‐Gamboa, 2006. Aurioles-Gamboa, Using carbon and nitrogen isotope values to investigate maternal strategies in northeast Pacific otariids. Marine Mammal Science 1: 556–572.
Newsome, S. D., M. T. Clementz & P. L. Koch, 2010. Using stable isotope biogeochemistry to study marine mammal ecology. Marine Mammal Science 1: 509–572.
Oliveira, A. C. B., M. G. M. Soares, L. A. Martinelli & M. Z. Moreira, 2006. Carbon sources of fish in an Amazonian floodplain lake. Aquatic Science 68: 229–238.
Osmond, C. B., N. Valaane, S. M. Haslam, P. Uotila & Z. Roksandic, 1981. Comparisons of δ13C values in leaves of aquatic macrophytes from different habitats in Britain and Finland: some implications for photosynthetic processes in aquatic plants. Oecologia 50: 117–124.
Pablo-Rodríguez, N., L. D. Olivera-Gómez, D. Aurioles-Gamboa & M. E. Vega-Cendejas, 2016. Seasonal differences in the feeding habits of the Antillean manatee population (Trichechus manatus manatus) in the fluvial-lagoon systems of Tabasco, Mexico. Marine Mammal Science 1: 363–375.
Parnell, A. C., R. Inger, S. Bearhop & A. L. Jackson, 2010. Source partitioning using stable isotopes: coping with too much variation. PLOS ONE 5: e9672.
Parnell, A. C., R. Inger, S & J. Cartwright, 2017. Stable isotope mixing models in R with simmr. https://cran.r-project.org/web/packages/simmr/vignettes/simmr.html.
Parolin, P. & L. V. Ferreira, 1998. Are there differences in specific wood gravities between trees in várzea and igapó (Central Amazonia)? Ecotropica 4: 25–32.
Phillips, D. L., R. Inger, S. Bearhop, A. L. Jackson, J. W. Moore, A. C. Parnell, B. X. Semmens & E. J. Ward, 2014. Best practices for use of stable isotope mixing models in food-web studies. Canadian Journal of Zoology 92: 823–835.
Piedade, M. T. F., W. J. Junk & S. P. Long, 1991. The productivity of the C4 grass Echinochloa polystachya on the Amazon floodplain. Ecology 72: 1456–1463.
Piedade, M. T. F., S. P. Long & W. J. Junk, 1993. Leaf and canopy photosynthetic CO2 uptake of a stand of Echinoclhoa polystachya on the Central Amazon floodplain. Are the high potential rates associated with the C4 syndrome realized under the near-optimal conditions provided by this exceptional natural habitat? Oecologia 97: 193–201.
Piedade, M. T. F., C. S. Ferreira & A. C. Franco, 2010. Estrategias reproductivas de la vegetación y sus respuestas al pulso de la inundación en las zonas inundables de la Amazonía Central. Ecosistemas 19: 52–66.
Polischuk, S. C., K. A. Hobson & M. A. Ramsay, 2001. Use of stable-carbon and nitrogen isotopes to assess weaning and fasting in female polar bears and their cubs. Canadian Journal of Zoology 79: 499–511.
Prance, G. T., 1979. Notes on vegetation of Amazonia. III. The terminology of Amazonian forest type subject to inundation. Brittonia 31: 26–38.
Prance, G. T., 1980. A terminologia dos tipos de florestas amazônicas sujeitas à inundação. Acta Amazonica 10: 495–504.
Putz, R., 1997. Periphyton communities in Amazonian black- and whitewater habitats: Community structure, biomass and productivity. Aquatic Sciences 59: 74–93.
R Development Core Team, 2016. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing, 2014. Retrieved from http://www.R-project.org/.
Reich, K. J. & G. A. J. Worthy, 2006. An isotopic assessment of the feeding habits of free-ranging manatees. Marine Ecology-Progress Series 322: 303–309.
Rosas, F. C. W., 1994. Biology, conservation and status of the Amazonian Manatee Trichechus inunguis. Mammal Review 24: 49–59.
Sand-Jensen, K., 1989. Environmental variables and their effect on photosynthesis of aquatic plant communities. Aquatic Botany 34: 5–25.
Sare, D. T., J. S. Millar & F. J. Longstaffe, 2005. Nitrogen-and carbon-isotope fractionation between mothers and offspring in red-backed voles (Clethrionomys gapperi). Canadian Journal of Zoology 1: 712–716.
Schoeninger, M. J. & M. J. DeNiro, 1984. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochimica et Cosmochimica Acta 48: 625–639.
Schöngart, J., F. Wittmann & M. Worbes, 2010. Biomass and NPP of Central Amazonian floodplain forests. In Junk, W. J., M. T. F. Piedade, F. Wittmann, J. Schongart & P. Parolin (eds), Amazonian floodplain Forests: Ecophysiology, Biodiversity and Sustainable Management. Ecological Studies. Springer, New York: 347–388.
Schwarcz, H. P. & M. J. Schoeninger, 1991. Stable isotope analyses in human nutritional ecology. American Journal of Physical Anthropology 1: 283–321.
Secchi, E. R., S. Botta, M. M. Wiegand, L. A. Lopez, P. F. Fruet, R. C. Genoves & J. C. Di Tullio, 2016. Long-term and gender-related variation in the feeding ecology of common bottlenose dolphins inhabiting a subtropical estuary and the adjacent marine coast in the western South Atlantic. Marine Biology Research 2: 121–134.
Self-Sullivan, C., G. W. Smith, J. M. Packard & K. S. LaCommare, 2003. Seasonal occurrence of male Antillean manatees (Trichechus manatus manatus) on the Belize Barrier Reef. Aquatic Mammals 29: 342–354.
Sheppard, J. K., A. R. Preen, H. Marsh, I. R. Lawler, S. D. Whiting & R. E. Jones, 2006. Movement heterogeneity of Dugongs, Dugong dugon (Muller), over large spatial scales. Journal of Experimental Marine Biology and Ecology 334: 64–83.
Sioli, H., 1950. Das Wasser in Amazonas gebiet. Fosch Fortschr 26: 274–280.
Sioli, H., 1984. The Amazon and its main effluents: Hydrography, morphology of the river courses, and river types. In Sioli, H. (ed.), The Amazon—Limnology and Landscape Ecology of a Mighty Tropical River and its Basin. Junk, Dordrecht: 127–166.
Smith, B. N. & S. Epstein, 1970. Biogeochemistry of the stable isotopes of hydrogen and carbon in salt marsh biota. Plant Physiology 46: 738–742.
Smith, F. A. & N. A. Walker, 1980. Photosynthesis by aquatic plants: effects of unstirred layers in relation to assimilation of CO2 and HCO3− and to carbon isotopic discrimination. New Phytologist 86: 245–259.
Stevens, R. E. & R. E. Hedges, 2004. Carbon and nitrogen stable isotope analysis of northwest European horse bone and tooth collagen, 40,000 BP–present: Palaeoclimatic interpretations. Quaternary Science Reviews 30: 977–991.
Tieszen, L. L. & T. Fagre, 1993. Effect of diet quality and composition on the isotopic composition of respiratory CO2, bone collagen, bioapatite, and soft tissues. In Lambert, J. B. & G. Grupe (eds), Prehistoric Human Bone—Archaeology at the Molecular Level. Springer, Berlin: 121–155.
Troina, G., S. Botta, E. R. Secchi & F. Dehairs, 2016. Ontogenetic and sexual characterization of the feeding habits of franciscanas, Pontoporia blainvillei, based on tooth dentin carbon and nitrogen stable isotopes. Marine Mammal Science 32: 1115–1137.
Tykot, R. H., 2004. Stable isotopes and diet: You are what you eat. In Martini, M., M. Milazzo & M. Piacentini (eds) Physics methods in archaeometry. Proceedings of the International School of Physics “Enrico Fermi.” Bologna: Società Italiana di Fisica: 433–444.
Valdevino, G. C. M., 2016. Variações morfológicas e geográficas no sincrânio do peixe-boi da Amazônia Trichechus inunguis (Natterer, 1883). Dissertation. Universidade Federal do Amazonas, Brasil.
van Klinken, G. J., 1999. Bone collagen quality indicators for palaeodietary and radiocarbon measurements. Journal of Archaeological Science 26: 687–695.
Walker, J. L. & S. A. Macko, 1999. Dietary studies of marine mammals using stable carbon and nitrogen isotopic ratios of teeth. Marine Mammal Science 15: 314–334.
Werner, R. A. & W. A. Brand, 2001. Referencing strategies and techniques in stable isotope ratio analysis. Rapid Communications in Mass Spectrometry 15(7): 501–519.
Witt, G. B. & L. K. Ayliffe, 2001. Carbon isotope variability in the bone collagen of red kangaroos (Macropus rufus) is age dependent: implications for palaeodietary studies. Journal of Archaeological Science 1: 247–252.
Zaniolo, G. R., 2006. Etnoconhecimento do peixe-boi amazônico Trichechus inunguis (Natterer, 1883) pelos pescadores de Novo Airão, Amazonas, Brasil. Dissertation, Universidade Federal do Amazonas.
Acknowledgements
We thank the Instituto Chico Mendes de Conservação da Biodiversidade for project funding, and for granting research permits. We are deeply grateful to the Isolab from Max Planck Institute for Biogeochemistry, especially Dr. Heiko Moossen and Heike Geilmann. Moreover, we thank the Laboratório de Mamíferos Aquáticos from Instituto Nacional de Pesquisas da Amazônia, especially MSc Gisele Valdevino; the Centro Nacional Pesquisa e Conservação de Mamíferos Aquáticos; and the Zoológico da Faculdade de Amazônia—Santarém, especialy Jairo Moura and Sidicley Matos. We thank also all people from estudied protected areas, residents and managers. We are grateful to Dr. Ulrich Struk and Marianne Grønholdt Falk, from Museum für Naturkunde Berlin, for the reception and advices. We also thank Dr. Cristiano Albuquerque, Dr. Rodrigo Amaral, Dra. Joana D’Arc de Paula, Dr. Charles Clement, and Dr. Eduardo Rios-Villamizar who reviewed an earlier version of this paper. This article is part of LCC’s Ph.D. thesis under the supervision of MTFP and VMFS. We are grateful for all suggestions given by the reviewer and editor of Hydrobiologia. This research received additional financial support from the Long Term Research Project of the Grupo Ecologia Monitoramento e Uso Sustentável de Áreas Úmidas (PELD-MAUA), Projeto Adaptações da Biota Aquática Amazônica (ADAPTA), and from the Associação Amigos do Peixe-boi and Projeto Petrobras Ambiental.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: David Hoeinghaus
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Crema, L.C., da Silva, V.M.F., Botta, S. et al. Does water type influence diet composition in Amazonian manatee (Trichechus inunguis)? A case study comparing black and clearwater rivers. Hydrobiologia 835, 1–19 (2019). https://doi.org/10.1007/s10750-019-3900-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-3900-4