Abstract
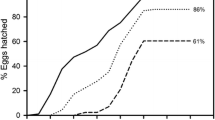
In the rotifer Brachionus calyciflorus mictic-female production is density-dependent and appears to be induced by a chemical—a quorum sensing molecule—produced by the females themselves. Even at the highest densities, however, populations never become entirely mictic: i.e., some amictic females continue to be produced. Surprisingly, the phenomenon also occurs in clonal laboratory populations with genetically identical individuals. Here, we study how this ecologically adaptive phenomenon is generated at the level of individual reproducing females. In a life-history experiment we subjected 123 amictic females of a clone of B. calyciflorus separately to a daily renewed stimulus of culture medium conditioned at a density of 30 females ml−1. For each of these mothers we isolated the lifetime offspring individually and recorded whether these females were amictic or mictic. Mothers produced on average 16 offspring but none of the mothers produced 100% mictic offspring; the average proportion of mictic females was 30%, despite the extremely strong stimulus. The distribution of amictic vs. mictic offspring was not uniform over the mothers’ lifetime. Early and late offspring had a low probability of being mictic whereas mid-aged mothers produced the highest proportion of mictic daughters (up to 56%). We conclude that not all oocytes of B. calyciflorus can be turned into mictic females, even when the mictic-female-inducing stimulus is extremely high. Propensity to become a mictic female also depends on the rank of an egg within a female’s offspring production. Despite these regularities, we observed considerable stochastic variability with respect to individual mothers’ life histories.





Similar content being viewed by others
References
Carmona, M. J., M. Serra & M. R. Miracle, 1993. Relationships between mixis in Brachionus plicatilis and preconditioning of culture medium by crowding. Hydrobiologia 255: 145–152.
Fussmann, G. F., S. P. Ellner & N. G. Hairston, 2003. Evolution as a critical component of plankton dynamics. Proceedings of the Royal Society of London Series B-Biological Sciences 270: 1015–1022.
Gilbert, J. J., 1963. Mictic female production in the rotifer Brachionus calyciflorus. Journal of Experimental Zoology 153: 113–124.
Gilbert, J. J., 2003a. Environmental and endogenous control of sexuality in a rotifer life cycle: developmental and population biology. Evolution & Development 5: 19–24.
Gilbert, J. J., 2003b. Specificity of crowding response that induces sexuality in the rotifer Brachionus. Limnology and Oceanography 48: 1297–1303.
Gilbert, J. J. & T. Schröder, 2004. Rotifers from diapausing, fertilized eggs: unique features and emergence. Limnology and Oceanography 49: 1341–1354.
Gilbert, J. J. & T. Schröder, 2007. Intraclonal variation in propensity for mixis in several rotifers: variation among females and with maternal age. Hydrobiologia doi:10.1007/s10750-007-9040-2.
Gotelli, N. J. & A. M. Ellison, 2004. A Primer of Ecological Statistics. Sinauer, Sunderland, MA.
Kussell, E. & S. Leibler, 2005. Phenotypic diversity, population growth, and information in fluctuating environments. Science 309: 2075–2078.
Lubzens, E. & G. Minkoff, 1988. Influence of the age of algae fed to rotifers (Brachionus plicatilis O. F. Müller) on the expression of mixis in their progenies. Oecologia 75: 430–435.
Philippi, T. & J. Seger, 1989. Hedging ones evolutionary bets, revisited. Trends in Ecology & Evolution 4: 41–44.
Pourriot, R., 1963. Influence du rythme nycthemeral sur le cycle sexuel de quelques rotifères. Comptes Rendus Hebdomadaires des Séances de l’Académie des Sciences 256: 5216–5219.
Pourriot, R., 1965. Sur le déterminisme du mode de reproduction chez les rotifères. Schweizerische Zeitschrift für Hydrologie 27: 76–87.
Pourriot, R. & C. Rougier, 1976. Influence de l’âge des parents sur la production de femelles mictiques chez Brachionus calyciflorus (Pallas) et B. rubens Ehr. (Rotifères). Comptes Rendus Hebdomadaires des Séances de l’Académie des Sciences Série D 283: 1497–1500.
Pourriot, R. & C. Rougier, 1986. Rhythmes de production de femelles sexuées chez le rotifère Brachionus calyciflorus, en élevage à température constante. Bulletin de la Societé Zoologique de France 111: 203–209.
Raser, J. M. & E. K. O’Shea, 2005. Noise in gene expression: origins, consequences, and control. Science 309: 2010–2013.
Rougier, C. & R. Pourriot, 1977. Aging and control of reproduction in Brachionus calyciflorus (Pallas) (Rotatoria). Experimental Gerontology 12: 137–151.
Ruttner-Kolisko, A., 1964. Űber die labile Periode im Fortpflanzungszyklus der Rädertiere. Internationale Revue der gesamten Hydrobiologie 49: 473–482.
Serra, M. & C. E. King, 1999. Optimal rates of bisexual reproduction in cyclical parthenogens with density-dependent growth. Journal of Evolutionary Biology 12: 263–271.
Snell, T. W. & E. M. Boyer, 1988. Thresholds for mictic female production in the rotifer Brachionus plicatilis (Muller). Journal of Experimental Marine Biology and Ecology 124: 77–85.
Snell, T. W., J. Kubanek, W. Carter, A. B. Payne, J. Kim, M. K. Hicks & C. P. Stelzer, 2006. A protein signal triggers sexual reproduction in Brachionus plicatilis (Rotifera). Marine Biology 149: 763–773.
Stelzer, C. P. & T. W. Snell, 2003. Induction of sexual reproduction in Brachionus plicatilis (Monogononta, Rotifera) by a density-dependent chemical cue. Limnology and Oceanography 48: 939–943.
Wallace, R. L., T.W. Snell, C. Ricci & T. Nogrady, 2006. In Segers, H. (ed.) Rotifera: Volume 1 Biology, Ecology and Systematics, 2nd edn. Guides to the Identification of the Microinvertebrates of the Continental Waters of the World 23. Kenobi Productions, Ghent, and Backhuys Publishers, Leiden.
Acknowledgements
We acknowledge financial support through an NSERC discovery grant to GFF. We thank Mark Romer for growth chamber set-up.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: S. S. S. Sarma, R. D. Gulati, R. L. Wallace, S. Nandini, H. J. Dumont & R. Rico-Martínez.
Advances in Rotifer Research.
Rights and permissions
About this article
Cite this article
Fussmann, G.F., Kramer, G. & Labib, M. Incomplete induction of mixis in Brachionus calyciflorus: patterns of reproduction at the individual level. Hydrobiologia 593, 111–119 (2007). https://doi.org/10.1007/s10750-007-9041-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-007-9041-1