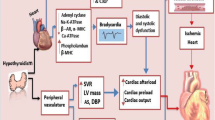
Abstract
The evolution of cardiac disease after an acute ischemic event depends on a complex and dynamic network of mechanisms alternating from ischemic damage due to acute coronary occlusion to reperfusion injury due to the adverse effects of coronary revascularization till post-ischemic remodeling. Cardioprotection is a new purpose of the therapeutic interventions in cardiology with the goal to reduce infarct size and thus prevent the progression toward heart failure after an acute ischemic event. In a complex biological system such as the human one, an effective cardioprotective strategy should diachronically target the network of cross-talking pathways underlying the disease progression. Thyroid system is strictly interconnected with heart homeostasis, and recent studies highlighted its role in cardioprotection, in particular through the preservation of mitochondrial function and morphology, the antifibrotic and proangiogenetic effect and also to the potential induction of cell regeneration and growth. The objective of this review was to highlight the cardioprotective role of triiodothyronine in the complexity of post-ischemic disease evolution.


Similar content being viewed by others
Abbreviations
- HF:
-
Heart failure
- CV:
-
Cardiovascular
- TH:
-
Thyroid hormone
- ATP:
-
Adenosine triphosphate
- H+ :
-
Hydrogen ions
- PKC:
-
Protein kinase C
- ERK:
-
Extracellular signal-regulated kinases
- AKT:
-
Protein kinase B
- ROS:
-
Reactive oxygen species
- I/R:
-
Ischemic/reperfusion
- BCL-2:
-
B cell lymphoma-2
- PTP:
-
Permeability transition pore
- PTPO:
-
Opening of the PTP
- L-T3S:
-
Low T3 syndrome
- AMI:
-
Acute myocardial infarction
- T3:
-
Triiodothyronine
- rT3:
-
Reverse T3
- TRs:
-
TH-specific receptors
- p38MAPK:
-
p38 mitogen-activated protein kinases
- HSP:
-
Heat-shock protein
- p53:
-
Tumor suppressor protein
- PI3K:
-
Phosphoinositide 3-kinase
- T4:
-
Tiroxine
- MMPs:
-
Metalloproteinases
- TIMPs:
-
Tissue inhibitors of MMPs
- TGF:
-
Transforming growth factor
- bFGF:
-
Fibroblastic growth factor
- VEGF:
-
Vascular endothelial growth factor
- HIF-1α:
-
Hypoxia-inducible factor 1 alpha
- mTOR:
-
Mammalian target of rapamycin
- MHC:
-
Myosin heavy chain
References
Van de Werf F, Bax J, Betriu A, Blomstrom-Lundqvist C, Crea F, Falk V et al (2008) ESC committee for practice guidelines (CPG). Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: the task force on the management of ST-segment elevation acute myocardial infarction of the European society of cardiology. Eur Heart J 29:2909–2945. doi:10.1093/eurheartj/ehn416
Gerber Y, Weston SA, Redfield MM, Chamberlain AM, Manemann SM, Jiang R, Killian JM, Roger VL (2015) A contemporary appraisal of the heart failure epidemic in Olmsted County, Minnesota, 2000–2010. JAMA Intern Med 175:996. doi:10.1001/jamainternmed.2015.0924
Gerber Y, Weston SA, Enriquez-Sarano M, Berardi C, Chamberlain AM, Manemann SM, Jiang R, Dunlay SM, Roger VL (2016) Mortality associated with heart failure after myocardial infarction: a contemporary community perspective. Circ Heart Fail 9:e002460. doi:10.1161/CIRCHEARTFAILURE.115.002460
West BJ, Geneston EL, Grigolini P (2008) Maximizing information exchange between complex networks. Phys Rep 468:1–99. doi:10.1016/j.physrep.2008.06.003
Heusch G (2015) Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116:674–699. doi:10.1161/CIRCRESAHA.116.305348
Perricone AJ, Vander Heide RS (2014) Novel therapeutic strategies for ischemic heart disease. Pharmacol Res 89:36–45
Altamirano F, Wang ZV, Hill JA (2015) Cardioprotection in ischemia-reperfusion injury: novel mechanisms and clinical translation. J Physiol 593:3773–3788. doi:10.1113/JP270953
Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD (2012) Third universal definition of myocardial infarction. Circulation 126:2020–2035. doi:10.1161/CIR.0b013e31826e1058
Sanada S, Komuro I, Kitakaze M (2011) Pathophysiology of myocardial reperfusion injury: preconditioning, postconditioning, and translational aspects of protective measures. Am J Physiol Heart Circ Physiol 301:H1723–H1741. doi:10.1152/ajpheart.00553.2011
Marín-García J (2008) Goldenthal. Mitochondrial centrality in heart failure. J Heart Fail Rev 13:137–150. doi:10.1007/s10741-007-9079-1
Whelan RS, Kaplinskiy V, Kitsis RN (2010) Cell death in the pathogenesis of heart disease mechanisms and significance. Annu Rev Physiol 72:19–44. doi:10.1146/annurev.physiol.010908.163111
Baines CP (2009) The mitochondrial permeability transition pore and ischemia-reperfusion injury. Basic Res Cardiol 104:181–188. doi:10.1007/s00395-009-0004-8
Galluzzi L, Kepp O, Kroemer G (2012) Mitochondria: master regulators of danger signaling. Nat Rev Mol Cell Biol 13:780–788. doi:10.1038/nrm3479
Baines CP (2010) The cardiac mitochondrion: nexus of stress. Annu Rev Physiol 72:61–80. doi:10.1146/annurev-physiol-021909-135929
Brady NR, Hamacher-Brady A, Gottlieb RA (2006) Proapoptotic BCL-2 family members and mitochondrial dysfunction during ischemia/reperfusion injury, a study employing cardiac HL-1 cells and GFP biosensors. Biochim Biophys Acta 1757:667–678
Zorov DB, Filburn CR, Klotz LO, Zweier JL, Sollott SJ (2000) Reactive oxygen species (ROS)-induced ROS release: a new phenomenon accompanying induction of the mitochondrial permeability transition in cardiac myocytes. J Exp Med 192:1001–1101
Assaly R, de d’Anglemont TA, Paradis S, Jacquin S, Berdeaux A, Morin D (2012) Oxidative stress, mitochondrial permeability transition pore opening and cell death during hypoxia-reoxygenation in adult cardiomyocytes. Eur J Pharmacol 675:6–14. doi:10.1016/j.ejphar.2011.11.036
Garcia-Dorado D, Ruiz-Meana M, Inserte J, Rodriguez-Sinovas A, Piper HM (2012) Calcium-mediated cell death during myocardial reperfusion. Cardiovasc Res 94:168–180. doi:10.1093/cvr/cvs116
Pfeffer MA (1995) Left ventricular remodeling after acute myocardial infarction. Annu Rev Med 46:455–466
Gerdes AM, Kellerman SE, Moore JA, Muffly KE, Clark LC, Reaves PY, Malec KB, McKeown PP, Schocken DD (1992) Structural remodelling of cardiac myocytes in patients with ischemic cardiomyopathy. Circulation 86:426–430
Daly PA, Sole MJ (1990) Myocardial catecholamines and the pathophysiology of heart failure. Circulation 82(2 Suppl):I35–I43
Wong GH, Goeddel DV (1998) Induction of manganous superoxide dismutase by tumor necrosis factor: possible protective mechanism. Science 242:941–944
Finkel MS, Oddis CV, Jacob TD et al (1992) Negative inotropic effects of cytokines on the heart mediated by nitric oxide. Science 257:387–389
Li L, Guo CY, Yang J, Jia EZ, Zhu TB, Wang LS, Cao KJ, Ma WZ, Yang ZJ (2011) Negative association between free triiodothyronine level and international normalized ratio in euthyroid subjects with acute myocardial infarction. Acta Pharmacol Sin 32:1351–1356. doi:10.1038/aps.2011.118
Friberg L, Werner S, Eggetsen G, Ahnve S (2002) Rapid down-regulation of thyroid hormones in acute myocardial infarction: Is it cardioprotective in patients with angina? Arch Int Med 162:1388–1394
Kimur T, Kotajima N, Kanda T, Kuwabara A, Fukumura Y, Kobayashi I (2001) Correlation of circulating interleukin-10 with thyroid hormone in acute myocardial infarction. Res Commun Mol Pathol Pharmacol 110:63–68
Friberg L, Drvota V, Bjelak AH, Eggertsen G, Ahnve S (2001) Association between increased levels of reverse triiodothyronine and mortality after acute myocardial infarction. Am J Med 111:699–703
Forini F, Lionetti V, Ardehali H, Pucci A, Cecchetti F, Ghanefar M, Nicolini G, Ichikawa Y, Nannipieri M, Recchia FA, Iervasi G (2011) Early long-term L-T3 replacement rescues mitochondria and prevents ischemic cardiac remodelling in rats. J Cell Mol Med 15:514–524. doi:10.1111/j.1582-4934.2010.01014.x
Forini F, Paolicchi A, Pizzorusso T, Ratto GM, Saviozzi M, Vanini V, Iervasi G (2001) 3,5,3′-triiodothyronine deprivation affects phenotype and intracellular [Ca2+]i of human cardiomyocytes in culture. Cardiovasc Res 51:322–330
Pingitore A, Chen Y, Gerdes AM, Iervasi G (2012) Acute myocardial infarction and thyroid function: new pathophysiological and therapeutic perspectives. Ann Med 44:745–757. doi:10.3109/07853890.2011.573501
Cheng SY, Leonard JL, Davis PJ (2010) Molecular aspects of thyroid hormone actions. Endocr Rev 31:139–170
Pantos C, Mourouzis I (2014) The emerging role of TRα1 in cardiac repair: potential therapeutic implications. Oxid Med Cell Longev 2014:481482. doi:10.1155/2014/481482
Chassandre O (2003) Do unliganded thyroid hormone receptors have physiological functions? Endocrinology 31:9–20
Mourouzis I, Forini F, Pantos C, Iervasi G (2011) Thyroid hormone and cardiac disease: from basic concepts to clinical application. J Thyroid Res 2011:958626. doi:10.4061/2011/958626
Pantos C, Mourouzis I, Saranteas T, Brozou V, Galanopoulos G, Kostopanagiotou G, Cokkinos DV (2011) Acute T3 treatment protects the heart against ischemia-reperfusion injury via TRα1 receptor. Mol Cell Biochem 353:235–241. doi:10.1007/s11010-011-0791-8
Mourouzis I, Kostakou E, Galanopoulos G, Mantzouratou P, Pantos C (2013) Inhibition of thyroid hormone receptor α1 impairs post-ischemic cardiac performance after myocardial infarction in mice. Mol Cell Biochem 379:97–105. doi:10.1007/s11010-013-1631-9
Engel FB, Schebesta M, Duong MT, Lu G, Ren S, Madwed JB, Jiang H, Wang Y, Keating MT (2005) p38 MAP kinase inhibition enables proliferation of adult mammalian cardiomyocytes. Genes Dev 19:1175–1187
Mourouzis I, Mantzouratou P, Galanopoulos G, Kostakou E, Roukounakis N, Kokkinos AD, Cokkinos DV, Pantos C (2012) Dose-dependent effects of thyroid hormone on post-ischemic cardiac performance: potential involvement of Akt and ERK signaling. Mol Cell Biochem 363:235–243. doi:10.1007/s11010-011-1175-9
Chen YF, Kobayashi S, Chen J, Redetzke RA, Said S, Liang Q, Gerdes AM (2008) Short term triiodo-L-thyronine treatment inhibits cardiac myocyte apoptosis in border area after myocardial infarction in rats. J Mol Cell Cardiol 44:180–187
Kuzman JA, Gerdes AM, Kobayashi S, Liang Q (2005) Thyroid hormone activates Akt and prevents serum starvation-induced cell death in neonatal rat cardiomyocytes. J Mol Cell Cardiol 39:841–844
Rybin V, Steinberg SF (1996) Thyroid hormone represses protein kinase C isoform expression and activity in rat cardiac myocytes. Circ Res 79:388–398
Pantos C, Mourouzis I, Saranteas T, Clavé G, Ligeret H, Noack-Fraissignes P et al (2009) Thyroid hormone improves postischaemic recovery of function while limiting apoptosis: a new therapeutic approach to support hemodynamics in the setting of ischaemia-reperfusion? Basic Res Cardiol 104:69–77. doi:10.1007/s00395-008-0758-4
Pantos C, Malliopoulou V, Paizis I, Moraitis P, Mourouzis I, Tzeis S, Karamanoli E, Cokkinos DD, Carageorgiou H, Varonos D, Cokkinos DV (2003) Thyroid hormone and cardioprotection: study of p38 MAPK and JNKs during ischaemia and at reperfusion in isolated rat heart. Mol Cell Biochem 242:173–180
Kehat I, Davis J, Tiburcy M, Accornero F, Saba-El-Leil MK, Maillet M et al (2010) Extracellular signal-regulated kinases 1 and 2 regulate the balance between eccentric and concentric cardiac growth. Circ Res 108:176–183. doi:10.1161/CIRCRESAHA.110.231514
Wrutniak-Cabello C, Casas F, Cabello G (2001) Thyroid hormone action in mitochondria. J Mol Endocrinol 26:67–77
Goldenthal MJ, Ananthakrishnan R, Marín-García J (2005) Nuclear-mitochondrial cross-talk in cardiomyocyte T3 signaling: a time-course analysis. J Mol Cell Cardiol 39:319–326
Marín-García J (2010) Thyroid hormone and myocardial mitochondrial biogenesis. Vasc Pharmacol 52:120–130. doi:10.1016/j.vph.2009.10.008
Forini F, Ucciferri N, Kusmic C, Nicolini G, Cecchettini A, Rocchiccioli S, Citti L, Iervasi G (2015) Low T3 state is correlated with cardiac mitochondrial impairments after ischemia reperfusion injury: evidence from a proteomic approach. Int J Mol Sci 16:26687–26705. doi:10.3390/ijms161125973
Forini F, Kusmic C, Nicolini G, Mariani L, Zucchi R, Matteucci M, Iervasi G, Pitto L (2014) Triiodothyronine prevents cardiac ischemia/reperfusion mitochondrial impairment and cell loss by regulating miR30a/p53 axis. Endocrinology 155:4581–4590. doi:10.1210/en.2014-1106
Spinale FG (2007) Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiol Rev 87:1285–1342
Collen A, Hanemaaijer R, Lupu F, Quax PH, van Lent N, Grimbergen J, Peters E, Koolwijk P, van Hinsbergh VW (2003) Membrane-type matrix metalloproteinase-mediated angiogenesis in a fibrin-collagen matrix. Blood 101:1810–1817
Givvimani S, Tyagi N, Sen U, Mishra PK, Qipshidze N, Munjal C, Vacek JC, Abe OA, Tyagi SC (2010) MMP-2/TIMP-2/TIMP-4 versus MMP-9/TIMP-3 in transition from compensatory hypertrophy and angiogenesis to decompensatory heart failure. Arch Physiol Biochem 116:63–72. doi:10.3109/13813451003652997
Lindsey ML, Zamilpa R (2012) Temporal and spatial expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases following myocardial infarction. Cardiovasc Ther 30:31–41. doi:10.1111/j.1755-5922.2010.00207.x
Wilson EM, Moainie SL, Baskin JM, Lowry AS, Deschamps AM, Mukherjee R, Guy TS, St John-Sutton MG, Gorman MGJH, Edmunds LH, Gorman RC, Spinale FG (2003) Region- and type-specific induction of matrix metalloproteinases in post-myocardial infarction remodeling. Circulation 107:2857–2863
Chen YF, Weltman NY, Li X, Youmans S, Krause D, Gerdes AM (2013) Improvement of left ventricular remodeling after myocardial infarction with 8 weeks L-thyroxine treatment in rats. J Transl Med 11:40. doi:10.1186/1479-5876-11-40
Weltman NY, Pol CJ, Zhang Y, Wang Y, Koder A, Raza S, Zucchi R, Saba A, Colligiani D, Gerdes AM (2015) Long-term physiological T3 supplementation in hypertensive heart disease in rats. Am J Physiol Heart Circ Physiol 309:H1059–H1065. doi:10.1152/ajpheart.00431.2015
Ziegelhöffer-Mihalovicová B, Briest W, Baba HA, Rassler B, Zimmer HG (2003) The expression of mRNA of cytokines and of extracellular matrix proteins in triiodothyronine-treated rat hearts. Mol Cell Biochem 247:61–68
Ghose Roy S, Mishra S, Ghosh G, Bandyopadhyay A (2007) Thyroid hormone induces myocardial matrix degradation by activating matrix metalloproteinase-1. Matrix Biol 26:269–279
Nicolini G, Forini F, Kusmic C, Pitto L, Mariani L, Iervasi G (2015) Early and short-term triiodothyronine supplementation prevents adverse post-ischemic cardiac remodeling: role of transforming growth factor-β1 and anti-fibrotic miRNA signaling. Mol Med. doi:10.2119/molmed.2015.00140
Liu Y, Sherer BA, Redetzke RA, Gerdes AM (2010) Regulation of arteriolar density in adult myocardium during low thyroid conditions. Vasc Pharmacol 52:146–150. doi:10.1016/j.vph.2009.10.003
Savinova OV, Liu Y, Aasen GA, Mao K, Weltman NY, Nedich BL, Liang Q, Gerdes AM (2011) Thyroid hormone promotes remodeling of coronary resistance vessels. PLoS One 6:e25054. doi:10.1371/journal.pone.0025054
Gerdes AM (2015) Restoration of thyroid hormone balance: a game changer in the treatment of heart failure? Am J Physiol Heart Circ Physiol 308(1):H1–H10. doi:10.1152/ajpheart.00704.2014
Balzan S, Del Carratore R, Nicolini G, Beffy P, Lubrano V, Forini F, Iervasi G (2012) Proangiogenic effect of TSH in human microvascular endothelial cells through its membrane receptor. J Clin Endocrinol Metab 97:1763–1770. doi:10.1210/jc.2011-2146
Eckle T, Köhler D, Lehmann R, El Kasmi K, Eltzschig HK (2008) Hypoxia-inducible factor-1 is central to cardioprotection: a new paradigm for ischemic preconditioning. Circulation 118:166–175. doi:10.1161/CIRCULATIONAHA.107.758516
Moeller LC, Dumitrescu AM, Refetoff S (2005) Cytosolic action of thyroid hormone leads to induction of hypoxia-inducible factor-1alpha and glycolytic genes. Mol Endocrinol 19:2955–29634
Makino A, Suarez J, Wang H, Belke DD, Scott BT, Dillmann WH (2009) Thyroid hormone receptor-beta is associated with coronary angiogenesis during pathological cardiac hypertrophy. Endocrinology 150:2008–2015
Taegtmeyer S, Sen S, Vela D (2010) Return to the fetal gene program: a suggested metabolic link to gene expression in the heart. Ann NY Acad Sci 1188:191–198. doi:10.1111/j.1749-6632.2009.05100.x
Pantos C, Mourouzis I, Dimopoulos A, Markakis K, Panagiotou M, Xinaris C, Tzeis S, Kokkinos AD, Cokkinos DV (2007) Enhanced tolerance of the rat myocardium to ischemia and reperfusion injury early after acute myocardial infarction. Basic Res Cardiol 102:327–333
Pantos C, Mourouzis I, Saranteas T, Paizis I, Xinaris C, Malliopoulou V, Cokkinos DV (2005) Thyroid hormone receptors alpha1 and beta1 are downregulated in the post-infarcted rat heart: consequences on the response to ischaemia-reperfusion. Basic Res Cardiol 100:422–432
Kinugawa K, Jeong MY, Bristow MR, Long CS (2005) Thyroid hormone induces cardiac myocyte hypertrophy in a thyroid hormone receptor alpha1-specific manner that requires TAK1 and p38 mitogen-activated protein kinase. Mol Endocrin 19:1618–1628
Tavi P, Sjögren M, Lunde PK, Zhang SJ, Abbate F, Vennström B, Westerblad H (2005) J Mol Cell Cardiol 38:655–663
Pantos C, Xinaris C, Mourouzis I (2008) Thyroid hormone receptor alpha 1: a switch to cardiac cell “metamorphosis”? J Physiol Pharmacol 59:253–269
Rolfe M, McLeod LE, Pratt PF, Proud CG (2005) Activation of protein synthesis in cardiomyocytes by the hypertrophic agent phenylephrine requires the activation of ERK and involves phosphorylation of tuberous sclerosis complex 2 (TSC2). Biochemical J 388:973–984
Pantos C, Mourouzis I, Galanopoulos G, Gavra M, Perimenis P, Spanou D, Cokkinos DV (2010) Thyroid hormone receptor alpha1 downregulation in postischemic heart failure progression: the potential role of tissue hypothyroidism. Horm Metab Res 42:718–724. doi:10.1055/s-0030-1255035
van Rooij E, Quiat D, Johnson BA, Sutherland LB, Qi X, Richardson JA, Kelm RJ Jr, Olson EN (2009) A family of microRNAs encoded by myosin genes governs myosin expression and muscle performance. Dev Cell 17:662–673. doi:10.1016/j.devcel.2009.10.013
Callis TE, Pandya K, Seok HY, Tang RH, Tatsuguchi M, Huang ZP, Chen JF, Deng Z, Gunn B, Shumate J, Willis MS, Selzman CH, Wang DZ (2009) MicroRNA-208a is a regulator of cardiac hypertrophy and conduction in mice. J Clin Invest 119:2772–2786. doi:10.1172/JCI36154
Sabatino L, Iervasi G, Pingitore A (2015) Thyroid hormone and heart failure: from myocardial protection to systemic regulation. Expert Rev Cardiovasc Ther 13:733–743. doi:10.1586/14779072.2015.1043094
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors of the paper “Cardioprotection and Thyroid Hormones,” Alessandro Pingitore, Giuseppina Nicolini, Claudia Kusmic, Giorgio Iervasi, Paolo Grigolini and Simona Forini declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Pingitore, A., Nicolini, G., Kusmic, C. et al. Cardioprotection and thyroid hormones. Heart Fail Rev 21, 391–399 (2016). https://doi.org/10.1007/s10741-016-9545-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-016-9545-8