Abstract
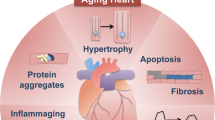
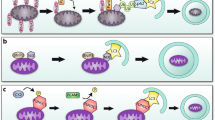
Heart failure is a progressive disease, leading to reduced quality of life and premature death. Adverse ventricular remodeling involves changes in the balance between cardiomyocyte protein synthesis and degradation, forcing these myocytes in equilibrium between life and death. In this context, autophagy has been recognized to play a role in the pathophysiology of heart failure. At basal levels, autophagy performs housekeeping functions, maintaining cardiomyocyte function and ventricular mass. Autophagy also occurs in the failing human heart, and upregulation has been reported in animal models of pressure overload–induced heart failure. Although the factors that determine whether autophagy will be protective or detrimental are not well known, the level and duration of autophagy seem important. Autophagy may antagonize ventricular hypertrophy by increasing protein degradation, which decreases tissue mass. However, the rate of protective autophagy declines with age. The inability to remove damaged structures results in the progressive accumulation of ‘garbage’, including abnormal intracellular proteins aggregates and undigested materials such as lipofuscin. Eventually, the progress of these changes results in enhanced oxidative stress, decreased ATP production, collapse of the cellular catabolic machinery, and cell death. By contrast, in load-induced heart failure, the extent of autophagic flux can rise to maladaptive levels. Excessive autophagy induction leads to autophagic cell death and loss of cardiomyocytes and may contribute to the worsening of heart failure. Accordingly, the development of therapies that up-regulate the repair qualities of the autophagic process and down-regulate the cell death aspects would be of great value in the treatment of heart failure.



Similar content being viewed by others
References
Heart disease & stroke. Statistics. http://www.americanheart.org/downloadable/heart/1240250946756LS-1982+Heart+and+Stroke+Update.042009.pdf. 2009. American Heart Association. Accessed 1 Oct 2009
De Keulenaer GW, Brutsaert DL (2009) The heart failure spectrum: time for a phenotype-oriented approach. Circulation 119:3044–3046
van Heerebeek L, Borbely A, Niessen HW, Bronzwaer JG, van der Velden J, Stienen GJ, Linke WA, Laarman GJ, Paulus WJ (2006) Myocardial structure and function differ in systolic and diastolic heart failure. Circulation 113:1966–1973
Martinet W, Knaapen MW, Kockx MM, De Meyer GRY (2007) Autophagy in cardiovascular disease. Trends Mol Med 13:482–491
Rothermel BA, Hill JA (2008) Autophagy in load-induced heart disease. Circ Res 103:1363–1369
De Meyer GRY, Martinet W (2009) Autophagy in the cardiovascular system. Biochim Biophys Acta 1793:1485–1495
Martinet W, Agostinis P, Vanhoecke B, Dewaele M, De Meyer GRY (2009) Autophagy in disease: a double-edged sword with therapeutic potential. Clin Sci (Lond) 116:697–712
Cuervo AM, Bergamini E, Brunk UT, Droge W, Ffrench M, Terman A (2005) Autophagy and aging: the importance of maintaining “clean” cells. Autophagy 1:131–140
Brunk UT, Jones CB, Sohal RS (1992) A novel hypothesis of lipofuscinogenesis and cellular aging based on interactions between oxidative stress and autophagocytosis. Mutat Res 275:395–403
Terman A, Brunk UT (2005) Autophagy in cardiac myocyte homeostasis, aging, and pathology. Cardiovasc Res 68:355–365
Terman A, Kurz T, Navratil M, Arriaga EA, Brunk UT (2010) Mitochondrial turnover and aging of long-lived postmitotic cells. Antioxid Redox Signal 12:503–535
Kurz T, Terman A, Brunk UT (2007) Autophagy, ageing and apoptosis: the role of oxidative stress and lysosomal iron. Arch Biochem Biophys 462:220–230
Rajawat YS, Hilioti Z, Bossis I (2009) Aging: central role for autophagy and the lysosomal degradative system. Ageing Res Rev 8:199–213
Gottlieb RA, Finley KD, Mentzer RM Jr (2009) Cardioprotection requires taking out the trash. Basic Res Cardiol 104:169–180
Gustafsson AB, Gottlieb RA (2009) Autophagy in ischemic heart disease. Circ Res 104:150–158
Nishida K, Kyoi S, Yamaguchi O, Sadoshima J, Otsu K (2008) The role of autophagy in the heart. Cell Death Differ 16:31–38
Kanamori H, Takemura G, Maruyama R, Goto K, Tsujimoto A, Ogino A, Li L, Kawamura I, Takeyama T, Kawaguchi T, Nagashima K, Fujiwara T, Fujiwara H, Seishima M, Minatoguchi S (2009) Functional significance and morphological characterization of starvation-induced autophagy in the adult heart. Am J Pathol 174:1705–1714
Baehrecke EH (2005) Autophagy: dual roles in life and death? Nat Rev Mol Cell Biol 6:505–510
Rothermel BA, Hill JA (2007) Myocyte autophagy in heart disease: friend or foe? Autophagy 3:632–634
Kostin S, Pool L, Elsasser A, Hein S, Drexler HC, Arnon E, Hayakawa Y, Zimmermann R, Bauer E, Klovekorn WP, Schaper J (2003) Myocytes die by multiple mechanisms in failing human hearts. Circ Res 92:715–724
Hein S, Arnon E, Kostin S, Schonburg M, Elsasser A, Polyakova V, Bauer EP, Klovekorn WP, Schaper J (2003) Progression from compensated hypertrophy to failure in the pressure-overloaded human heart: structural deterioration and compensatory mechanisms. Circulation 107:984–991
Nakai A, Yamaguchi O, Takeda T, Higuchi Y, Hikoso S, Taniike M, Omiya S, Mizote I, Matsumura Y, Asahi M, Nishida K, Hori M, Mizushima N, Otsu K (2007) The role of autophagy in cardiomyocytes in the basal state and in response to hemodynamic stress. Nat Med 13:619–624
Zhu H, Tannous P, Johnstone JL, Kong Y, Shelton JM, Richardson JA, Le V, Levine B, Rothermel BA, Hill JA (2007) Cardiac autophagy is a maladaptive response to hemodynamic stress. J Clin Invest 117:1782–1793
Gustafsson AB, Gottlieb RA (2008) Recycle or die: the role of autophagy in cardioprotection. J Mol Cell Cardiol 44:654–661
Kuzman JA, O’Connell TD, Gerdes AM (2007) Rapamycin prevents thyroid hormone-induced cardiac hypertrophy. Endocrinology 148:3477–3484
Ha T, Li Y, Gao X, McMullen JR, Shioi T, Izumo S, Kelley JL, Zhao A, Haddad GE, Williams DL, Browder IW, Kao RL, Li C (2005) Attenuation of cardiac hypertrophy by inhibiting both mTOR and NFkappaB activation in vivo. Free Radic Biol Med 39:1570–1580
McMullen JR, Sherwood MC, Tarnavski O, Zhang L, Dorfman AL, Shioi T, Izumo S (2004) Inhibition of mTOR signaling with rapamycin regresses established cardiac hypertrophy induced by pressure overload. Circulation 109:3050–3055
Rubinsztein DC (2006) The roles of intracellular protein-degradation pathways in neurodegeneration. Nature 443:780–786
Tannous P, Zhu H, Nemchenko A, Berry JM, Johnstone JL, Shelton JM, Miller FJ Jr, Rothermel BA, Hill JA (2008) Intracellular protein aggregation is a proximal trigger of cardiomyocyte autophagy. Circulation 117:3070–3078
Weekes J, Morrison K, Mullen A, Wait R, Barton P, Dunn MJ (2003) Hyperubiquitination of proteins in dilated cardiomyopathy. Proteomics 3:208–216
Fidzianska A, Bilinska ZT, Walczak E, Witkowski A, Chojnowska L (2010) Autophagy in transition from hypertrophic cardiomyopathy to heart failure. J Electron Microsc (Tokyo) in press doi:10.1093/jmicro/dfp048
Knaapen MW, Davies MJ, De BM, Haven AJ, Martinet W, Kockx MM (2001) Apoptotic versus autophagic cell death in heart failure. Cardiovasc Res 51:304–312
Elsasser A, Vogt AM, Nef H, Kostin S, Mollmann H, Skwara W, Bode C, Hamm C, Schaper J (2004) Human hibernating myocardium is jeopardized by apoptotic and autophagic cell death. J Am Coll Cardiol 43:2191–2199
Akazawa H, Komazaki S, Shimomura H, Terasaki F, Zou Y, Takano H, Nagai T, Komuro I (2004) Diphtheria toxin-induced autophagic cardiomyocyte death plays a pathogenic role in mouse model of heart failure. J Biol Chem 279:41095–41103
Shimomura H, Terasaki F, Hayashi T, Kitaura Y, Isomura T, Suma H (2001) Autophagic degeneration as a possible mechanism of myocardial cell death in dilated cardiomyopathy. Jpn Circ J 65:965–968
Takemura G, Miyata S, Kawase Y, Okada H, Maruyama R, Fujiwara H (2006) Autophagic degeneration and death of cardiomyocytes in heart failure. Autophagy 2:212–214
Dhesi P, Tehrani F, Fuess J, Schwarz ER (2010) How does the heart (not) die? The role of autophagy in cardiomyocyte homeostasis and cell death. Heart Fail Rev 15:15–21
Martinet W, De Meyer GRY (2009) Autophagy in atherosclerosis: a cell survival and death phenomenon with therapeutic potential. Circ Res 104:304–317
Salminen A, Kaarniranta K (2009) SIRT1: regulation of longevity via autophagy. Cell Signal 21:1356–1360
Hsu CP, Odewale I, Alcendor RR, Sadoshima J (2008) Sirt1 protects the heart from aging and stress. Biol Chem 389:221–231
Opie LH, Lecour S (2007) The red wine hypothesis: from concepts to protective signalling molecules. Eur Heart J 28:1683–1693
Hsu CP, Oka S, Shao D, Hariharan N, Sadoshima J (2009) Nicotinamide phosphoribosyltransferase regulates cell survival through NAD + synthesis in cardiac myocytes. Circ Res 105:481–491
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy fights disease through cellular self-digestion. Nature 451:1069–1075
Shintani T, Klionsky DJ (2004) Autophagy in health and disease: a double-edged sword. Science 306:990–995
Pattingre S, Tassa A, Qu X, Garuti R, Liang XH, Mizushima N, Packer M, Schneider MD, Levine B (2005) Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122:927–939
Nishida K, Kyoi S, Yamaguchi O, Sadoshima J, Otsu K (2009) The role of autophagy in the heart. Autophagy 16:31–38
Razeghi P, Myers TJ, Frazier OH, Taegtmeyer H (2002) Reverse remodeling of the failing human heart with mechanical unloading. Emerging concepts and unanswered questions. Cardiology 98:167–174
Razeghi P, Sharma S, Ying J, Li YP, Stepkowski S, Reid MB, Taegtmeyer H (2003) Atrophic remodeling of the heart in vivo simultaneously activates pathways of protein synthesis and degradation. Circulation 108:2536–2541
Kassiotis C, Ballal K, Wellnitz K, Vela D, Gong M, Salazar R, Frazier OH, Taegtmeyer H (2009) Markers of autophagy are downregulated in failing human heart after mechanical unloading. Circulation 120:S191–S197
de Gasparo M, Catt KJ, Inagami T, Wright JW, Unger T (2000) International union of pharmacology XXIII. The angiotensin II receptors. Pharmacol Rev 52:415–472
Timmermans PB, Wong PC, Chiu AT, Herblin WF, Benfield P, Carini DJ, Lee RJ, Wexler RR, Saye JA, Smith RD (1993) Angiotensin II receptors and angiotensin II receptor antagonists. Pharmacol Rev 45:205–251
Griendling KK, Lassegue B, Alexander RW (1996) Angiotensin receptors and their therapeutic implications. Annu Rev Pharmacol Toxicol 36:281–306
Unger T, Li J (2004) The role of the renin-angiotensin-aldosterone system in heart failure. J Renin Angiotensin Aldosterone Syst 5(Suppl 1):S7–S10
Porrello ER, D’Amore A, Curl CL, Allen AM, Harrap SB, Thomas WG, Delbridge LM (2009) Angiotensin II type 2 receptor antagonizes angiotensin II type 1 receptor-mediated cardiomyocyte autophagy. Hypertension 53:1032–1040
Steckelings UM, Unger T (2009) Angiotensin receptors and autophagy: live and let die. Hypertension 53:898–899
Acknowledgments
The authors thank Liliane Van Den Eynde for secretarial assistance. The authors’ research that is cited in this review was supported by the Fund for Scientific Research (FWO)-Flanders (Belgium) (Projects G.0308.04, G.0113.06 and G.0112.08), the University of Antwerp (NOI-BOF and TOP-BOF), and the Bekales Foundation. W. Martinet is a postdoctoral fellow of the FWO-Flanders.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Meyer, G.R.Y., De Keulenaer, G.W. & Martinet, W. Role of autophagy in heart failure associated with aging. Heart Fail Rev 15, 423–430 (2010). https://doi.org/10.1007/s10741-010-9166-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-010-9166-6