Abstract
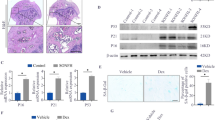
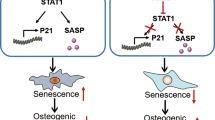
Age-related jawbone loss directly impact the function of oral cavity resulted from tooth loss, implant failure, and jaw fracture. Numerous evidences show that age-related senescence of bone marrow stromal cells (BMSCs) play a critical role in bone loss, but little attention has been paid to jawbone. Here, we delineated the critical role of sirtuin family protein 6 (SIRT6) in senescence, autophagy, and osteogenesis of BMSCs from jawbones. Radiography analysis showed less jawbone quality in elderly than young people. We also showed that SIRT6 expression decreased in bone tissue and BMSCs from the elderly by immunochemical staining. BMSCs from the elderly exhibited decreased osteogenic differentiation and inclined senescence which these phenotypes could be simulated by SIRT6 knockdown. Furthermore, accompanied with the inhibition of SIRT6, the autophagy level and ostogenesis of BMSCs was also decreased. However, using rapamycin, an autophagy activator, could rescue these adverse effects of BMSCs caused by SIRT6 inhibition. Mechanistically, SIRT6 regulated the autophagy and osteogenesis of BMSCs by activating AKT-mTOR pathway, at least in part. Finally, a decreased jawbone quality was shown in SIRT6 haploinsufficiency mice by Wnt1 specific tissue knockdown (Wnt1-Cre;SIRT6fl/+) model. Taken together, our data revealed that SIRT6 adjusted senescence and osteogenesis of BMSCs via altering autophagy level, and associated with age-related bone loss. SIRT6 could be as a promising therapeutic target for age-related osteoporosis of jawbone.





Similar content being viewed by others
References
Bianco P, Robey PG, Simmons PJ (2008) Mesenchymal stem cells: revisiting history, concepts, and assays. Cell Stem Cell 2:313–319. https://doi.org/10.1016/j.stem.2008.03.002
Bodic F, Hamel L, Lerouxel E, Baslé MF, Chappard D (2005) Bone loss and teeth. Joint Bone Spine 72:215–221. https://doi.org/10.1016/j.jbspin.2004.03.007
Chai Y et al (2000) Fate of the mammalian cranial neural crest during tooth and mandibular morphogenesis. Development 127:1671–1679
Cuervo AM, Bergamini E, Brunk UT, Droge W, Ffrench M, Terman A (2005) Autophagy and aging: the importance of maintaining "clean" cells. Autophagy 1:131–140
Fan Y et al (2019) Sirt6 suppresses high glucose-induced mitochondrial dysfunction and apoptosis in podocytes through AMPK activation. Int J Biol Sci 15:701–713. https://doi.org/10.7150/ijbs.29323
Fu Y et al (2014) Histone deacetylase 8 suppresses osteogenic differentiation of bone marrow stromal cells by inhibiting histone H3K9 acetylation and RUNX2 activity. Int J Biochem Cell Biol 54:68–77. https://doi.org/10.1016/j.biocel.2014.07.003
García-Prat L et al (2016) Autophagy maintains stemness by preventing senescence. Nature 529:37–42. https://doi.org/10.1038/nature16187
Gertler AA, Cohen HY (2013) SIRT6, a protein with many faces. Biogerontology 14:629–639. https://doi.org/10.1007/s10522-013-9478-8
He C, Klionsky DJ (2009) Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet 43:67–93. https://doi.org/10.1146/annurev-genet-102808-114910
He J et al (2017) SIRT6 reduces macrophage foam cell formation by inducing autophagy and cholesterol efflux under ox-LDL condition. Febs J 284:1324–1337. https://doi.org/10.1111/febs.14055
Heras-Sandoval D, Perez-Rojas JM, Hernandez-Damian J, Pedraza-Chaverri J (2014) The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal 26:2694–2701. https://doi.org/10.1016/j.cellsig.2014.08.019
Hocking LJ, Whitehouse C, Helfrich MH (2012) Autophagy: a new player in skeletal maintenance? J Bone Miner Res 27:1439–1447. https://doi.org/10.1002/jbmr.1668
Huang N et al (2017) Sirtuin 6 plays an oncogenic role and induces cell autophagy in esophageal cancer cells. Tumour Biol 39:1010428317708532. https://doi.org/10.1177/1010428317708532
Koh K, Kim K (2011) Utility of the computed tomography indices on cone beam computed tomography images in the diagnosis of osteoporosis in women. Imaging Sci Dent 41:101. https://doi.org/10.5624/isd.2011.41.3.101
Lu J et al (2016) SIRT6 suppresses isoproterenol-induced cardiac hypertrophy through activation of autophagy. Transl Res 172:96–112.e6. https://doi.org/10.1016/j.trsl.2016.03.002
Ma Y et al (2018) Autophagy controls mesenchymal stem cell properties and senescence during bone aging. Aging Cell. https://doi.org/10.1111/acel.12709
McClung M, Baron R, Bouxsein M (2017) An update on osteoporosis pathogenesis, diagnosis, and treatment. Bone 98:37. https://doi.org/10.1016/j.bone.2017.02.013
Michishita E et al (2008) SIRT6 is a histone H3 lysine 9 deacetylase that modulates telomeric chromatin. Nature 452:492–496. https://doi.org/10.1038/nature06736
Mostoslavsky R et al (2006) Genomic instability and aging-like phenotype in the absence of mammalian SIRT6. Cell 124:315–329. https://doi.org/10.1016/j.cell.2005.11.044
Mu W et al (2018) Metformin promotes the proliferation and differentiation of murine preosteoblast by regulating the expression of sirt6 and oct4. Pharmacol Res 129:462–474. https://doi.org/10.1016/j.phrs.2017.11.020
Ono T, Takayanagi H (2017) Osteoimmunology in bone fracture healing. Curr Osteoporos Rep 15:367–375. https://doi.org/10.1007/s11914-017-0381-0
Qi M et al (2017) Autophagy maintains the function of bone marrow mesenchymal stem cells to prevent estrogen deficiency-induced osteoporosis. Theranostics 7:4498–4516. https://doi.org/10.7150/thno.17949
Revuelta M, Matheu A (2017) Autophagy in stem cell aging. Aging Cell 16:912–915. https://doi.org/10.1111/acel.12655
Schlundt C et al (2018) Macrophages in bone fracture healing: their essential role in endochondral ossification. Bone 106:78–89. https://doi.org/10.1016/j.bone.2015.10.019
Shih MS, Cook MA, Spence CA, Palnitkar S, McElroy H, Parfitt AM (1993) Relationship between bone formation rate and osteoblast surface on different subdivisions of the endosteal envelope in aging & osteoporosis. Bone 14:519–521
Smith M, Wilkinson S (2017) ER homeostasis and autophagy. Essays Biochem 61:625–635. https://doi.org/10.1042/EBC20170092
Song C, Song C, Tong F (2014) Autophagy induction is a survival response against oxidative stress in bone marrow-derived mesenchymal stromal cells. Cytotherapy 16:1361–1370. https://doi.org/10.1016/j.jcyt.2014.04.006
Sugatani T, Agapova O, Malluche HH, Hruska KA (2015) SIRT6 deficiency culminates in low-turnover osteopenia. Bone 81:168–177. https://doi.org/10.1016/j.bone.2015.07.018
Sun H, Wu Y, Fu D, Liu Y, Huang C (2014) SIRT6 regulates osteogenic differentiation of rat bone marrow mesenchymal stem cells partially via suppressing the nuclear factor-kappaB signaling pathway. Stem Cells 32:1943–1955. https://doi.org/10.1002/stem.1671
Wang L et al (2018) Aberrant SIRT6 expression contributes to melanoma growth: role of the autophagy paradox and IGF-AKT signaling. Autophagy 14:518–533. https://doi.org/10.1080/15548627.2017.1384886
Xiao C et al (2012) Progression of chronic liver inflammation and fibrosis driven by activation of c-JUN signaling in Sirt6 mutant mice. J Biol Chem 287:41903–41913. https://doi.org/10.1074/jbc.M112.415182
Xu R et al (2016) Transplantation of osteoporotic bone marrow stromal cells rejuvenated by the overexpression of SATB2 prevents alveolar bone loss in ovariectomized rats. Exp Gerontol 84:71–79. https://doi.org/10.1016/j.exger.2016.09.001
Xu R et al (2018) Simvastatin improves oral implant osseointegration via enhanced autophagy and osteogenesis of BMSCs and inhibited osteoclast activity. J Tissue Eng Regen Med 12:1209–1219. https://doi.org/10.1002/term.2652
Xue JF, Shi ZM, Zou J, Li XL (2017) Inhibition of PI3K/AKT/mTOR signaling pathway promotes autophagy of articular chondrocytes and attenuates inflammatory response in rats with osteoarthritis. Biomed Pharmacother 89:1252–1261. https://doi.org/10.1016/j.biopha.2017.01.130
Yang B, Zwaans BM, Eckersdorff M, Lombard DB (2009) The sirtuin SIRT6 deacetylates H3 K56Ac in vivo to promote genomic stability. Cell Cycle 8:2662–2663. https://doi.org/10.4161/cc.8.16.9329
Yu Y et al (2012) Insulin-like growth factor 1 enhances the proliferation and osteogenic differentiation of human periodontal ligament stem cells via ERK and JNK MAPK pathways. Histochem Cell Biol 137:513–525. https://doi.org/10.1007/s00418-011-0908-x
Zhang P et al (2017) SIRT6 promotes osteogenic differentiation of mesenchymal stem cells through BMP signaling. Sci Rep. https://doi.org/10.1038/s41598-017-10323-z
Zhang W et al (2018) SIRT6 deficiency results in developmental retardation in cynomolgus monkeys. Nature 560:661–665. https://doi.org/10.1038/s41586-018-0437-z
Zhou, P. et al. (2016) SATB2-Nanog axis links age-related intrinsic changes of mesenchymal stem cells from craniofacial bone. Aging 8:2006–2011. https://doi.org/10.18632/aging.101041
Zhu W et al (2019) Zoledronic acid promotes TLR-4-mediated M1 macrophage polarization in bisphosphonate-related osteonecrosis of the jaw. Faseb J. https://doi.org/10.1096/fj.201801791RR
Acknowledgements
This work was supported by the National Natural Science Fundation of China (81970910) and a project funded by the Priority Academic Program for the Development of Jiangsu Higher Education Institutions (2018-87).
Author information
Authors and Affiliations
Contributions
XS performed experiments, analyzed data, and wrote the manuscript. XC, JDH prepared the figures and analyzed data. RYX and JC contributed to the reagents and materials. HBJ designed the experimental study and analyzed the data. All authors participated in discussing and revising the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shen, X., Chen, X., Huang, J. et al. Age-dependent role of SIRT6 in jawbone via regulating senescence and autophagy of bone marrow stromal cells. J Mol Hist 51, 67–76 (2020). https://doi.org/10.1007/s10735-020-09857-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-020-09857-w