Abstract
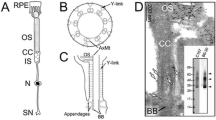
Numerous biochemical and morphological studies have provided insight into the distribution pattern of caveolin-1 and the presence of membrane rafts in the vertebrate retina. To date however, studies have not addressed the localization profile of raft specific proteins during development. Therefore the purpose of our studies was to follow the localization pattern of caveolin-1, phospho-caveolin-1 and c-src in the developing retina and compare it to that observed in adults. Specific antibodies were used to visualize the distribution of caveolin-1, c-src, a kinase phosphorylating caveolin-1, and phospho-caveolin-1. The labeling pattern of this scaffolded complex was compared to those of rhodopsin and rhodopsin kinase. Samples were analyzed at various time points during postnatal development and compared to adult retinas. The immunocytochemical studies were complemented with immunoblots and immunoprecipitation studies. In the mature retina caveolin-1 and c-src localized mainly to the cell body and IS of photoreceptors, with only very weakly labeled OS. In contrast, phospho-caveolin-1 was only detectable in the OS of photoreceptors. During development we followed the expression and distribution profile of these proteins in a temporal sequence with special attention to the period when OS formation is most robust. Double labeling immunocytochemistry and immunoprecipitation showed rhodopsin to colocalize and co-immunoprecipitate with caveolin-1 and c-src. Individual punctate structures between the outer limiting membrane and the outer plexiform layer were seen at P10 to be labeled by both rhodopsin and caveolin-1 as well as by rhodopsin and c-src, respectively. These studies suggest that membrane raft specific proteins are co-distributed during development, thereby pointing to a role for such complexes in OS formation. In addition, the presence of small punctate structures containing caveolin-1, c-src and rhodopsin raise the possibility that these proteins may transport together to OS during development and that caveolin-1 exists predominantly in a phosphorylated form in the OS.







Similar content being viewed by others
References
Baker SA, Haeri M, Yoo P, Gospe SM III, Skiba NP, Knox BE, Arshavsky VY (2008) The outer segment serves as a default destination for the trafficking of membrane proteins in photoreceptors. J Cell Biol 183:485–498
Berta AI, Kiss AL, Kemeny-Beke A, Lukats A, Szabo A, Szel A (2007a) Different caveolin isoforms in the retina of melanoma malignum affected human eye. Mol Vis 13:881–886
Berta AI, Kiss AL, Lukats A, Szabo A, Szel A (2007b) Distribution of caveolin isoforms in the lemur retina. J Vet Sci 8:295–297
Boesze-Battaglia K, Dispoto J, Kahoe MA (2002) Association of a photoreceptor-specific tetraspanin protein, ROM-1, with triton X-100-resistant membrane rafts from rod outer segment disk membranes. J Biol Chem 277:41843–41849
Calderone JB, Jacobs GH (1999) Cone receptor variations and their functional consequences in two species of hamster. Vis Neurosci 16:53–63
del Pozo MA, Balasubramanian N, Alderson NB, Kiosses WB, Grande-Garcia A, Anderson RG, Schwartz MA (2005) Phospho-caveolin-1 mediates integrin-regulated membrane domain internalization. Nat Cell Biol 7:901–908
Deretic D (2006) A role for rhodopsin in a signal transduction cascade that regulates membrane trafficking and photoreceptor polarity. Vision Res 46:4427–4433
Elliott MH, Ghalayini AJ (2008) Phosphorylation of caveolin-1 in bovine rod outer segments in vitro by an endogenous tyrosine kinase. Adv Exp Med Biol 613:335–341
Elliott MH, Fliesler SJ, Ghalayini AJ (2003) Cholesterol-dependent association of caveolin-1 with the transducin alpha subunit in bovine photoreceptor rod outer segments: disruption by cyclodextrin and guanosine 5′-O-(3-thiotriphosphate). Biochemistry 42:7892–7903
Elliott MH, Nash ZA, Takemori N, Fliesler SJ, McClellan ME, Naash MI (2008) Differential distribution of proteins and lipids in detergent-resistant and detergent-soluble domains in rod outer segment plasma membranes and disks. J Neurochem 104:336–352
Greiner JV, Weidman TA (1978) Development of the hamster retina: a morphologic study. Am J Vet Res 39:665–670
Kachi S, Yamazaki A, Usukura J (2001) Localization of caveolin-1 in photoreceptor synaptic ribbons. Invest Ophthalmol Vis Sci 42:850–852
Khorana HG (1992) Rhodopsin, photoreceptor of the rod cell. An emerging pattern for structure and function. J Biol Chem 267:1–4
Kim H, Lee T, Lee J, Ahn M, Moon C, Wie MB, Shin T (2006) Immunohistochemical study of caveolin-1 and -2 in the rat retina. J Vet Sci 7:101–104
Kurzchalia TV, Parton RG (1999) Membrane microdomains and caveolae. Curr Opin Cell Biol 11:424–431
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li S, Seitz R, Lisanti MP (1996) Phosphorylation of caveolin by src tyrosine kinases. The alpha-isoform of caveolin is selectively phosphorylated by v-Src in vivo. J Biol Chem 271:3863–3868
Liu H, Seno K, Hayashi F (2003) Active transducin alpha subunit carries PDE6 to detergent-resistant membranes in rod photoreceptor outer segments. Biochem Biophys Res Commun 303:19–23
Maeda T, Imanishi Y, Palczewski K (2003) Rhodopsin phosphorylation: 30 years later. Prog Retin Eye Res 22:417–434
Martin RE, Elliott MH, Brush RS, Anderson RE (2005) Detailed characterization of the lipid composition of detergent-resistant membranes from photoreceptor rod outer segment membranes. Invest Ophthalmol Vis Sci 46:1147–1154
Nair KS, Balasubramanian N, Slepak VZ (2002) Signal-dependent translocation of transducin, RGS9–1-Gbeta5L complex, and arrestin to detergent-resistant membrane rafts in photoreceptors. Curr Biol 12:421–425
Nair KS, Hanson SM, Kennedy MJ, Hurley JB, Gurevich VV, Slepak VZ (2004) Direct binding of visual arrestin to microtubules determines the differential subcellular localization of its splice variants in rod photoreceptors. J Biol Chem 279:41240–41248
Pike LJ (2003) Lipid rafts: bringing order to chaos. J Lipid Res 44:655–667
Rohlich P, Szel A (1993) Binding sites of photoreceptor-specific antibodies COS-1, OS-2 and AO. Curr Eye Res 12:935–944
Schlegel A, Volonte D, Engelman JA, Galbiati F, Mehta P, Zhang XL, Scherer PE, Lisanti MP (1998) Crowded little caves: structure and function of caveolae. Cell Signal 10:457–463
Senin II, Hoppner-Heitmann D, Polkovnikova OO, Churumova VA, Tikhomirova NK, Philippov PP, Koch KW (2004) Recoverin and rhodopsin kinase activity in detergent-resistant membrane rafts from rod outer segments. J Biol Chem 279:48647–48653
Seno K, Kishimoto M, Abe M, Higuchi Y, Mieda M, Owada Y, Yoshiyama W, Liu H, Hayashi F (2001) Light- and guanosine 5’-3-O-(thio)triphosphate-sensitive localization of a G protein and its effector on detergent-resistant membrane rafts in rod photoreceptor outer segments. J Biol Chem 276:20813–20816
Swaney JS, Patel HH, Yokoyama U, Head BP, Roth DM, Insel PA (2006) Focal adhesions in (myo)fibroblasts scaffold adenylyl cyclase with phosphorylated caveolin. J Biol Chem 281:17173–17179
von Schantz M, Argamaso-Hernan SM, Szel A, Foster RG (1997) Photopigments and photoentrainment in the Syrian golden hamster. Brain Res 770:131–138
Acknowledgments
We would like to thank Dr. Pál Röhlich for critically reading the manuscript and Dr. Gustavo D. Aguirre for his beneficial advices about the manuscript. We are grateful to Dr. Krzysztof Palczewski for the donation of the antibody against rhodopsin kinase, to Dr. Robert Molday for the donation of the RDS antibody, to Nándor Müllner for the immunoprecipitation and also to Katalin Lőcsey and Margit Kutasi for their help in the sample preparation for electron microscopy. The work was supported by Hungarian OTKA Grant, #73000 (Ágoston Szél), EY10420 and EY18705 (Kathleen Boesze-Battaglia).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berta, Á.I., Boesze-Battaglia, K., Magyar, A. et al. Localization of caveolin-1 and c-src in mature and differentiating photoreceptors: raft proteins co-distribute with rhodopsin during development. J Mol Hist 42, 523–533 (2011). https://doi.org/10.1007/s10735-011-9360-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-011-9360-4