Abstract
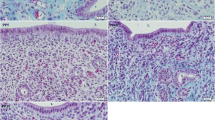
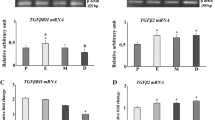
Activin A is a growth factor expressed in the endometrium, where it modulates tissue remodeling and enhances decidualization. The effects of activin A are counteracted by two binding proteins, namely follistatin and follistatin-like 3 (FSTL3). We have evaluated the effects of estrogen and progestin on the endometrial expression of activin βA subunit, follistatin and FSTL3 in ovariectomized rats. Adult female Wistar rats (n = 21) were ovariectomized and received one week later a single dose of estradiol benzoate (1.5 mg/kg body weight, i.m. injection), either alone (n = 7) or associated with depot medroxyprogesterone acetate (3 mg/kg body weight, i.m. injection, n = 7), or oil vehicle (control group, n = 7). One week later, activin βA subunit mRNA levels had increased significantly in the uteri of rats treated with estradiol alone (7.4 fold increase over controls, P < 0.05) and to the same extent in rats receiving estradiol plus medroxyprogesterone (6.1 fold increase over controls, P < 0.05). This was accompanied by increase of βA subunit immunostaining in estradiol and estroprogestin treated rats, which was noted only in the surface endometrial epithelium. Follistatin mRNA expression, conversely, showed a significant decrease in the groups treated with estrogen alone and estrogen plus progestin (P < 0.05), and follistatin immunostaining in the glandular epithelium was weaker in estradiol and estroprogestin-treated rats compared to controls. FSTL3 expression was similar in the 3 groups. In conclusion, the expression of activin βA subunit increases and that of follistatin decreases following estrogen replacement in the endometrium of ovariectomized rats, and these effects are not further altered by the addition of progestin.


Similar content being viewed by others
References
Ciarmela P, Florio P, Sigurdardottir M, Toti P, Maguer-Satta V, Rimokh R et al (2004) Follistatin-related gene expression, but not follistatin expression, is decreased in human endometrial adenocarcinoma. Eur J Endocrinol 151:251–257. doi:10.1530/eje.0.1510251
de Winter JP, ten Dijke P, de Vries CJ, van Achterberg TA, Sugino H, de Waele P et al (1996) Follistatins neutralize activin bioactivity by inhibition of activin binding to its type ii receptors. Mol Cell Endocrinol 116:105–114. doi:10.1016/0303-7207(95)03705-5
Florio P, Severi FM, Luisi S, Ciarmela P, Calonaci G, Cobellis L et al (2003) Endometrial expression and secretion of activin A, but not follistatin, increase in the secretory phase of the menstrual cycle. J Soc Gynecol Investig 10:237–243. doi:10.1016/S1071-5576(03)00045-5
Florio P, Ciarmela P, Toti P, Maguer-Satta V, Rimokh R, Buonocore G et al (2004) Human endometrium and decidua express follistatin-related gene (FLRG) mRNA and peptide. Mol Cell Endocrinol 218:129–135. doi:10.1016/j.mce.2003.12.019
Florio P, Rossi M, Vigano P, Luisi S, Torricelli M, Torres PB et al (2007) Interleukin 1beta and progesterone stimulate activin A expression and secretion from cultured human endometrial stromal cells. Reprod Sci 14:29–36. doi:10.1177/1933719106298191
Gu Y, Srivastava RK, Ou J, Krett NL, Mayo KE, Gibori G (1995) Cell-specific expression of activin and its two binding proteins in the rat decidua: role of alpha 2-macroglobulin and follistatin. Endocrinology 136:3815–3822. doi:10.1210/en.136.9.3815
Jones RL, Salamonsen LA, Critchley HO, Rogers PA, Affandi B, Findlay JK (2000) Inhibin and activin subunits are differentially expressed in endometrial cells and leukocytes during the menstrual cycle, in early pregnancy and in women using progestin-only contraception. Mol Hum Reprod 6:1107–1117. doi:10.1093/molehr/6.12.1107
Jones RL, Salamonsen LA, Findlay JK (2002) Activin A promotes human endometrial stromal cell decidualization in vitro. J Clin Endocrinol Metab 87:4001–4004. doi:10.1210/jc.87.8.4001
Jones RL, Findlay JK, Farnworth PG, Robertson DM, Wallace E, Salamonsen LA (2006a) Activin A and inhibin A differentially regulate human uterine matrix metalloproteinases: potential interactions during decidualization and trophoblast invasion. Endocrinology 147:724–732. doi:10.1210/en.2005-1183
Jones RL, Findlay JK, Salamonsen LA (2006b) The role of activins during decidualisation of human endometrium. Aust NZJ Obstet Gynaecol 46:245–249. doi:10.1111/j.1479-828X.2006.00581.x
Leung PH, Salamonsen LA, Findlay JK (1998) Immunolocalization of inhibin and activin subunits in human endometrium across the menstrual cycle. Hum Reprod 13:3469–3477. doi:10.1093/humrep/13.12.3469
Ling N, Ying SY, Ueno N, Shimasaki S, Esch F, Hotta M et al (1986) Pituitary FSH is released by a heterodimer of the beta-subunits from the two forms of inhibin. Nature 321:779–782. doi:10.1038/321779a0
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative pcr and the 2(-delta delta c(t)) method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Mathews LS, Vale WW (1991) Expression cloning of an activin receptor, a predicted transmembrane serine kinase. Cell 65:973–982. doi:10.1016/0092-8674(91)90549-E
Mathews LS, Vale WW (1993) Characterization of type II activin receptors. Binding, processing, and phosphorylation. J Biol Chem 268:19013–19018
Mathews LS, Vale WW, Kintner CR (1992) Cloning of a second type of activin receptor and functional characterization in xenopus embryos. Science 255:1702–1705. doi:10.1126/science.1313188
Mylonas I, Jeschke U, Wiest I, Hoeing A, Vogl J, Shabani N et al (2004) Inhibin/activin subunits alpha, beta-A and beta-B are differentially expressed in normal human endometrium throughout the menstrual cycle. Histochem Cell Biol 122:461–471. doi:10.1007/s00418-004-0709-6
Ocal G, Kokcu A, Cetinkaya MB, Tosun M, Kefeli M, Kandemir B (2007) Efficacy of levamisole on experimental endometriosis. Int J Gynaecol Obstet 99:38–42. doi:10.1016/j.ijgo.2007.04.041
Otani T, Minami S, Kokawa K, Shikone T, Yamoto M, Nakano R (1998) Immunohistochemical localization of activin A in human endometrial tissues during the menstrual cycle and in early pregnancy. Obstet Gynecol 91:685–692. doi:10.1016/S0029-7844(98)00053-2
Popovici RM, Kao LC, Giudice LC (2000) Discovery of new inducible genes in in vitro decidualized human endometrial stromal cells using microarray technology. Endocrinology 141:3510–3513. doi:10.1210/en.141.9.3510
Sahlin L, Masironi B, Akerberg S, Eriksson H (2006) Tissue- and hormone-dependent progesterone receptor distribution in the rat uterus. Reprod Biol Endocrinol 4:47. doi:10.1186/1477-7827-4-47
Schneyer A, Tortoriello D, Sidis Y, Keutmann H, Matsuzaki T, Holmes W (2001) Follistatin-related protein (FSRP): a new member of the follistatin gene family. Mol Cell Endocrinol 180:33–38. doi:10.1016/S0303-7207(01)00501-9
Schneyer A, Sidis Y, Xia Y, Saito S, del Re E, Lin HY et al (2004) Differential actions of follistatin and follistatin-like 3. Mol Cell Endocrinol 225:25–28. doi:10.1016/j.mce.2004.02.009
Spritzer PM, Ribeiro MF, Oliveira MC, Barbosa-Coutinho LM, Silva IS, Dahlem N et al (2001) Effects of tamoxifen on serum prolactin levels, pituitary immunoreactive prolactin cells and uterine growth in estradiol-treated ovariectomized rats. Horm Metab Res 28:171–176
Tsuchida K, Mathews LS, Vale WW (1993) Cloning and characterization of a transmembrane serine kinase that acts as an activin type I receptor. Proc Natl Acad Sci USA 90:11242–11246. doi:10.1073/pnas.90.23.11242
Vale W, Rivier J, Vaughan J, McClintock R, Corrigan A, Woo W et al (1986) Purification and characterization of an FSH releasing protein from porcine ovarian follicular fluid. Nature 321:776–779. doi:10.1038/321776a0
Wang HQ, Takebayashi K, Tsuchida K, Nishimura M, Noda Y (2003) Follistatin-related gene (FLRG) expression in human endometrium: sex steroid hormones regulate the expression of FLRG in cultured human endometrial stromal cells. J Clin Endocrinol Metab 88:4432–4439. doi:10.1210/jc.2002-021758
Wool IG, Chan YL, Gluck A, Suzuki K (1991) The primary structure of rat ribosomal proteins p0, p1, and p2 and a proposal for a uniform nomenclature for mammalian and yeast ribosomal proteins. Biochimie 73:861–870. doi:10.1016/0300-9084(91)90127-M
Wrana JL, Attisano L (2000) The Smad pathway. Cytokine Growth Factor Rev 11:5–13. doi:10.1016/S1359-6101(99)00024-6
Acknowledgments
Research supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil, and the Ministry of University and Scientific Research (MURST), Italy. We thank Ms. Solange Aquino and Mr. André Pimenta for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferreira, M.C., Cavallo, I.K.D., Florio, P. et al. Activin βA subunit, follistatin and follistatin-like 3 are expressed in the endometrium of ovariectomized rats and regulated by estrogen replacement. J Mol Hist 39, 535–541 (2008). https://doi.org/10.1007/s10735-008-9194-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-008-9194-x