Abstract
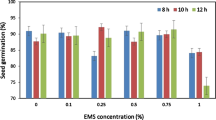
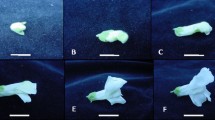
A tissue culture screen for ethylene tolerance utilizing 1-aminocyclopropane-1-carboxylic acid (ACC) was optimized for Antirrhinum majus L. (snapdragon) inbred line OAK564. The influence of various concentrations of ACC, including 0, 0.25, 0.50, 1.0, 2.5, 5.0, 10, 25, 50, 75, and 100 µM on seedling growth was investigated. It was determined that 5 µM ACC was optimum in elucidating biological activity. This screen was then used to evaluate responses of 48 snapdragon hybrid lines along with an inbred line to ethylene based on hypocotyl elongation inhibition. Moreover, 40,000 mutagenized snapdragon seeds from three M2 populations, derived from ethyl methanesulfonate (EMS) treatments of 0.50, 0.75, and 1.0%, were also assessed for ethylene insensitivity based on presence/absence of an ethylene ‘triple response’. From this ethylene screen, a total of 231 putative ethylene mutants were recovered, exhibiting eight distinct phenotypes corresponding to the ‘triple response’. Of these, 16 mutants were selected for further analysis that included at least one and up to three lines from each of the eight identified phenotypic groups. M2 plants were grown in the greenhouse, and selfed seeds were collected. These M3 seeds were then screened with 5 µM ACC to assess their tolerance to ethylene at the population level. Responses varied from complete ethylene tolerance to ethylene sensitivity among the different putative mutant lines.






Similar content being viewed by others
Change history
11 January 2023
A Correction to this paper has been published: https://doi.org/10.1007/s10725-022-00952-4
References
Alonso JM, Stepanova AN, Solano R, Wisman E, Ferrari S, Ausubel FM, Ecker JR (2003) Five components of the ethylene-response pathway identified in a screen for weak ethylene-insensitive mutants in Arabidopsis. Proc Natl Acad Sci USA 100:2992–2997
Amist N, Singh NB (2022) Ethylene and phytohormone crosstalk in plant defense against abiotic stress. In: Singh S, Husain T, Singh VP, Tripathi DK, Prasad SM, Dubey NK (eds) Ethylene in Plant Biology. Wiley, New york, pp 277–290
Booker MA, DeLong A (2015) Producing the ethylene signal: regulation and diversification of ethylene biosynthetic enzymes. Plant Physiol 169:42–50. https://doi.org/10.1104/pp.15.00672
Cerveny CB, Miller WB (2010) Residual effects of ethylene on tulip growth and flowering. HortSci 45:1164–1166
Chang C (2016) Q&A: how do plants respond to ethylene and what is its importance? BMC Biol 14:7. https://doi.org/10.1186/s12915-016-0230-0
Clark DG, Dervinis C, Barrett JE, Nell TA (2001) Using a seedling hypocotyl assay as a genetic screen for ethylene sensitivity of seedling geranium cultivars. HortTech 11:297–302
Costa LC, Finger FL (2016) Flower opening and vase life of gladiolus cultivars: the sensitivity to ethylene and the carbohydrate content. Ornam Hortic 22:147–153. https://doi.org/10.14295/oh.v22i2.901
Costa LC, Luz LM, Nascimento VL, Araujo FF, Santos MNS, França CFM, Silva TP, Fugate KK, Finger FL (2020) Selenium-ethylene interplay in postharvest life of cut flowers. Front Plant Sci 11:584698. https://doi.org/10.3389/fpls.2020.584698
De Paepe A, De Grauwe L, Bertrand S, Smalle J, Van Der Straeten D (2005) The Arabidopsis mutant eer2 has enhanced ethylene responses in the light. J Exp Biol 56:2409–2420
Dhaliwal AK, Mohan A, Sidhu G, Maqbool R, Gill KS (2015) An ethylmethane sulfonate mutant resource in pre-green revolution hexaploid wheat. PLoS ONE 10(12):1–15. https://doi.org/10.1371/journal.pone.0145227
Dolgikh VA, Pukhovaya EM, Zemlyanskaya EV (2019) Shaping ethylene response: the role of EIN3/EIL1 transcription factors. Front Plant Sci 10:1030. https://doi.org/10.3389/fpls.2019.01030
Espina MJ, Ahmed CMS, Bernardini A, Adeleke E, Yadegari Z, Arelli P, Pantalone V, Taheri A (2018) Development and phenotypic screening of an ethyl methanesulfonate mutant population in soybean. Front Plant Sci 9:394. https://doi.org/10.3389/fpls.2018.0039
Eun HD, Ali S, Jung H, Kim K, Kim W-C (2019) Profiling of ACC synthase gene (ACS11) expression in Arabidopsis induced by abiotic stresses. Appl Biol Chem 62:42. https://doi.org/10.1186/s13765-019-0450-4
Hassan FAS, Ali E (2014) Physiological response of gladiolus flowers to anti-ethylene treatments and their relation to senescence. Int J Adv Res 2(10):188–199. https://www.journalijar.com/uploads/72_IJAR-4190.pdf
Heffron LM, Korban SS (2022a) Mutagenic responses to ethyl methanesulfonate and phenotypic characterization of an M1 generation of snapdragon, Antirrhinum majus. Euphytica 218:76. https://doi.org/10.1007/s10681-022-03034-8
Heffron LM, Korban SS (2022b) Evaluation of ethylene mutant snapdragon lines for rooting, gravitropism, 1-MCP, ethylene, and vase-life responses. Scient Hortic 304:11274. https://doi.org/10.1016/j.scienta.2022.111274
Heffron LM, Korban SS (2022c) Molecular and genetic characterization of ethylene insensitive mutants in snapdragon. Plant Growth Regul 98:625–639. https://doi.org/10.1007/s10725-022-00895-w
Hoffman T, Schmidt JS, Zheng X, Bent AF (1999) Isolation of ethylene-insensitive soybean mutants that are altered in pathogen susceptibility and gene-for-gene disease resistance. Plant Physiol 119:935–949
Houben M, Van de Poel B (2019) 1-Aminocyclopropane-1-carboxylic acid oxidase (ACO): the enzyme that makes the plant hormone ethylene. Front Plant Sci 10:695. https://doi.org/10.3389/fpls.2019.00695
Hu Y, Vandenbussche F, Van Der Straeten D (2017) Regulation of seedling growth by ethylene and the ethylene–auxin crosstalk. Planta 245:467–489. https://doi.org/10.1007/s00425-017-2651-6
Iqbal N, Khan NA, Ferrante A, Trivellini A, Francini A, Khan MIR (2017) Ethylene role in plant growth, development and senescence: Interaction with other phytohormones. Front Plant Sci 8:475. https://doi.org/10.3389/fpls.2017.00475
Jander G, Baerson SR, Hudak JA, Gonzalez KA, Gruys KJ, Last RL (2003) Ethylmethane sulfonate saturation mutagenesis in Arabidopsis to determine frequency of herbicide resistance. Plant Physiol 131:139–164
Jasrotia S, Jasrotia R (2022) Role of ethylene in combating biotic stress. In: Singh S, Husain T, Singh VP, Tripathi DK, Prasad SM, Dubey NK (eds) Ethylene in Plant Biology. Wiley, New York, pp 388–397
Ke C, Guan W, Bu S, Li X, Deng Y, Wei Z, Wu W, Zheng Y (2019) Determination of absorption dose in chemical mutagenesis in plants. PLoS ONE 14(1):e0210596. https://doi.org/10.1371/journal.pone.0210596
Khan N, Bano A, Ali S, Ali Babar M (2020) Crosstalk amongst phytohormones from planta and PGPR under biotic and abiotic stresses. Plant Growth Regul 90:189–203. https://doi.org/10.1007/s10725-020-00571-x
Kushwah S, Jones AM, Laxmi A (2011) Cytokinin interplay with ethylene, auxin, and glucose signaling controls Arabidopsis seedling root directional growth. Plant Physiol 156:1851–1866. https://doi.org/10.1104/pp.111.175794
Lacey RF, Binder BM (2014) How plants sense ethylene gas—the ethylene receptors. J Inorg Biochem 133:58–62. https://doi.org/10.1016/j.jinorgbio.2014.01.006
Latado RR, Adames AH, Neto AT (2004) In vitro mutation of chrysanthemum (Dendranthema grandiflora Tzvelev) with ethylmethanesulphonate (EMS) in immature floral pedicels. Plant Cell Tiss Organ Cult 77:103–106
Lee JH, Lee SY (2002) Selection of stable mutants from cultured rice anthers treated with ethyl methane sulfonic acid. Plant Cell Tiss Organ Cult 71:165–171
Lethin J, Shakil SSM, Hassan S, Sirijovski N, Töpel M, Olsson O, Aronsson H (2020) Development and characterization of an EMS-mutagenized wheat population and identification of salt-tolerant wheat lines. BMC Plant Biol 20:18. https://doi.org/10.1186/s12870-019-2137-8
Locke JM, Bryce JH, Morris PC (2000) Contrasting effects of ethylene perception and biosynthesis inhibitors on germination and seedling growth of barley (Hordeum vulgare L.). J Exp Bot 51:1843–1849
Lu Y, Luo S, Li Q, Li N, Du W, Yu P, Wang X, Zhang W, Xuan S, Zhou X, Shen J, Zhao J, Wang Y, Chen X, Shen S (2021) Phenotypic characterization and differential gene expression analysis reveal that dwarf mutant dwf dwarfism is associated with gibberellin in eggplant. Horticulturae 7:114. https://doi.org/10.3390/horticulturae7050114
Ma Q, Dong CH (2021) Regulatory functions and molecular mechanisms of ethylene receptors and receptor-associated proteins in higher plants. Plant Growth Regul 93:39–52. https://doi.org/10.1007/s10725-020-00674-5
Ma N, Ma C, Liu Y, Shahid M, Wang C, Gao J (2018) Petal senescence: a hormone view. J Exp Bot 69:719–732. https://doi.org/10.1093/jxb/ery009
Merchante C, Stepanova AN (2017) The triple response assay and its use to characterize ethylene mutants in Arabidopsis. In: Binder B, Schaller GE (eds) Ethylene Signaling. Methods in Molecular Biology, vol 1573. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6854-1_13
Qu L-J, Qin G (2014) Generation and identification of Arabidopsis mutants. Methods Mol Biol 1062:225–239. https://doi.org/10.1007/978-1-62703-580-4_12
Rabiza-Świder J, Skutnik E, Jędrzejuk A, Rochala-Wojciechowska J (2020) Nanosilver and sucrose delay the senescence of cut snapdragon flowers. Postharv Biol Technol 165:111165. https://doi.org/10.1016/j.postharvbio.2020.111165
Schaller GE, Kieber JJ (2002) Ethylene. In: Somerville, C., Meyerowitz, E. (eds) The Arabidopsis Book 1. 2002: e0071
Shaharoona B, Arshad M, Khalid A (2007) Differential response of etiolated pea seedlings to inoculation with rhizoctonia capable of utilizing 1-aminocyclopropane-1-carboxylic acid or L-methionine. J Microbiol 45:15–20
Singh P, Singh AP, Sane AP (2019) Differential and reciprocal regulation of ethylene pathway genes regulates petal abscission in fragrant and non-fragrant roses. Plant Sci 80:330–339. https://doi.org/10.1016/j.plantsci.2018.12.013
Stepanova AN, Hoyt JM, Hamilton AA, Alonso JM (2005) A link between ethylene and auxin uncovered by the characterization of two root-specific ethylene-insensitive mutants in Arabidopsis. Plant Cell 17:2230–2242
Suzuki Y, Sano Y, Hirano H-Y (2002) Isolation and characterization of a rice mutant insensitive to cool temperatures on amylose synthase. Euphytica 123:95–100
Tanase K, Otsu S, Satoh S, Onozaki T (2015) Expression levels of ethylene biosynthetic genes and senescence-related genes in carnation (Dianthus caryophyllus L.) with ultra-long-life flowers. Scient Hortic 183:31–38
Vanderstraeten L, Depaepe T, Bertrand S, Van Der Straeten D (2019) The ethylene precursor ACC affects early vegetative development independently of ethylene signaling. Front Plant Sci 10:1591. https://doi.org/10.3389/fpls.2019.01591
Velasco L, Perez-Vich B, Fernandez-Matinez JM (2004) Novel variation for the tocopherol profile in a sunflower created by mutagenesis and recombination. Plant Breed 123:490–492
Viana VE, Pegoraro C, Busanello C, Costa de Oliveira A (2019) Mutagenesis in rice: the basis for breeding a new super plant. Front Plant Sci 10:1326. https://doi.org/10.3389/fpls.2019.01326
Vogel JP, Woeste KE, Theologis A, Kieber JJ (1998) Recessive and dominant mutations in the ethylene biosynthetic gene ACS5 of Arabidopsis confer cytokinin insensitivity and ethylene overproduction, respectively. Proc Natl Acad Sci USA 95:4766–4771
Vriezen WH, Achard P, Harberd NP, Van Der Straeten D (2004) Ethylene-mediated enhancement of apical hook formation in etiolated Arabidopsis thaliana seedlings is gibberellin dependent. Plant J 37:505–516
Wu L, Luo P, Di DW, Wang L, Wang M, Lu C-K, Wei S-D, Zhang L, Zhang T-Z, Amakorová P, Strnad M, Novák O, Guo G-Q (2015) Forward genetic screen for auxin-deficient mutants by cytokinin. Sci Rep 5:11923. https://doi.org/10.1038/srep11923
Wu F, Zhang C, Wang X, Guo J, Dong L (2017) Ethylene-influenced development of tree peony cut flowers and characterization of genes involved in ethylene biosynthesis and perception. Postharv Biol Technol 125:150–160
Xu J, Shi S, Wang L, Tang Z, Lv T, Zhu X, Ding X, Wang Y, Zhao F-J, Wu Z (2017) OsHAC4 is critical for arsenate tolerance and regulates arsenic accumulation in rice. New Phytol 215:1090–1101. https://doi.org/10.1111/nph.14572
Acknowledgements
The authors would like to acknowledge the help and support provided by the late Dr. Alan D. Blowers, Ball Horticultural Co., and would like to thank Dr. Michael Uchneat, then at Ball Horticultural Co. and now at the Huck Institute of Life Sciences at Penn State University, for providing seed material and support.
Funding
This study was supported by the Jonathan Baldwin Turner Graduate Fellowship, a grant received from the Ball Helix (Ball Horticultural, Inc.), the Eugene S. Boerner Scholarship Award, and the ACES Office of Research project 65-325.
Author information
Authors and Affiliations
Contributions
LMH and SSK planned the research project; SSK supervised the project; LMH conducted all experiments, analyzed all data, and interpreted results; LMH and SSK wrote the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Zhong-Hua Chen.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
"The original online version of this article was revised”: The table 2 is revised.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heffron, L.M., Korban, S.S. Elucidating the ethylene response and tolerance in non-mutagenized and mutagenized snapdragon (Antirrhinum majus L.) lines using 1-aminocyclopropane-1-carboxylic acid (ACC). Plant Growth Regul 100, 133–145 (2023). https://doi.org/10.1007/s10725-022-00945-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-022-00945-3