Abstract
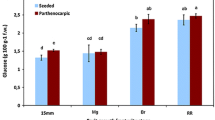
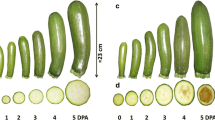
The auxin treatment in tomato plants during anthesis has been extensively used for setting fruits in adverse climatic conditions (e.g., low temperatures and inadequate light), which is well known that reduces pollen availability and fertility. Since auxin application may affect fruit composition and quality, we examined l-ascorbic acid metabolism in seeded fruit (set by natural pollination) and parthenocarpic fruit (set by auxin) in cherry tomato cv. Conchita. Specifically, we studied the oxidized and total ascorbic acid contents, the expression of all characterized genes of l-ascorbic acid metabolism, the activity of ascorbate peroxidase and dehydroascorbate reductase and the immunolocalization of ascorbate peroxidase. Differences were detected between seeded and parthenocarpic fruits, in the expression of some of the genes of ascorbic acid metabolism. However, strong presence of l-ascorbic acid peroxidase protein was detected on the developing seeds. Our data indicate that induced parthenocarpy in auxin treated plants has a significant influence in ascorbic acid metabolism comparing to seeded tomato fruits.






Similar content being viewed by others
Abbreviations
- AsA:
-
l-Ascorbic acid
- DHA:
-
Dehydroascorbic acid
- tAsA:
-
Total ascorbate (reduced AsA + DHA)
- ImG:
-
Immature green
- Mg:
-
Mature green
- Br:
-
Breaker
- RR:
-
Red ripe
- APX:
-
Ascorbate peroxidase
- MDHAR:
-
Monodehydroascorbate reductase
- DHAR:
-
Dehydroascorbate reductase
- GR:
-
Glutathione reductase
- ROS:
-
Reactive oxygen species
References
Alhagdow M, Mounet F, Gilbert L, Nunes-Nesi A, Garcia V, Just D, Petit J, Beauvoit B, Fernie AR, Rothan C, Baldet P (2007) Silencing of the mitochondrial ascorbate synthesizing enzyme l-galactono-1,4-lactone dehydrogenase affects plant and fruit development in tomato. Plant Physiol 145:1408–1422
Aoki K, Yano K, Suzuki A, Kawamura S, Sakurai N, Suda K, Kurabayashi A, Suzuki T, Tsugane T, Watanabe M, Ooga K, Torii M, Narita T, Shin IT, Kohara Y, Yamamoto N, Takahashi H, Watanabe Y, Egusa M, Kodama M, Ichinose Y, Kikuchi M, Fukushima S, Okabe A, Arie T, Sato Y, Yazawa K, Satoh S, Omura T, Ezura H, Shibata D (2010) Large-scale analysis of full-length cDNAs from the tomato (Solanum lycopersicum) cultivar Micro-Tom, a reference system for the Solanaceae genomics. BMC Genomics 11:210–215
Arrigoni O, De Gara L, Tommasi F, Liso R (1992) Changes in the ascorbate system during seed development of Vicia faba L. Plant Physiol 99:235–238
Badejo AA, Wada K, Gao Y, Maruta T, Sawa Y, Shigeoka S, Ishikawa T (2012) Translocation and the alternative d-galacturonate pathway contribute to increasing the ascorbate level in ripening tomato fruits together with the d-mannose/l-galactose pathway. J Exp Bot 63:229–239
Bartoli CG, Yu J, Gomez F, Fernandez L, McIntosh L, Foyer CH (2006) Inter-relationships between light and respiration in the control of ascorbic acid synthesis and accumulation in Arabidopsis thaliana leaves. J Exp Bot 57:1621–1631
Bulley SM, Rassam M, Hoser D, Otto W, Schunemann N, Wright M, MacRae E, Gleave A, Laing W (2009) Gene expression studies in kiwifruit and gene over-expression in Arabidopsis indicates that GDP-l-galactose guanyltransferase is a major control point of vitamin C biosynthesis. J Exp Bot 60:765–778
Chen Z, Gallie DR (2006) Dehydroascorbate reductase affects leaf growth, development, and function. Plant Physiol 142:775–787
Chen Z, Young TE, Ling J, Chang SC, Gallie DR (2003) Increasing vitamin C content of plants through enhanced ascorbate recycling. PNAS 100:3525–3530
Davey M (2003) Rocket-powered high-performance liquid chromatographic analysis of plant ascorbate and glutathione. Anal Biochem 316:74–81
Davletova S, Rizhsky L, Liang H, Shengqiang Z, Oliver DJ, Coutu J, Shulaev V, Schlauch K, Mittler R (2005) Cytosolic ascorbate peroxidase 1 is a central component of the reactive oxygen gene network of Arabidopsis. Plant Cell 17:268–281
de Jong M, Wolters-Arts M, Feron R, Mariani C, Vriezen WH (2009) The Solanum lycopersicum auxin response factor 7 (SlARF7) regulates auxin signaling during tomato fruit set and development. Plant J 57:160–170
de Pinto MC, de Gara L (2004) Changes in the ascorbate metabolism of apoplastic and symplastic spaces are associated with cell differentiation. J Exp Bot 55:2559–2569
Delis C, Krokida A, Georgiou S, Pena-Rodriguez LM, Kavroulakis N, Ioannou E, Roussis V, Osbourn AE, Papadopoulou KK (2011) Role of lupeol synthase in Lotus japonicus nodule formation. New Phytol 189:335–346
di Matteo A, Sacco A, Anacleria M, Pezzotti M, Delledonne M, Ferrarini A, Frusciante L, Barone A (2010) The ascorbic acid content of tomato fruits is associated with the expression of genes involved in pectin degradation. BMC Plant Biol 10:163–173
Eltelib HA, Fujikawa Y, Esaka M (2012) Overexpression of the acerola (Malpighia glabra) monodehydroascorbate reductase gene in transgenic tobacco plants results in increased ascorbate levels and enhanced tolerance to salt stress. S Afr J Bot 78:295–301
Forti G, Elli G (1995) The function of ascorbic acid in photosynthetic phosphorylation. Plant Physiol 109:1207–1211
Foyer CH, Noctor G (2011) Ascorbate and glutathione: the heart of the redox hub. Plant Physiol 155:2–18
Gest N, Gautier H, Steevens R (2013) Ascorbate as seen through plant evolution: the rise of a successful molecule? J Exp Bot 64:33–53
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Grantz AA, Brummell DA, Bennett AB (1995) Ascorbate free radical reductase mRNA levels are induced by wounding. Plant Physiol 108:411–418
Haroldsen VM, Chi-Ham CL, Kulkarni S, Lorence A, Bennett AB (2011) Constitutively expressed DHAR and MDHAR influence fruit, but not foliar ascorbate levels in tomato. Plant Physiol Biochem 49:1244–1249
Hazra P, Dutta AK (2010) Inheritance of parthenocarpy in tomato (Solanum lycopersicum) and its association with two marker characters. Int J Plant Sci 1:144–149
Henmi K, Demura T, Tsuboi S, Fukuda H, Iwabuchi M, Ogawa K (2005) Change in the redox state of glutathione regulates differentiation of tracheary elements in Zinnia cells and Arabidopsis roots. Plant Cell Physiol 46:1757–1765
Ioannidi E, Kalamaki MS, Engineer C, Pateraki I, Alexandrou D, Mellidou I, Giovannonni J, Kanellis AK (2009) Expression profiling of ascorbic acid-related genes during tomato fruit development and ripening and in response to stress conditions. J Exp Bot 60:663–678
Ishikawa T, Shigeoka S (2008) Recent advances in ascorbate biosynthesis and the physiological significance of ascorbate peroxidase in photosynthesizing organisms. Biosci Biotech Biochem 72:1143–1154
Ishikawa T, Dowdle J, Smirnoff N (2006) Progress in manipulating ascorbic acid biosynthesis and accumulation in plants. Physiol Plant 126:343–355
Jimenez A, Creissen G, Kular B, Firmin J, Robinson S, Verhoeyen M, Mullineaux P (2002) Changes in oxidative processes and components of the antioxidant system during tomato fruit ripening. Planta 214:751–758
Kataya AR, Reumann S (2010) Arabidopsis glutathione reductase 1 is dually targeted to peroxisomes and the cytosol. Plant Signal Behav 5:171–175
Kim IJ, Chung WI (1998) Molecular characterization of a cytosolic ascorbate peroxidase in strawberry fruit. Plant Sci 133:69–77
Li MJ, Ma FW, Zhang M, Pu F (2008) Distribution and metabolism of ascorbic acid in apple fruits (Malus domestica Borkh cv. Gala). Plant Sci 174:606–612
Li F, Wu QY, Sun YL, Wang LY, Yang XH, Meng QW (2010a) Overexpression of chloroplastic monodehydroascorbate reductase enhanced tolerance to temperature and methyl viologen-mediated oxidative stresses. Physiol Plant 139:421–434
Li M, Ma F, Liu J, Li J (2010b) Shading the whole vines during young fruit development decreases ascorbate accumulation in kiwi. Physiol Plant 140:225–237
Marin A, Ferreres F, Tomás-Barberán F, Gil MI (2004) Characterization and quantification of antioxidant constituents of sweet pepper. J Agric Food Chem 52:3861–3869
Najami N, Janda T, Barriah W, Kayam G, Tal M, Guy M, Volokita M (2008) Ascorbate peroxidase gene family in tomato: its identification and characterization. Mol Genet Genomics 279:171–182
Noctor G, Gomez L, Vanacker H, Foyer CH (2002) Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signaling. J Exp Bot 53:1283–1304
Panchuk II, Volkov RA, Schoffl F (2002) Heat stress and heat shock transcription factor-dependent expression and activity of ascorbate peroxidase in Arabidopsis. Plant Physiol 129:838–853
Pandolfini T, Rotino GL, Camerini S, Defez R, Spena A (2002) Optimization of transgene action at the post-transcriptional level: high quality parthenocarpic fruits in industrial tomatoes. BMC Biotech 2:1–11
Raffo A, Leonardi C, Fogliano V, Ambrosino P, Salucci M, Gennaro L, Bugianesi R, Giuffrida F, Quaglia G (2002) Nutritional value of cherry tomatoes (Lycopersicon esculentum cv. Naomi F1) harvested at different ripening stages. J Agric Food Chem 50:6550–6556
Schantz ML, Schreiber H, Guillemaut P, Schantz R (1995) Changes in ascorbate peroxidase activities during fruit ripening in Capsicum annuum. FEBS Lett 23:149–152
Shigeoka S, Ishikawa T, Tamoi M, Miyagawa Y, Takeda T, Yabuta Y (2002) Regulation and function of ascorbate peroxidase isoenzymes. J Exp Bot 53:1305–1319
Shu DF, Wang LW, Duan M, Deng YS, Meng QW (2011) Antisense-mediated depletion of tomato chloroplast glutathione reductase enhances susceptibility to chilling stress. Plant Physiol Biochem 49:1228–1237
Smirnoff N (2011) Vitamin C: the metabolism and functions of ascorbic acid in plants. Adv Bot Res 9:109–177. doi:10.1016/B978-0-12-385853-5.00003-9
Stevens R, Buret M, Duffé P, Garchery C, Baldet P, Rothan C, Causse M (2007) Candidate genes and quantitative trait loci affecting fruit ascorbic acid content in three tomato populations. Plant Physiol 1431:943–953
Tommasi F, Paciolla C, Arrigoni O (1999) The ascorbate system in recalcitrant and orthodox seeds. Physiol Plant 105:193–198
Tsaniklidis G, Delis C, Liakopoulos G, Karapanos I, Katinakis P, Passam HC, Aivalakis G (2012) Induced parthenocarpic cherry tomato fruits did not shown significant differences in l-ascorbate content but showed different pattern in GalLDH and GME expression. Plant Growth Regul 68:493–502
Zhang W, Lorence A, Gruszewski HA, Chevone BI, Nessler CL (2009) AMR1, an Arabidopsis gene that coordinately and negatively regulates the mannose/l-galactose ascorbic acid biosynthetic pathway. Plant Physiol 150:942–950
Zou L, Li H, Ouyang B, Zhang J, Ye Z (2006) Cloning and mapping of genes involved in tomato ascorbic acid biosynthesis and metabolism. Plant Sci 170:120–127
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tsaniklidis, G., Delis, C., Nikoloudakis, N. et al. l-Ascorbic acid metabolism in parthenocarpic and seeded cherry tomatoes. Plant Growth Regul 72, 141–153 (2014). https://doi.org/10.1007/s10725-013-9845-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-013-9845-0