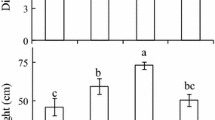
Abstract
Plants growing under canopy shade or in near-neighboring proximity of taller vegetation are the receivers of shade light conditions. The effect of light irradiance (photosynthetically active radiation [PAR]), one of the main components of shade light, on the growth of various tissues of sunflower seedlings and the possible role of auxin were investigated. Gradual reductions in PAR irradiance level from near-normal to low and very low result in significant and gradual increases in sunflower hypocotyl growth and endogenous auxin content. Similar reductions in PAR level resulted in significant and gradual decreases in sunflower cotyledon and leaf growth, and endogenous auxin content. Exogenously applied auxin increased hypocotyl elongation under near-normal PAR, where IAA levels are below optimum, but decreased elongation under very low PAR, where IAA levels are already at optimum. These results suggests that auxin acts as positive growth regulator of sunflower hypocotyls subjected to low light irradiance stress. This is further supported by the transfer experiments where seedlings transferred, for example, from near-normal PAR to very low PAR showed increased elongation associated with increased IAA levels. Therefore, it is reasonable to conclude that light irradiance-mediated changes in hypocotyl elongation of young sunflower seedlings are regulated by endogenous auxin levels.




Similar content being viewed by others
References
Ballare CL (1999) Keeping up with the neighbours: phytochrome sensing and other signalling mechanisms. Trends Plant Sci 4:97–102
Ballare CL, Scopel AL, Sanchez RA (1990) Far-red radiation reflected from adjacent leaves: an early signal of competition in plant canopies. Science 247:329–331
Behringer FJ, Davies PJ (1992) Indole-3-acetic acid levels after phytochrome-mediated changes in the stem elongation rate of dark-grown and light-grown Pisum seedlings. Planta 188:85–92
Franklin KA, Whitelam GC (2005) Phytochromes and shade-avoidance responses in plants. Ann Bot (Lond) 96:169–175
Gaskin P, MacMillan J (1991) GC-MS of the gibberellins and related compounds. Methodology and a library of spectra. University of Bristol (Cantock’s Enterprises), Bristol, UK
Hedden P (1987) Gibberellins. In: Rivier L, Crozier A (eds) Principles and practice of plant hormone analysis, vol 1. Academic Press, London, pp 9–110
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Cal Agr Exp Stat Circ 347:1–32
Jacobsen JV, Pearce DW, Poole AT, Pharis RP, Mander LN (2002) Abscisic acid, phaseic acid and gibberellin contents associated with dormancy and germination in barley. Physiol Plant 115:428–441
Koshioka M, Takeno K, Beall FD, Pharis RP (1983) Purification and separation of gibberellins from their precursors and glucosyl conjugates. Plant Physiol 73:398–406
Kurepin LV, Walton LJ, Reid DM (2007a) Interaction of red to far red light ratio and ethylene in regulating stem elongation of Helianthus annuus. Plant Growth Regul 51:53–61
Kurepin LV, Emery RJN, Pharis RP, Reid DM (2007b) The interaction of light quality and irradiance with gibberellins, cytokinins and auxin in regulating growth of Helianthus annuus hypocotyls. Plant Cell Environ 30:147–155
Kurepin LV, Emery RJN, Pharis RP, Reid DM (2007c) Uncoupling light quality from light irradiance effects in Helianthus annuus shoots: putative roles for plant hormones in leaf and internode growth. J Exp Bot 58:2145–2157
Kurepin LV, Walton LJ, Reid DM, Chinnappa CC (2010a) Light regulation of endogenous salicylic acid levels in Helianthus annuus hypocotyls. Botany 88:668–674
Kurepin LV, Walton LJ, Yeung EC, Chinnappa CC, Reid DM (2010b) The interaction of light irradiance with ethylene in regulating growth of Helianthus annuus shoot tissues. Plant Growth Regul 62:43–50
Lee SH, Reid DM (1997) The role of endogenous ethylene in the expansion of Helianthus annuus leaves. Can J Bot 78:501–508
McKeon T, Yang SF (1987) Biosynthesis and metabolism of ethylene. In: Davies PJ (ed) Plant hormones and their role in plant growth and development. Martinus Nijhoff, Boston, pp 94–112
Reid DM, Sheffer MG, Pierce RC, Bezdicek DF, Linzon SN, Revven T, Spenser MS, Vena F (1985) Ethylene in the environment: scientific criteria for assessing its effects on environmental quality. National Research Council of Canada, Ottawa, p 110 (publication 22497)
Smith H (2000) Phytochromes and light signal perception by plants—an emerging synthesis. Nature 407:585–591
Steindler C, Matteucci A, Sessa G, Weimar T, Ohgishi M, Aoyama T, Morelli G, Ruberti I (1999) Shade avoidance responses are mediated by the ATHB-2 HD-Zip protein, a negative regulator of gene expression. Development 126:4235–4245
Vandenbussche F, Vriezen WH, Smalle J, Laarhoven LJ, Harren FJ, Van Der Straeten D (2003) Ethylene and auxin control the Arabidopsis response to decreased light irradiance. Plant Physiol 133:517–527
Vandenbussche F, Pierik R, Millenaar FF, Voesenek LA, Van Der Straeten D (2005) Reaching out of the shade. Curr Opin Plant Biol 8:462–468
Walton LJ, Kurepin LV, Reid DM, Chinnappa CC (2010) Narrow-band light regulation of ethylene and gibberellin levels in hydroponics-grown Helianthus annuus hypocotyls and roots. Plant Growth Regul 61:53–59
Zobel RW, Robert LW (1978) Effect of low concentration of ethylene on cell division and cell differentiation in lettuce pit explants. Can J Bot 56:987–990
Acknowledgments
We would like to thank Ms. Bonnie Smith and Mr. Ken Girard for excellent greenhouse assistance. This work was funded by NSERC (Canada) grant to DMR and ECY.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurepin, L.V., Walton, L.J., Yeung, E.C. et al. The interaction of light irradiance with auxin in regulating growth of Helianthus annuus shoots. Plant Growth Regul 65, 255–262 (2011). https://doi.org/10.1007/s10725-011-9596-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-011-9596-8