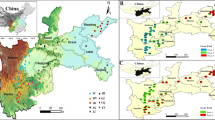
Abstract
Crataegus bretschneideri C. K. Schneid. originated in Northeast China, it has remarkable characteristics of cold resistance and early maturity. Due to its unclear taxonomic status, the conservation and utilization of C. bretschneideri have been limited. In this study, the molecular and phenotypic diversity of C. bretschneideri and relative species were systematically assessed. The results showed the high levels of genetic variation in leaf and fruit phenotypic traits in thirty-six Crataegus accessions based on the Shannon–wiener index and principal component analyses. According to the phenotypic cluster analysis, Crataegus accessions were clustered into five main clades. The genetic diversity and population structure of seven Crataegus species were analyzed by thirteen reported and five newly designed SCoT markers. In total, eighteen SCoT markers produced 148 polymorphic bands with 94.87% polymorphic loci. Thirty-six Crataegus accessions were divided into five clades in the phylogenetic tree constructed by SCoT markers. C. bretschneideri accessions were clustered into a single clade which had a closer relationship with C. pinnatifida Bunge accessions. Population structure analyses indicated that there was a mixed gene pool in C. bretschneideri accessions. These findings revealed that C. bretschneideri may be of hybrid origin. Newly designed SCoT markers are practical for genetic diversity analyses of genus Crataegus.






Similar content being viewed by others
Data Availability
The data presented in this study are available in supplementary material.
References
Amom T, Tikendra L, Apana N, Goutam M, Sonia P, Koijam AS, Potshangbam AM, Rahaman H, Nongdam P (2020) Efficiency of RAPD, ISSR, iPBS, SCoT and phytochemical markers in the genetic relationship study of five native and economical important bamboos of North-East India. Phytochemistry 174:112330. https://doi.org/10.1016/j.phytochem.2020.112330
Betancourt-Olvera M, Nieto-Ángel R, Urbano B, González-Andrés F (2018) Analysis of the biodiversity of hawthorn (Crataegus spp.) from the morphological, molecular, and ethnobotanical approaches, and implications for genetic resource conservation in scenery of increasing cultivation: the case of Mexico. Genet Resour Crop Evol 65:897–916. https://doi.org/10.1007/s10722-017-0583-4
Bhattacharyya P, Kumaria S, Kumar S, Tandon P (2013) Start Codon Targeted (SCoT) marker reveals genetic diversity of Dendrobium nobile Lindl., an endangered medicinal orchid species. Gene 529:21–26. https://doi.org/10.1016/j.gene.2013.07.096
Christensen KI (1992) Revision of Crataegus Sect. Crataegus and Nothosect. Crataeguineae (Rosaceae-Maloideae) in the Old World. Syst Bot Monogr 35:1–199. https://doi.org/10.2307/25027810
Collard BCY, Mackill DJ (2009) Start Codon Targeted (SCoT) polymorphism: a simple, novel dna marker technique for generating gene-targeted markers in plants. Plant Mol Biol Rep 27:86. https://doi.org/10.1007/s11105-008-0060-5
Dai HY, Guo XW, Zhang Y, Li YY, Li H, Zhou CS, Zhang ZH (2008) Genetic diversity of Crataegus pinnatifida Bge. as evaluated by RAPD and ISSR markers. Acta Hortic Sin 35:1117–1124
Dickinson TA, Belaoussoff S, Love RM, Muniyamma M (1996) North American black-fruited hawthorns. I. Variation in floral construction, breeding system correlates, and their possible evolutionary significance in Crataegus sect. Douglasii London. Folia Geobot 31:355–371. https://doi.org/10.1007/BF02815380
Dickinson TA, Proctor S, Shipley PR, Lee J, Coughlan J, Zarrei M (2014) Natural health products and Crataegus of the Pacific Northwest. Paper presented at the Proceedings of the Annual Meeting. https://www.rom.on.ca/sites/default/files/imce/tadickinson.pdf. Accessed June 2014
Dong WX (2013) Technical specifications for crop germplasm characterization and evaluation of Hawthorn. China Agriculture Press, Beijing
Dong WX, Li ZX (2015) The science and practice of Chinese fruit tree: Hawthorn. Science Press, Shanxi
Dönmez EO (2008) Pollen morphology in Turkish Crataegus (Rosaceae). Bot Helv 118:59–70. https://doi.org/10.1007/s00035-008-0823-5
Du X, Zhang X, Bo HD, Zhang TC, Lao YC, Dong WX (2019) Molecular analysis of evolution and origins of cultivated hawthorn (Crataegus spp.) and related species in China. Front Plant Sci 10:443. https://doi.org/10.3389/fpls.2019.00443
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-011-9548-7
Edwards JE, Brown PN, Talent N, Dickinson TA, Shipley PR (2012) A review of the chemistry of the genus Crataegus. Phytochemistry 79:5–26. https://doi.org/10.1016/j.phytochem.2012.04.006
Emami A, Shabanian N, Rahmani MS, Khadivi A, Mohammad-Panah N (2018) Genetic characterization of the Crataegus genus: implications for in situ conservation. Sci Hortic 231:56–65. https://doi.org/10.1016/j.scienta.2017.12.014
Feng SG, He RF, Jiang MY, Lu JJ, Shen XX, Liu JJ, Wang ZA, Wang HZ (2016) Genetic diversity and relationships of medicinal Chrysanthemum morifolium revealed by start codon targeted (SCoT) markers. Sci Hortic 201:118–123. https://doi.org/10.1016/j.scienta.2016.01.042
Guo T, Jiao P (1995) Hawthorn (Crataegus) resources in China. Hort Sci 30:1132–1134. https://doi.org/10.21273/HORTSCI.30.6.1132
Han X, Liang Y, Wang Y, Li F, Guo T, Xue Y (2009) Analysis of the origin and classification of C. brettschnederi by ISSR markers. J Jilin Agric Univ 31:164–167
Hong DY, Blackmore S (2015) Plants of China: a companion to the flora of China. Cambridge University Press, London
Hu G, Wang Y, Wang Y, Zheng S, Dong W, Dong N (2021) New insight into the phylogeny and taxonomy of cultivated and related species of Crataegus in China, based on complete chloroplast genome sequencing. Horticulturae 7:301. https://doi.org/10.3390/horticulturae7090301
Hubisz MJ, Falush D, Stephens M, Pritchard JK (2009) Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour 9:1322–1332. https://doi.org/10.1111/j.1755-0998.2009.02591.x
Igwe DO, Afiukwa CA, Ubi BE, Ogbu KI, Ojuederie OB, Ude GN (2017) Assessment of genetic diversity in Vigna unguiculata L. (Walp) accessions using inter-simple sequence repeat (ISSR) and start codon targeted (SCoT) polymorphic markers. BMC Genet 18:98. https://doi.org/10.1186/s12863-017-0567-6
Jiang YL, Dong WX (2009) Diversity analysis of morphological characters in hawthorn germplasm resources. Northern Fruits 1:8–10. https://doi.org/10.16376/j.cnki.bfgs.2009.01.022
Khadivi A, Heidari P, Rezaei M, Safari-Khuzani A, Sahebi M (2019) Morphological variabilities of Crataegus monogyna and C. pentagyna in northeastern areas of Iran. Ind Crops Prod 139:111531. https://doi.org/10.1016/j.indcrop.2019.111531
Long ZJ, Fan LZ, Xu G, Hu SL, Han GH (2015) Application advance of SCoT molecular markers in plants. J Plant Genet Resour 16:336–343. https://doi.org/10.13430/j.cnki.jpgr.2015.02.019
Lu BR (2014) Genetic and evolutionary effects of hybridization-introgression and their implications for conservation of crop wild relative species. Sci Bull 59:479–492. https://doi.org/10.1360/972013-660
Pakseresht F, Talebi R, Karami E (2013) Comparative assessment of ISSR, DAMD and SCoT markers for evaluation of genetic diversity and conservation of landrace chickpea (Cicer arietinum L.) genotypes collected from north-west of Iran. Physiol Mol Biol Plants 19:563–574. https://doi.org/10.1007/s12298-013-0181-7
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Phipps JB (2005) A review of hybridization in north american hawthorns. Another look at “the Crataegus problem.” Ann Missouri Bot Gard 92(1):113–126
Phipps J, Dvorsky KA (2008) A taxonomic revision of Crataegus series Lacrimatae (Rosaceae). J Bot Res Inst Tex 2:1101–1162
Phipps JB (2014) Flora of North America Editorial Committee (Eds.). Flora of North America North of Mexico. Oxford University Press, New York
Rohlf FJ (2000) NTSYS-pc: numerical taxonomy and multivariate analysis system version 2.1. Exeter Publishing Setauket, New York
Saboori S, Noormohammadi Z, Sheidai M, Marashi S (2020) SCoT molecular markers and genetic fingerprinting of date palm (Phoenix dactylifera L.) cultivars. Genet Resour Crop Evol 67:73–82. https://doi.org/10.1007/s10722-019-00854-x
Sheng F, Chen SY, Tian J, Li P, Qin X, Wang L, Li J (2017) Morphological and ISSR molecular markers reveal genetic diversity of wild hawthorns (Crataegus songorica K. Koch.) in Xinjiang, China. J Integr Agric 16:2482–2495. https://doi.org/10.1016/S2095-3119(17)61688-5
Šnjegota D, Stronen AV, Boljte B, Ćirović D, Djan M, Huber D, Jelenčič M, Konec M, Kusak J, Skrbinšek T (2021) Population genetic structure of wolves in the northwestern Dinaric-Balkan region. Ecol Evol 11:18492–18504. https://doi.org/10.1002/ece3.8444
Talent N, Dickinson T (2007) Ploidy level increase and decrease in seeds from crosses between sexual diploids and asexual triploids and tetraploids in Crataegus L. (Rosaceae, Spiraeoideae, Pyreae). Can J Bot 85:570–584. https://doi.org/10.1139/B07-028
Wu F, Zhang Z, Dai H, Zhang Y, Chang L (2008) Genetic relationship of some Crataegus spp. (Hawthorn) revealed by chloroplast DNA PCR-RFLP. J Biotechnol 136:S103. https://doi.org/10.1016/j.jbiotec.2008.07.235
Xiao P, Liu H, Wang D, Tang W, Yang H, Wang C, Tian Y (2020) Assessment of genetic diversity in Camellia oleifera Abel. accessions using inter-simple sequence repeat (ISSR) and start codon targeted (SCoT) polymorphic markers. Genet Resour Crop Evol 67:1115–1124. https://doi.org/10.1007/s10722-020-00924-5
Yeh FC, Yang RC, Boyle TB (1999) POPGENE v.3.2. The user-friendly shareware for population genetics analysis. University of Alberta, Canadá: Edmonton Molecular Biology and Biotechnology Centre
Zhang X, Zhang Q, Sun X, Du X, Liu W, Dong W (2019) Differential expression of genes encoding phenylpropanoid enzymes in an apricot cultivar (Prunus armeniaca L.) with cleavable endocarp. Trees 33:1695–1710. https://doi.org/10.1007/s00468-019-01890-x
Zhao HC, Feng BT (1996) China fruit-plant monograph of hawthorn (Crataegus) Flora. China Forest Press, Beijing, China
Funding
This work was supported by “The Conservation and Utilization of Crop Germplasm Resource–Hawthorn (Project # 19190178; 19200357)”.
Author information
Authors and Affiliations
Contributions
XZ wrote the manuscript, analyzed the data and designed the research. WD provided the funding for the experiment. XS, JW, MX, and SC conducted experiments. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Information Fig. S1
Amplification profiles of primers SCoT4 (a) and SCoTH1 (b). Lane M: Trans2K®DNA Marker, Lanes 1–14: fourteen individuals belonging to C. bretschneideri, Lanes 15–16: two individuals belonging to C. maximowiczii, Lanes 17–19: three individuals belonging to C. sanguinea, Lanes 20–27: eight individuals belonging to C. pinnatifida, Lanes 28–31: four individuals belonging to C. hupehensis, Lanes 32–34: three individuals belonging to C. scabrifolia, Lanes 35–36: two individuals belonging to C. altaica. Details of Crataegus individuals were in Table 1. Supplementary file1 (TIF 2452 KB)
Supplementary Information Fig. S2
△K value calculated by Structure Harvester; When K = 5, △K was maximum. Supplementary file2 (TIF 189 KB)
Supplementary Information Table S1
Scales of qualitative traits of genus Crataegus. Supplementary file3 (XLSX 16 KB)
Supplementary Information Table S2
SCoT markers statistical data of Crataegus individuals. Supplementary file4 (XLSX 36 KB)
Supplementary Information Table S3
Assessment results of phenotypic traits of Crataegus accessions. Supplementary file5 (XLSX 23 KB)
Supplementary Information Table S4
Diversity analyses of qualitative traits of Crataegus accessions. Supplementary file6 (XLSX 18 KB)
Supplementary Information Table S5
Principal component analysis for Crataegus accessions based multiply phenotypic traits. Supplementary file7 (XLSX 20 KB)
Supplementary Information Table S6
Genetic similarities of Crataegus accessions based on the Jaccard’s coefficients. Supplementary file8 (XLSX 21 KB)
Supplementary Information Table S7
Genetic Distance of Crataegus accessions based on SCoT markers. Supplementary file9 (XLSX 18 KB)
Supplementary Information Table S8
Principal coordinate analysis of Crataegus accessions based on genetic distances. Supplementary file10 (XLSX 36 KB)
Rights and permissions
About this article
Cite this article
Zhang, X., Sun, X., Wang, J. et al. Evaluation of molecular and phenotypic diversity of Crataegus bretschneideri C. K. Schneid. and related species in China. Genet Resour Crop Evol 70, 221–234 (2023). https://doi.org/10.1007/s10722-022-01428-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-022-01428-0