Abstract
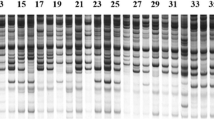
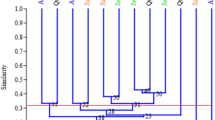
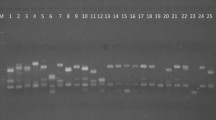
Erianthus arundinaceus, a wild relative of sugarcane has a number of traits including tolerance to environmental stresses desired by sugarcane breeders. The value of genetic diversity studies in providing useful information for introgression breeding and germplasm conservation is well known. A previous study reported little genetic diversity in a number of Indonesian E. arundinaceus while Indian E . arundinaceus was more diverse. A small number of Chinese E. arundinaceus was observed as being relatively diverse. However, an assessment of the genetic diversity of E. arundinaceus from the major geographical areas in the world has not been reported before. In this study, 207 E. arundinaceus accessions from China, Indonesia, India, New Guinea, Philippines and Vietnam were sampled for genetic diversity analysis using AFLP markers. Three AFLP primer combinations generated 286 markers in total, with the percentage of polymorphism observed being 99.3%. A dendrogram and PCA diagram were constructed based on the matrix of similarity analyzed with UPGMA method by NTSYS software. The results showed that: (1) a high level of diversity found within the Chinese collection compared with the Indonesian collection and (2) there was a general division amongst clones from China based on those collected from eastern versus western parts of the country, and clones from Yunnan province (western China) were the most diverse.



Similar content being viewed by others
References
Aitken KS, Li JC, Jackson PJ, Piperidis G, McIntyre CL (2006) AFLP analysis of genetic diversity within S. officinarum and comparison with sugarcane cultivars. Aust J Agric Res 57:1167–1184
Aitken KS, Li JC, Wang L, Qing C, Fan YH, Jackson P (2007) Characterization of intergeneric hybrids of Erianthus rockii and Saccharum using molecular markers. Genet Resour Crop Evol 54:1395–1405
Berding N, Koike H (1980) Germplasm conservation of the Saccharum complex: a collection from the Indonesian Archipelago. Hawaii Plant Rec 59:176–187
Berding N, Roach BT (1987) Germplasm collection, maintenance, and use. In: Heinz DJ (ed) Sugarcane improvement through breeding. Elsevier, Amsterdam, pp 143–210
Besse P, McIntyre CL, Berding N (1996) Ribosomal DNA variations in Erianthus, a wild sugarcane relative (Andropogoneae-Saccharinae). Theor Appl Genet 92:733–743
Besse P, McIntyre CL, Berding N (1997) Characterisation of Erianthus section Ripidium and Saccharum germplasm (Androponeae-Saccharum) using RFLP markers. Euphytica 93:283–292
Besse P, Taylor G, Carrol B, Berding N, Burner D, McIntyre CL (1998) Assessing genetic diversity in a sugar cane germplasm collection using an automated AFLP analysis. Genetica 104:143–153
Burner DM (1991) Cytogenetic analyses of sugarcane relatives (Andropogoneae: Saccharinae). Euphytica 54:125–133
Cai Q, Aitken K, Fan YH, Piperidis G, Jackson P, McIntyre CL (2005) A preliminary assessment of the genetic relationship between Erianthus rockii and the “Saccharum Complex” using microsatellite and AFLP markers. Plant Sci 169:976–984
Cai Q, Fan YH, Ma L,Ying XM, Liu XL, Wang LP (2006) In: Sharing and utilization research of perennial and asexual propagation crops. Exploration, conservation, characterization and utilization for sugarcane germplasm in China. Germplasm. China Agricultural Press, Beijing, pp 93-100
Chen SL (1997) Gramineae (5). In: Botanical Gazetteer of China Vol 10(2), Sciences publisher, Beijing China, pp 43-45
Deng HH, Liao ZZ, Li QW, Lao FY, Fu C, Chen XW (2002) Breeding and isozyme marker assisted selection of F2 hybrids from Saccharum spp. × Erianthus arundinaceus. Sugarcane Canesugar (Chinese) 1:1–5
D’Hont A, Rao PS, Feldmann P, Grivet L, Islam-Faridi N, Taylor P, Glaszmann JC (1995) Identification and characterisation of sugarcane intergeneric hybrids, Saccharum officinarum × Erianthus arundinaceus, with molecular markers and DNA in situ hybridisation. Theor Appl Genet 91:320–326
Glaszmann JC, Noyer JC, Fautret A, Feldmann P, Lanaud C (1989) Biochemical genetic markers in sugarcane. Theor Appl Genet 78:537–543
Hoisington DA (1992) Laboratory protocols: applied molecular genetics laboratory. CIMMYT, Mexico
Jackson P, Henry RJ (2011) Erianthus. In: Kole C (ed) Wild crop relatives: genomic and breeding resources: industrial crops. Springer, Berlin, pp 97–109
Lima MLA, Garcia AAF, Oliveira KM, Matsuoka S, Arizono H (2002) Analysis of genetic similarity detected by AFLP and coefficient of parentage among genotypes of sugarcane (Saccharum spp.). Theor Appl Genet 104:30–38
Lu YH, D’Hont A, Walker DIT, Rao PS, Feldmann P, Glaszmann JC (1994) Relationships among ancestral species of sugarcane revealed with RFLP using single copy maize nuclear probes. Euphytica 78:7–18
Mohan N, Sreenivasan TV (1983) Chromosome number in the genus Erianthus, Michx. (Poaceae) of Indonesian Arquipelago. Cell Chromosome Res 6:14–16
Nair NV, Mary Sheji (2006) RAPD analysis reveals the presence of mainland Indian and Indonesian forms of Erianthus arundinaceus (Retz.) Jeswiet in the Andaman–Nicobar Islands. Curr Sci 90(8):1118–1122
Piperidis N, Jian-wen C, Hai-hua D, Li-Ping W, Phillip J, George P (2010) GISH characterisation of Erianthus arundinaceus chromosomes in three generations of sugarcane intergeneric hybrids. Genome 53(5):331–336
Pu YS, Zhang ZY, Pu LN (2007) Strategic studies on the biodiversity sustainability in Yunnan Province, Southwest China. For Stud China 3:225–237
Rohlf FJ (1997) NTSYS-PC: numerical taxonomy and multivariate analysis system. Version 2.1. Applied Biostatistics, New York
Sneath PHA and Sokal RR (1973) Numerical taxonomy: the principles and practice of numerical classification. Series of books in biology. W.H. Freeman and Company, San Francisco
Vos P, Hogers R, Bleeker M, Reijans M, Van Der Lee T, Hornes M (1995) AFLP: a new concept for DNA fingerprinting. Nucleic Acids Res 23:4407–4414
Wu ZY, Raven PH (1994) Flora of China (English edition), vol 22, pp 571, 576, 589, Saccharum arundinaceum (online sources) .http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=200026230
Zhang MQ, Hong YX, Li QW, Liu SM, Zhang CM, Yang RZ (2004) Molecular polymorphic analyses for the germplasms of Erianthus arundinacues collected in China. J Plant Res Environ 13(1):1–6
Zhang HY, Li FS, Liu XZ, He LL, Yang QH, He SC (2008) Analysis of genetic variation in Erianthus arundinaceum by random amplified polymorphic DNA markers. Afr J Biotechnol 7(19):3414–3418
Acknowledgments
This work was supported by the Australian Centre for International Agricultural Research (ACIAR project code CS1/2000/038); the Yunnan Provincial Science and Technology Department (YPST project code 2006C0013Z and 2009CI059). We would like to thank L. Ma, J. Mao and Y. Bi at YSRI (Yunnan Sugarcane Research Institute) People’s Republic of China, for their assistance with the leaf materials collecting and figures colored. Thanks also to J.C. Li at CSIRO for her assistance with the molecular experiments, and to Dr Raymond Schnell (USDA, Miami) and Dr William Burnquist (Copersucar) for allowing access to the germplasm collections to collect leaf material.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cai, Q., Aitken, K.S., Fan, Y.H. et al. Assessment of the genetic diversity in a collection of Erianthus arundinaceus . Genet Resour Crop Evol 59, 1483–1491 (2012). https://doi.org/10.1007/s10722-011-9776-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-011-9776-4