Abstract
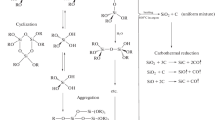
This paper reports on the results of rheological investigations into the kinetics of gelation of the sol-gel systems based on a water-alcohol solution of tetraethoxysilane (TEOS) modified by a number of metal salts and organic polyhydroxyl compounds, such as glycerol, poly(ethylene glycol) (PEG-300), and hyperbranched aliphatic complex polyol polyethers (HBP-49, HBP-64). It is demonstrated that, at a dispersed phase concentration of approximately 10 wt %, water-ethanol sol-gel systems containing 2–4 g/l of high-molecular additives possess a shear strength and are characterized by anomalous properties in their flow. The gelation time of sols in the presence of organic additives is affected by the ratio between the numbers of OH− hydrophilic and CH3 hydrophobic groups in the polymer molecule, the concentration of the introduced additive, and the electrolyte multivalence. The presence of metal salts enhances the structuring effect of polymers.
Similar content being viewed by others
REFERENCES
Iler, R.K., The Chemistry of Silica: Solubility, Polymerization, Colloid and Surface Properties, and Biochemistry of Silica, New York: Wiley, 1979. Translated under the title Khimiya kremnezema, Moscow: Mir, 1982.
Brinker, C.F. and Scherer, G.W., Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing, San Diego: Academic, 1990.
Yoldas, B.E., Introduction and Effect of Structural Variations in Inorganic Polymers and Glass Network, J. Non-Cryst. Solids, 1982, vol. 51, no.1, pp. 105–121.
Mackenzie, J., Sol-Gel Researches—Achievements Since 1981 and Prospects for the Future, Sol-Gel Sci. Technol., 2003, vol. 26, nos.1–3, pp. 23–27.
Sviderskii, V.A., Voronkov, M.G., Klimenko, V.S., and Klimenko, S.V., Hydrolytic Polycondensation of Tetraethoxysilane with Salts and Oxides of Metals in the Sol-Gel Process, Zh. Prikl. Khim. (St. Petersburg), 1997, vol. 70, no.10, pp. 1698–1703.
Suikovskaya, N.V., Khimicheskie metody polucheniya tonkikh prozrachnykh plenok (Chemical Methods Used for the Preparation of Thin Transparent Films), Leningrad: Khimiya, 1971.
Nikolaeva, L.V. and Borisenko, A.I., Tonkosloinye stekloemalevye i steklokeramicheskie pokrytiya (Thin-Layer Glass-Enamel and Glass-Ceramic Coatings), Leningrad: Nauka, 1980.
Shilova, O.A., Tarasyuk, E.V., Shevchenko, V.V., Klimenko, N.S., Movchan, T.G., Hashkovsky, S.V., and Shilov, V.V., The Influence of Low-and High-Molecular Hydroxyl-Containing Additives on the Stability of Sol-Gel Tetraethoxysilane-Based Systems and on the Structure of Hybrid Organic-Inorganic Coatings, Fiz. Khim. Stekla, 2003, vol. 29, no.4, pp. 527–541 [Glass Phys. Chem. (Engl. transl.), 2003, vol. 29, no. 4, pp. 378–389].
Movchan, T.G., Ur’ev, N.B., Khamova, T.V., Potapov, A.V., Izhik, A.P., Tarasyuk, E.V., Shilova, O.A., and Pisarev, S.A., Rheology and Structuring of Nanodisperse Systems Based on Tetraethoxysilane, Mekhan. Kompozits. Mater. Konstr., 2004, vol. 10, no.1, pp. 118–132.
Prokopenko, V.A., Pertsov, N.V., Shilov, V.N., and Dul’neva, T.Yu., Aggregative Stability of Mineral Disperse Systems at High Electrolyte Concentrations, Kolloidn. Zh., 1994, vol. 56, no.6, pp. 820–823.
Andreeva, I.A., Romanova, L.G., and Sushko, V.A., Investigation into the Colloidal Chemical Processes in Aqueous Dispersions of Ethoxysilanes, Kolloidn. Zh., 1982, vol. 44, no.2, pp. 195–200.
Frolov, Yu.G., Shabanova, N.A., and Molodchikova, S.I., Regularities of the Variations in the Viscosity of Silica Hydrosol, Kolloidn. Zh., 1983, vol. 45, no.5, pp. 970–974.
Shchukin, E.D. and Rebinder, P.A., On the Mechanism of Elastic Aftereffect in Structured Suspensions of Bentonite with a Low Concentration, Kolloidn. Zh., 1971, vol. 33, no.3, pp. 450–458.
Golikova, E.V., Khamova, T.V., and Shilova, O.A., Investigation into the Factors Responsible for the Stability of Hydrolyzed Tetraethoxysilane-Based Colloidal Systems Used for Forming Thin Films and Surface Coatings, in Temperaturoustoichivye funktsional’nye pokrytiya. Trudy XIX Vserossiiskogo soveshchaniya (Proceedings of the XIX All-Russia Conference on Thermally Stable Functional Coatings), St. Petersburg: Yanus, 2003, vol. 1, pp. 83–86.
Batchelor, G.K., The Effect of Brownian Motion on the Bulk Stress in a Suspension of Spherical Particles, J. Fluid Mechan., 1977, vol. 83, no.1, pp. 97–117.
Efremov, I.F., Gelation of Dilute Sols and Suspensions, Kolloidn. Zh., 1956, vol. 18, no.3, pp. 276–284.
Pakhovchishin, S.V., Gritsenko, V.F., Bogatyrev, V.M., and Artemov, A.S., The Influence of Ageing on the Rheological Properties of Aqueous Dispersions of Phosphorus-Containing Silica, Kolloidn. Zh., 1996, vol. 58, no.5, pp. 656–658.
Makarov, A.S., Andreeva, I.A., and Tretinnik, V.Yu., Rheological Properties of Polymer-Containing Aqueous Dispersions of Aerosil, Kolloidn. Zh., 2001, vol. 63, no.6, pp. 801–807.
Rebinder, P.A., Physicochemical Mechanics of Disperse Structures, in Fiziko-khimicheskaya mekhanika dispersnykh struktur (Physicochemical Mechanics of Disperse Structures), Moscow: Nauka, 1966, pp. 3–16.
Akopova, O.V. and Eremenko, B.V., Effect of Electrolytes on the Stability of Silica Suspensions in Poly(ethylene oxide) Solutions, Kolloidn. Zh., 1994, vol. 56, no.1, pp. 5–10.
Efremov, I.F., Lukashenko, G.M., and Terent’eva, E.A., Dilatancy of Colloidal Structures, Kolloidn. Zh., 1980, vol. 42, no.5, pp. 859–865.
Trapeznikov, A.A. and Frolova, E.A., Change in the Rheological Properties of Concentrated Suspensions of Soot in Oil Due to Coagulation and Peptization at Different Shear Rates, Kolloidn. Zh., 1978, vol. 40, no.4, pp. 728–734.
Tikhonov, A.P., Senapova, O.V., and Krivoshchepov, A.F., Investigation into the Properties of Al2O3 Suspensions in Relation to the Fabrication of Ceramic Products by the Electrophoretic Technique, Kolloidn. Zh., 1976, vol. 38, no.5, pp. 1022–1025.
Tikhonov, A.P. and Krivoshchepov, A.F., Dilatant-Thixotropic Transformations of Periodic Colloidal Structures, Kolloidn. Zh., 1979, vol. 41, no.6, pp. 1212–1213.
Adsorption from Solutions on the Solid/Liquid Interface, Parfitt, G. and Rochester, C., Eds., London: Academic, 1983. Translated under the title Adsorbtsiya iz rastvorov na poverkhnosti tverdykh tel, Moscow: Mir, 1986.
Yakhnin, E.D. and Taubman, A.P., Macromosaic Structure of the Surface of Adsorptively Modified Particles of the Dispersed Phase and Its Role in the Structure Formation, in Doklady konferentsii “Fiziko-khimicheskaya mekhanika pochv, gruntov, glin i stroitel’nykh materialov” (Proceedings of the Conference “Physicochemical Mechanics of Soils, Grounds, Clays, and Building Materials”), Tashkent: FAN, 1966.
Shabanova, N.A. and Kodintseva, E.Yu., The Influence of the Initial Conditions on the Kinetics of Gelation in Silica Hydrosols, Kolloidn. Zh., 1990, vol. 52, no.3, pp. 553–563.
Ravaine, D., Seminel, A., Charbouillot, Y., and Vincens, M., A New Family of Organically Modified Silicates Prepared from Gels, J. Non-Cryst. Solids, 1986, vol. 82, nos.1–3, pp. 210–219.
Khil’ko, S.L., Titov, E.V., and Fedoseeva, A.A., The Influence of Strong Electrolytes and High Temperature on the Formation of Discontinuities in Flow of Water-Coal Suspensions, Kolloidn. Zh., 2001, vol. 63, no.6, pp. 845–849.
Taubman, A.B., Yakhnin, E.D., and Voronkov, V.A., On the Kinetics of Structuring and Thixotropic Properties of Silica Suspensions As Models of Filled Polymeric Systems, in Fiziko-khimicheskaya mekhanika dispersnykh struktur (Physicochemical Mechanics of Disperse Structures), Moscow: Nauka, 1966, pp. 39–44.
Shabanova, N.A. and Silos, I.V., Transformation of Sols into Gels under the Conditions of Electrolytic Coagulation of Colloidal Silica, Kolloidn. Zh., 1996, vol. 58, no.2, pp. 266–271.
Author information
Authors and Affiliations
Additional information
Original Russian Text Copyright © 2005 by Fizika i Khimiya Stekla, Movchan, Ur’ev, Khamova, Tarasyuk, Potapov, Shilova, Klimenko, Shevchenko.
Rights and permissions
About this article
Cite this article
Movchan, T.G., Ur’ev, N.B., Khamova, T.V. et al. Kinetics of structuring in the sol-gel systems based on tetraethoxysilane with organic additives: I. Sols. Glass Phys Chem 31, 219–228 (2005). https://doi.org/10.1007/s10720-005-0045-5
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10720-005-0045-5