Abstract
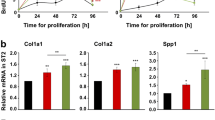
Glycosaminoglycans (GAG) and proteoglycans, which are components of the extracellular bone matrix, are also localized in and at the membrane of osteoblasts and in the pericellular matrix. Due to their interaction with several growth factors, water and cations these molecules play an important role in regulating proliferation and differentiation of osteoblasts and bone development. The aim of this study was to assess in vitro the effects of two chemically sulfated hyaluronan (HyaS) derivatives on the proliferation of rat calvarial osteoblasts and to compare with those of native hyaluronan (Hya) and natural sulfated GAG such as chondroitin-4-sulfate (C4S), chondroitin-6-sulfate (C6S), dermatan sulfate (DS) and heparan sulfate (HS). Moderately and highly sulfated HyaS derivatives caused a time-dependent reduction of osteoblast proliferation. The anti-proliferative effect of HyaS was accompanied by a cell cycle arrest in the G1 phase, but was not associated with cell death. Whereas non-sulfated high molecular weight (HMW)- and low molecular weight (LMW)-Hya as well as C4S, C6S, DS and HS showed no effect on the cell proliferation.



Similar content being viewed by others
Abbreviations
- GAG:
-
glycosaminoglycans
- HyaS:
-
sulfated hyaluronan
- CS:
-
chondroitin sulfate
- DS:
-
dermatan sulfate
- HS:
-
heparan sulfate
- HMW:
-
high molecular weight
- LMW:
-
low molecular weight
References
Manton, K.J., Haupt, L.M., Vengadasalam, K., Nurcombe, V., Cool, S.M.: Glycosaminoglycans and growth factor mediated murine calvarial cell proliferation. J. Mol. Hist. 38, 415–424 (2007)
Cool, S.M., Nurcombe, V.: The osteoblast-heparan axis: control of the bone cell lineage. Int. J. Biochem. Cell. Biol. 37, 1739–1745 (2005)
Taylor, K.R., Gallo, R.L.: Glycosaminoglycans and their proteoglycans: host associated molecular patterns for initiation and modulation of inflammation. FASEB J. 20, 9–22 (2006)
Hardingham, T.E., Fosang, A.J.: Proteoglycans: many forms and many functions. FASEB J. 6, 861–870 (1992)
Bali, J.P., Cousse, H., Neuzil, E.: Biochemical basis of the pharmacological action of chondroitin sulfates on the osteoarticular system. Semin. Arthritis Rheum. 31, 58–68 (2001)
Nikitovic, D., Zafiropoulos, A., Tzanakakis, G.N., Karamanos, N.K., Tsatsakis, A.M.: Effects of glycosaminoglycans on cell proliferation of normal osteoblasts and human osteosarcoma cells depend on their type and fine chemical structure. Anticancer Res. 25, 2851–2856 (2005)
Ling, L., Murali, S., Dombrowski, C., Haupt, L.M., Stein, G.S., van Wijnen, A.J., Nurcombe, V., Cool, S.M.: Sulfated glycosaminoglycans mediate the effects of FGF2 on the osteogenic potential of rat calvarial osteoprogenitor cells. J. Cell. Physiol. 209, 811–825 (2006)
Grünert, M., Dombrowski, C., Sadasivam, M., Manton, K., Cool, S.M.: Isolation of a native osteoblast matrix with a specific affinity for BMP2. J. Mol. Histol. 38, 393–404 (2007)
Jackson, R.A., Nurcombe, V., Cool, S.M.: Coordinated fibroblast growth factor and heparan sulfate regulation of osteogenesis. Gene 379, 79–91 (2006)
Manton, K.J., Sadasivam, M., Cool, S.M., Nurcombe, V.: Bone-specific heparan sulfates induce osteoblast growth arrest and downregulation of retinoblastoma protein. J. Cell. Physiol. 209, 219–229 (2006)
Isaksson, A., Önnervik, P.O., Malmström, A., Fransson, L.A.: L-iduronate-rich glycosaminoglycans inhibit growth of normal fibroblasts independently of serum or added growth factors. Exp. Cell. Res. 206, 93–99 (1993)
Syrokou, A., Tzanakakis, G., Tsegenidis, T., Hjerpe, A., Karamanos, N.K.: Effects of glycosaminoglycans on proliferation of epithelial and fibroblast human malignant mesothelioma cells: a structure-function relationship. Cell. Prolif. 32, 85–99 (1999)
Volpi, N., Petrini, M., Conte, A., Valentini, P., Venturelli, T., Bolognani, L., Ronca, G.: Effects of glycosaminoglycans on U-937 leukemia cell proliferation and differentiation: structure-function relationship. Exp. Cell. Res. 215, 119–130 (1994)
Cuff, C.A., Kothapalli, D., Azonobi, I., Chun, S., Zhan, Y., Belkin, R., Yeh, C., Secreto, A., Assoian, R.K., Rader, D.J., Puré, E.: The adhesion receptor CD44 promotes atherosclerosis by mediating inflammatory cell recruitment and vascular cell activation. J. Clin. Invest. 108, 1031–1040 (2001)
Garg, H.G., Joseph, P.A.M., Thompson, B.T., Hales, C.A., Toida, T., Imanari, T., Capila, I., Linhardt, R.J.: Effect of fully sulfated glycosaminoglycans on pulmonary artery smooth muscle cell proliferation. Arch. Biochem. Biophys. 371, 228–233 (1999)
Nagahata, M., Tsuchiya, T., Ishiguro, T., Matsuda, N., Nakatsuchi, Y., Teramoto, A., Hachimori, A., Abe, K.: A novel function of N-cadherin and Connexin43: marked enhancement of alkaline phosphatase activity in rat calvarial osteoblast exposed to sulfated hyaluronan. Biochem. Biophys. Res. Commun. 315, 603–611 (2004)
Magnani, A., Albanese, A., Lamponi, S., Barbucci, R.: Blood-interaction performance of differentially sulphated hyaluronic acids. Thromb. Res. 81, 383–395 (1996)
Isoyama, T., Thwaites, D., Selzer, M.G., Carey, R.I., Barbucci, R., Lokeshwar, V.B.: Differential selectivity of hyaluronidase inhibitors toward acidic and basic hyaluronidases. Glycobiology 16, 11–21 (2006)
Bothner, H., Waaler, T., Wik, O.: Limiting viscosity number and weight average molecular weight of hyaluronate samples produced by heat degradation. Int. J. Biol. Macromol. 10, 287–291 (1988)
Magnani, A., Lamponi, S., Rappuoli, R., Barbucci, R.: Sulphated hyaluronic acids: a chemical and biological characterisation. Polym. Int. 46, 225–240 (1998)
Barbucci, R., Magnani, A., Casolaro, M., Marchettini, N., Rossi, C., Bosco, M.: Modification of hyaluronic acid by insertion of sulphate groups to obtain a heparin-like molecule. Part I. Characterisation and behaviour in aqueous solution towards H+ and Cu2+ ions. Gazz. Chim. Ital. 125, 169–180 (1995)
Bociek, S.M., Darke, A.H., Welti, D., Rees, D.A.: The 13C-NMR spectra of hyaluronate and chondroitin sulphates. Further evidence on an alkali-induced conformation change. Eur. J. Biochem. 109, 447–456 (1980)
Naji, L., Kaufmann, J., Huster, D., Schiller, J., Arnold, K.: 13C NMR relaxation studies on cartilage and cartilage components. Carbohydr. Res. 327, 439–446 (2000)
Geissler, U., Hempel, U., Wolf, C., Scharnweber, D., Worch, H., Wenzel, K.: Collagen type I-coating of Ti6A14V promotes adhesion of osteoblasts. J. Biomed. Mater. Res. 51, 752–760 (2000)
Hempel, U., Reinstorf, A., Poppe, M., Fischer, U., Gelinsky, M., Pompe, W., Wenzel, K.W.: Proliferation and differentiation of osteoblasts on biocement D modified with collagen type I and citric acid. J. Biomed. Mater. Res. B Appl. Biomater. 71, 130–143 (2004)
Jackson, R.A., Murali, S., van Wijnen, A.J., Stein, G.S., Nurcombe, V., Cool, S.M.: Heparan sulfate regulates the anabolic activity of MC3T3-E1 preosteoblast cells by induction of Runx2. J. Cell. Physiol. 210, 38–50 (2007)
Huang, L., Cheng, Y.Y., Koo, P.L., Lee, K.M., Qin, L., Cheng, J.C.Y., Kumta, S.M.: The effect of hyaluronan on osteoblast proliferation and differentiation in rat calvarial-derived cell cultures. J. Biomed. Mater. Res. 66A, 880–884 (2003)
Kothapalli, D., Zhao, L., Hawthorne, E.A., Cheng, Y., Lee, E., Puré, E., Assoian, R.K.: Hyaluronan and CD44 antagonize mitogen-dependent cyclin D1 expression in mesenchymal cells. J. Cell. Biol. 176, 535–544 (2007)
Kothapalli, D., Flowers, J., Xu, T., Puré, E., Assoian, R.K.: Differential activation of ERK and Rac mediates the proliferative and anti-proliferative effects of hyaluronan and CD44. J. Biol. Chem. 283, 31823–31829 (2008)
Pietilä, K., Nikkari, T.: Acidic glycosaminoglycans inhibit the uptake of tritiated thymidine by rabbit aortic smooth muscle cells in culture. Med. Biol. 60, 34–37 (1982)
Acknowledgement
The authors thank the Deutsche Forschungsgemeinschaft for financial support (grant HE2763/5-1 & SCHN1099/1-1 & TR67), and Christine Kupke and Carolin Preissler for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kunze, R., Rösler, M., Möller, S. et al. Sulfated hyaluronan derivatives reduce the proliferation rate of primary rat calvarial osteoblasts. Glycoconj J 27, 151–158 (2010). https://doi.org/10.1007/s10719-009-9270-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-009-9270-9