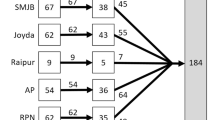
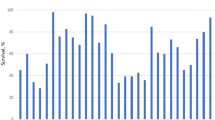
Abstract
Chinook salmon, Oncorhynchus tshawytscha, exhibit alternative reproductive tactics (ARTs) where males exist in two phenotypes: large “hooknose” males and smaller “jacks” that reach sexual maturity after only 1 year in seawater. The mechanisms that determine “jacking rate”—the rate at which males precociously sexually mature—are known to involve both genetics and differential growth rates, where individuals that become jacks exhibit higher growth earlier in life. The additive genetic components have been studied and it is known that jack sires produce significantly more jack offspring than hooknose sires, and vice versa. The current study was the first to investigate both additive and non-additive genetic components underlying jacking through the use of a full-factorial breeding design using all hooknose sires. The effect of dams and sires descendant from a marker-assisted broodstock program that identified “high performance” and “low performance” lines using growth- and survival-related gene markers was also studied. Finally, the relative growth of jack, hooknose, and female offspring was examined. No significant dam, sire, or interaction effects were observed in this study, and the maternal, additive, and non-additive components underlying jacking were small. Differences in jacking rates in this study were determined by dam performance line, where dams that originated from the low performance line produced significantly more jacks. Jack offspring in this study had a significantly larger body size than both hooknose males and females starting 1 year post-fertilization. This study provides novel information regarding the genetic architecture underlying ARTs in Chinook salmon that could have implications for the aquaculture industry, where jacks are not favoured due to their small body size and poor flesh quality.


Similar content being viewed by others
References
Aubin-Horth N, Bourque JF, Daigle G, Hedger R, Dodson JJ (2006) Longitudinal gradients in threshold sizes for alternative male life history tactics in a population of Atlantic salmon (Salmo salar). Can J Fish Aquat Sci 63:2067–2075
Bates D, Maechler M, Bolker B (2009) lme4: linear mixed-effects model using S4 classes. R packaged version 0.999375-31
Berejikian BA, Tezak EP, LaRae A (2000) Female mate choice and spawning behavior of Chinook salmon (Oncorhynchus tshawytscha) under experimental conditions. J Fish Biol 57:647–661
Berejikian BA, Van Doornik DM, Endicott RC, Hoffnagle TL, Tezak EP, Moore ME, Atkins J (2010) Mating success of alternative male phenotypes and evidence for frequency-dependent selection in Chinook salmon, Oncorhynchus tshawytscha. Can J Fish Aquat Sci 67:1933–1941
Berejikian BA, Van Doornik DM, Atkins JJ (2011) Alternative male reproductive phenotypes affect offspring growth rates in Chinook salmon. Trans Am Fish Soc 140:1206–1212
Berejikian BA, Bush RA, Campbell LA (2014) Maternal control over offspring life history in a partially anadromous species, Oncorhynchus mykiss. Trans Am Fish Soc 143:369–379
Brantley RK, Bass AH (1994) Alternative male spawning tactics and acoustic signals in the plainfin midshipman fish Porichthys notatus Girard (Teleostei, Batrachoididae). Ethology 96(3):213–232
Bryden CA, Heath JW, Heath DD (2004) Performance and heterosis in farmed and wild Chinook salmon (Oncorhynchus tshawyacha) hybrid and purebred crosses. Aquaculture 235:249–261
Butts IAE, Love OP, Farwell M, Pitcher TE (2012) Primary and secondary sexual characters in alternative reproductive tactics of Chinook salmon: associations with androgens and the maturation-inducing steroid. Gen Comp Endocrinol 175:449–456
Chernoff E, Curry RA (2007) First summer growth predetermined in anadromous and resident brook charr. J Fish Biol 70:334–346
Docker MF, Heath DD (2002) PCR-based markers detect genetic variation at growth and immune function-related loci in chinook salmon (Oncorhynchus tshawytscha). Mol Ecol Notes 2:606–609
Eilertsen EM, Bardsen BJ, Liljedal GR, Folstad I (2009) Experimental evidence for paternal effects on offspring growth rate in Arctic charr (Salvelinus alpinus). Proc R Soc B Biol Sci 276:129–136
Einum S, Fleming IA (1999) Maternal effects of egg size in brown trout (Salmo trutta): norms of reaction to environmental quality. R Soc Lond B Biol Sci 266:2095–2100
Evans ML, Neff BD, Heath DD (2010) Quantitative genetic and translocation experiments reveal genotype-by-environment effects on juvenile life-history traits in two populations of Chinook salmon (Oncorhynchus tshawytscha). J Evol Biol 23:687–698
Falica BK, Higgs DM (2013) Paternal genetic effects on offspring swimming performance vary with age of juvenile Chinook salmon. Evol Biol 40:355–365
Flannery EW, Butts IAE, Slowinska M, Ciereszko A, Pitcher TE (2013) Reproductive investment patterns, sperm characteristics, and seminal plasma physiology in alternative reproductive tactics of Chinook salmon (Oncorhynchus tshawytscha). Biol J Linn Soc 108:99–108
Gall GAE (1974) Influence of size of eggs and age of female on hatchability and growth in rainbow trout. Calif Fish Game 60:26–35
Garant D, Dodson JJ, Bernatchez L (2003) Differential reproductive success and heritability of alternative reproductive tactics in wild Atlantic salmon (Salmo salar L). Evolution 57(5):1133–1141
Gilbey J, Cauwelier E, Jones CS, Mclay A, Noble R, Verspoor E (2009) Size- dependent growth of individual Atlantic salmon Salmo salar alevins from hatch to first feeding. J Fish Biol 75:2820–2831
Gross M (1991a) Evolution of alternative reproductive strategies: frequency-dependent sexual selection in male bluegill sunfish. Philos Trans R Soc Lond B Biol Sci 332:59–66
Gross M (1991b) Salmon breeding behavior and life history evolution in changing environments. Ecology 72(4):1180–1186
Gross M (1994) Breeding competition in a pacific salmon (coho, Oncorhynchus kisutch)—measures of natural and sexual selection. Evolution 48:637–657
Gross M (1996) Alternative reproductive strategies and tactics: diversity within sexes. TREE 11(2):92–98
Healey MC, Prince A (1998) Alternative tactics in the breeding behaviour of male coho salmon. Behaviour 135(8):1099–1124
Heath DD (1992) Genetic, environmental, and physiological factors involved in the precocious sexual maturation of chinook salmon (Oncorhynchus tshawytscha). Ph.D. Thesis. University of British Columbia, Canada
Heath DD, Heath JW, Iwama GK (1991) Maturation in chinook salmon, Oncorhynchus tshawytscha (Walbaum): early identification based on the development of a bimodal weight-frequency distribution. J Fish Biol 39:565–575
Heath DD, Devlin RH, Heath JW, Iwama GK (1994) Genetic, environmental and interaction effects on the incidence of jacking in Oncorhynchus tshawytscha (Chinook salmon). Heredity 72:146–154
Heath DD, Devlin RH, Heath JW, Sweeting RM, McKeown BA, Iwama GK (1996) Growth and hormonal changes associated with precocious sexual maturation in male chinook salmon (Oncorhynchus tshawytscha (Walbaum)). J Exp Mar Biol Ecol 208:239–250
Heath DD, Fox CW, Heath JW (1999) Maternal effects on offspring size: variation through early development of Chinook salmon. Evolution 53(5):1605–1611
Heath DD, Rankin L, Bryden CA, Heath JW, Shrimpton JM (2002) Heritability and Y-chromosome influence male life history of Chinook salmon (Oncorhynchus tshawytscha). Heredity 89:311–317
Higgins PJ (1985) Metabolic differences between Atlantic salmon (Salmo salar) parr and smolts. Aquaculture 45:33–53
Houde ALS, Wilson CC, Neff BD (2013) Genetic architecture of survival and fitness-related traits in two populations of Atlantic salmon. Heredity 111:513–519
Hutchings J, Myers R (1994) The evolution of alternative mating strategies in variable environments. Evol Ecol 8:256–268
Johnson SL, Brockmann J (2013) Parental effects on early development: testing for indirect benefits of polyandry. Behav Ecol 24(5):1218–1228
Kause A, Ritola O, Paananen T, Mantysaari E, Eskelinen U (2003) Selection against early maturity in large rainbow trout Oncorhynchus mykiss: the quantitative genetics of sexual dimorphism and genotype-by-environment interactions. Aquaculture 228:53–68
Larsen DA, Beckman BR, Cooper KA, Barrett D, Johnston M, Swanson P, Dickoff WW (2004) Assessment of high rates of precocious male maturation in a spring Chinook salmon supplementation hatchery program. Trans Am Fish Soc 33:98–120
Lehnert SJ, Love OP, Pitcher TE, Higgs DM, Heath DD (2014) Multigenerational outbreeding effects in Chinook salmon (Oncorhynchus tshawytscha). Genetica 142:281–293
Lister JS, Neff BD (2006) Paternal genetic effects on foraging decision-making under the risk of predation. Ethology 112:963–970
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer Associates, Massachusetts
Magnhagen C (1998) Alternative reproductive tactics and courtship in the common goby. J Fish Biol 53:130–137
Martyniuk CJ, Perry GML, Mogahadam HK, Ferguson MM, Danzmann RG (2003) The genetic architecture of correlations among growth-related traits and male age at maturation in rainbow trout. J Fish Biol 63:746–764
Metcalfe NB (1998) The interaction between behavior and physiology in determining life history patterns in Atlantic salmon (Salmo salar). Can J Fish Aquat Sci 55(1):93–103
Nakagawa S, Schielzeth H (2010) Repeatability for Gaussian and non-Gaussian data: a practical guide for biologists. Biol Rev 85:935–956
Neff BD, Pitcher TE (2005) Genetic quality and sexual selection: an integrated framework for good genes and compatible genes. Mol Ecol 14:19–38
Oliveira RF, Canario AVM, Grober MS (2001) Male sexual polymorphism, alternative reproductive tactics, and androgens in combtooth blennies (Pisces: Bleniidae). Horm Behav 40:266–275
Pitcher TE, Neff BD (2006) MHC class IIB alleles contribute to both additive and nonadditive genetic effects on survival in Chinook salmon. Mol Ecol 15:2357–2365
Pitman RW (1979) Effects of female age and egg size on growth and mortality in rainbow trout. Progress Fish Cult 41(4):202–204
R Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Railsback SF, Rose KA (1999) Bioenergetics modeling of stream trout growth: temperature and food consumption effects. Trans Am Fish Soc 128:241–256
Sato T, Hirose M, Taborsky M, Kimura S (2004) Size-dependent male alternative reproductive tactics in the shell-brooding cichlid fish Lamprologus callipterus in Lake Tanganyika. Ethology 110:49–62
Semsar K, Kandel FLM, Godwin J (2001) Manipulations of the AVT system shift social status and related courtship and aggressive behavior in the bluehead wrasse. Horm Behav 40:21–31
Shrimpton JM, Heath JW, Devlin RH, Heath DD (2012) Effect of triploidy on growth and ionoregulatory performance in ocean-type Chinook salmon: A quantitative genetics approach. Aquaculture 362:248–254
Silverstein JT, Hershberger WK (1992) Precocious maturation in Coho salmon (Oncorhynchus kisutch): estimation of the additive genetic variance. J Hered 83(4):282–286
Sutterlin AM, MacLean D (1984) Age at first maturity and the early expression of oocyte recruitment processes in two forms of Atlantic salmon (Salmo salar) and their hybrids. Can J Fish Aquat Sci 41:1139–1149
Sutton SG, Bult TP, Haedrich RL (2000) Relationships among fat weight, body weight, water weight, and condition factors in wild Atlantic salmon parr. Trans Am Fish Soc 129:527–538
Taborsky M (2008) Alternative reproductive tactics in fish. In: Oliveira RF, Taborsky M, Brockmann JH (eds) Alternative reproductive tactics: an integrated approach. Cambridge University Press, New York, pp 1–21
Vainikka A, Huusko R, Hyvarinen P, Korhonen PK, Laaksonen T, Koskela J, Vielma J, Hirvonen H, Salminen M (2012) Food restriction prior to release reduces precocious maturity and improves migration tendency of Atlantic salmon (Salmo salar). Can J Fish Aquat Sci 69:1981–1993
Acknowledgments
The research was supported by the Natural Sciences and Engineering Research Council of Canada (Collaborative Research and Development Grant to TEP & DDH), Canada Foundation for Innovation (TEP), Ontario Research Fund (TEP), and Ontario Ministry of Innovation (TEP) and Early Researcher Award (TEP). We are grateful to John and Ann Heath all of the staff at Yellow Island Aquaculture Limited for assistance in the field.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Forest, A.R., Semeniuk, C.A.D., Heath, D.D. et al. Additive and non-additive genetic components of the jack male life history in Chinook salmon (Oncorhynchus tshawytscha). Genetica 144, 477–485 (2016). https://doi.org/10.1007/s10709-016-9917-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-016-9917-y