Abstract
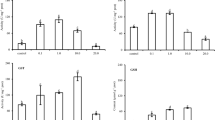
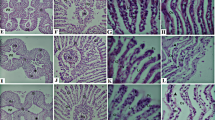
There were not any past studies about metallothionein isoforms (smtB and mt2) having anti-oxidative functions on zebrafish after Cd2+ exposure. On the other hand, the anti-oxidative enzymatic factors such as superoxide dismutase (sod), glutathione peroxidase (gpx1a), and catalase (cat) are used as references to investigate whether the smtB and mt2 have anti-oxidative responses on the gills and brain of zebrafish after 1–6 h of 0 and 1.78 μM Cd2+ exposure. The anti-oxidative system such as sod, cat, and gpx1a mRNA expressions demonstrated a cascade response upon Cd2+-induced oxidative stress in the present study. Interestingly, the smtB mRNA expression levels increased by 3.2- to 6.1-fold, and mt2 raised by 4.1- to 11.3-fold in gills at 1 and 3 h after exposure to Cd2+, respectively. On the other hand, the smtB mRNA levels increased by 10.6- to 58.6-fold, but mt2 mRNA levels increased by 2.3- to 11.1-fold in brain at 1 and 3 h after exposure to Cd2+, respectively. In addition, both tissues showed increased apoptosis levels at 3 h, and recovery after 6 h of Cd2+ exposure. From the results, we suggest that both mt2 and smtB play a role in anti-oxidation responses within 6 h after exposure to Cd2+. In conclusion, the smtB mRNA levels have a higher response than mt2 in the brain, but both mRNA expressions appear to have a similar pattern in the gill. We suggest that smtB plays an important role to defend oxidative stress in the brain of adult zebrafish upon acute Cd2+ exposure.








Similar content being viewed by others
References
Alvarez RM, Morales AE, Sanz A (2005) Antioxidant defenses in fish: biotic and abiotic factors. Rev Fish Biol Fish 15:75–88
An KW, Kim NN, Shin HS, Kil GS, Choi CY (2010) Profiles of antioxidant gene expression and physiological changes by thermal and hypoosmotic stresses in black porgy (Acanthopagrus schlegeli). Comp Biochem Physiol 156A:262–268
Atli G, Canli M (2010) Response of antioxidant system of freshwater fish Oreochromis niloticus to acute and chronic metal (Cd, Cu, Cr, Zn, Fe) exposures. Ecotoxicol Environ Saf 73:1884–1889
Bagnyukova TV, Lushchak OV, Storey KB, Lushchak VI (2007) Oxidative stress and antioxidant defense responses by goldfish tissues to acute changes of temperature from 3 to 23 °C. J Therm Biol 32:227–234
Barzilai A, Yamamoto KI (2004) DNA damage responses to oxidative stress. DNA Repair 3:1109–1115
Cao L, Huang W, Liu J, Yin X, Dou S (2010) Accumulation and oxidative stress biomarkers in Japanese flounder larvae and juveniles under chronic cadmium exposure. Comp Biochem Physiol 151C:386–392
Chen Y, Maret W (2001) Catalytic selenols couple the redox cycles of metallothionein and glutathione. Eur J Biochem 268:3346–3353
Chen YY, Zhu JY, Chan KM (2014) Effects of cadmium on cell proliferation, apoptosis, and proto-oncogene expression in zebrafish liver cells. Aquat Toxicol 157:196–206
Choi CY, An KW, Nelson ER, Habibi HR (2007) Cadmium affects the expression of metallothionein (MT) and glutathione peroxidase (GPX) mRNA in goldfish, Carassius auratus. Comp Biochem Physiol 145C:595–600
Choi JE, Kim J, Ahn JH, Youn P, Kang JS, Park K, Yi J, Ryu DY (2010) Induction of oxidative stress and apoptosis by silver nanoparticles in the liver of adult zebrafish. Aquat Toxicol 100:151–159
Crawshaw L, Grahn D, Wollmuth L, Simpson L (1985) Central nervous regulation of body temperature in vertebrates: comparative aspects. Pharmacol Ther 30:19–30
Cuypers A, Plusquin M, Remans T, Jozefczak M, Keunen E, Gielen H (2010) Cadmium stress: an oxidative challenge. Biometals 23:927–940
Firat O, Cogun HY, Aslanyavrusu S, Kargin F (2009) Antioxidant responses and metal accumulation in tissues of Nile tilapia Oreochromis niloticus under Zn, Cd and Zn+Cd exposures. J Appl Toxicol 29:295–301
Hsu T, Huang KM, Tsai HT, Sung ST, Ho TN (2013) Cadmium (Cd)-induced oxidative stress down-regulates the gene expression of DNA mismatch recognition proteins MutS homolog 2 (MSH2) and MSH6 in zebrafish (Danio rerio) embryos. Aquat Toxicol 126:9–16
Imada I, Sato EF, Miyamoto M, Ichimori Y, Minamiyama Y, Konaka R, Inoue M (1999) Analysis of reactive oxygen species generated by neutrophils using a chemiluminescence probe L-012. Anal Biochem 271:53–58
Kashiwagi A, Kashiwagi K, Takase M, Hanada H, Nakamura M (1997) Comparison of catalase in diploid and haploid Rana regosa using heat and chemical inactivation techniques. Comp Biochem Physiol 118B:499–503
Leonard SS, Harris GK, Shi X (2004) Metal-induced oxidative stress and signal transduction. Free Radic Biol Med 37:1921–1942
Liu CT, Chou MY, Lin CH, Wu SM (2012) Effects of ambient cadmium with calcium on mRNA expression of calcium uptake related transporters in zebrafish (Danio rerio) larvae. Fish Physiol Biochem 38:977–988
Porter NA, Mills KA, Caldwell SE (1995) Mechanisms of free radical oxidation of unsaturated lipids. Lipids 30:277–290
Regoli F, Gorbi S, Frenzilli G, Nigro M, Corsi I, Focardi S, Winston GW (2002) Oxidative stress in ecotoxicology: from the analysis of individual antioxidants to a more integrated approach. Mar Environ Res 54:419–423
Roesijadi G (1992) Metallothioneins in metal regulation and toxicity in aquatic animals. Aquat Toxicol 22:81–114
Sato M, Bremner I (1993) Oxygen free radicals and metallothionein. Free Radic Biol Med 14:325–337
Sellin MK, Eidem TM, Kolok AS (2007) Cadmium exposures in fathead minnows: Are there sex-specific differences in mortality, reproductive success, and Cd accumulation? Arch Environ Contam Toxicol 52:535–540
Shimoda R, Achanzar WE, Qu W, Nagamine T, Takagi H, Mori M, Waalkes MP (2003) Metallothionein is a potential negative regulator of apoptosis. Toxicol Sci 73:294–300
Tang R, Dodd A, Lai D, Mcnabb WC, Love DR (2007) Validation of zebrafish (Danio rerio) reference genes for quantitative real-time RT-PCR normalization. Acta Biochem Biophys Sin 39:384–390
Taylor EW, Beaumont MW, Butler PJ, Mair J, Mujallid MSI (1996) Lethal and sublethal effects of copper upon fish: a role for ammonia toxicity. In: Taylor EW (ed) Toxicology of aquatic pollution: physiological, cellular and molecular approaches. Cambridge University Press, Cambridge, pp 85–113
Tseng YC, Chen RD, Lucassen M, Schmidt MM, Dringen R, Abele D, Hwang PP (2011) Exploring upcoupling proteins and antioxidant mechanisms under acute cold exposure in brain of fish. PLoS One 6:e18180
Valavanidis A, Vlahogianni T, Dassenakis M, Scoullos M (2006) Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol Environ Saf 64:178–189
Wendel A (1980) Glutathione peroxidase. In: Jakoby WB (ed) Enzymatic basis of detoxification, vol 1. Academic Press, New York, pp 333–353
Wu SM, Jong KJ, Lee YJ (2006) Relationships among metallothionein, cadmium accumulation, and cadmium tolerance in three species of fish. Bull Environ Contam Toxicol 76:595–600
Wu SM, Shih MJ, Ho YC (2007) Toxicological stress response and cadmium distribution in hybrid tilapia (Oreochromis sp.) upon cadmium exposure. Comp Biochem Physiol 145C:218–226
Wu SM, Zheng YD, Kuo CH (2008a) Expression of mt2 and smt-B upon cadmium exposure and cold shock in zebrafish (Danio rerio). Comp Biochem Physiol 148C:184–193
Wu SM, Ding HR, Lin LY, Lin YS (2008b) Juvenile tilapia (Oreochromis mossambicus) strive to maintain physiological functions after waterborne copper exposure. Arch Environ Contam Toxicol 54:482–492
Wu SM, Tsai PR, Yan CJ (2012) Maternal cadmium exposure induces mt2 and smtB mRNA expression in zebrafish (Danio rerio) females and their offspring. Comp Biochem Physiol 156C:1–6
Wu SM, Liu JH, Shu LH, Chen CH (2015) Anti-oxidative responses of zebrafish (Danio rerio) gill, liver and brain tissues upon acute cold shock. Comp Biochem Physiol 187A:202–213
Acknowledgments
This study was financially supported by the National Science Council, Taiwan (NSC101-2313-B-415-003-MY3).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, S.M., Shu, LH. & Liu, JH. Anti-oxidative functions of mt2 and smtB mRNA expression in the gills and brain of zebrafish (Danio rerio) upon cadmium exposure. Fish Physiol Biochem 42, 1709–1720 (2016). https://doi.org/10.1007/s10695-016-0251-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-016-0251-1