Abstract
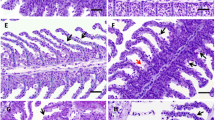
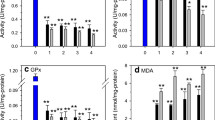
The organophosphate insecticide diazinon is widely used to control pest in Iran. The purpose of the present study was to investigate the antioxidant and histopathological changes in the liver tissue of rainbow trout (Oncorhynchus mykiss) exposed to 0.1 and 0.2 mg/L of a commercial formula of diazinon for a period of 28 days. Antioxidant enzyme activities—catalase, superoxide dismutase, glutathione peroxidase and glutathione reductase—were determined after 7, 14 and 28 days of exposure. Histopathological analyses were performed at the 28th day. All antioxidant enzymes were induced after 7 days of diazinon treatment in both concentrations of diazinon. Catalase and superoxide dismutase maintained elevated activities during all the treatment period. Glutathione peroxidase activity returned to the control values at the 14th day, decreasing to values below control at the 28th day in both diazinon concentrations. Glutathione reductase maintained increased activities at the 14th day in the 0.1 mg/L diazinon, decreasing to control values at the 28th day. In the 0.2 mg/L group, the activity returned to control values at the 14th and decreased below the control at the 28th day. Total antioxidant capacity of hepatocytes significantly decreased in fishes exposed to diazinon during all experimental periods. Hypertrophy of hepatocytes, vacuolization of cell cytoplasm and hepatocyte cloudy swelling were observed in the liver tissue of fish exposed to both concentrations of diazinon. The results showed that diazinon altered the activity of antioxidant enzymes and the cellular total antioxidant capacity inducing oxidative stress and cellular damage in hepatocytes evidenced by histopathological analysis.



Similar content being viewed by others
References
Achuba FI, Osakwe SA (2003) Petroleum-induced free radical toxicity in African catfish (Clarias gariepinus). Fish Physiol Biochem 29:97–103. doi:10.1023/B:FISH.000035905.14420.eb
Aebi H (1984) Catalase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press, New York, pp 671–684
Albanis T, Hela D, Sakellarides T, Konstantinou I (1998) Monitoring of pesticide residues and their metabolites in surface and underground waters of Imathia (N. Greece) by means of solid-phase extraction disks and gas chromatography. J Chromatogr A 823:59–71. doi:10.1016/S0021-9673(98)00304-5
Allender WJ, Britt AG (1994) Analysis of liquid diazinon formulations and breakdown products: an Australia-wide survey. Bull Environ Contam Toxicol 53:902–906. doi:10.1007/BF00196222
Arjmandi R, Tavakol M, Shayeghi M (2010) Determination of organophosphorus insecticide residues in the rice paddies. Int J Environ Sci Technol 7(1):175–182
Aydın R, Köprücü K (2005) Acute toxicity of diazinon on the common carp (Cyprinus carpio L.) embryos and larvae. Pestic Biochem Physiol 82:220–225. doi:10.1016/j.pestbp.2005.03.001
Bagheri F (2007) Study of pesticide residues (Diazinon, Azinphosmethyl) in the rivers of Golestan province (Gorgan Roud and Gharehsou), M.Sc. thesis, Tehran University of Medical Science. Tehran, Iran, pp 1–125
Bailey H, Deanovic L, Reyes E, Kimball T, Larson K, Cortright K, Connor V, Hinton D (2000) Diazinon and chlorpyrifos in urban waterways in northern California, USA. Environ Toxicol Chem 19:82–87. doi:10.1002/etc.5620190109
Banaee M (2012) Adverse effect of insecticides on various aspects of fish’s biology and physiology. In: Sonia Soloneski, Marcelo Larramendy (eds) Insecticides—basic and other applications book. InTech, Chapter 6, pp 101–126
Banaee M, Mirvagefei AR, Rafei GR, Majazi Amiri B (2008) Effect of sub-lethal diazinon concentrations on blood plasma biochemistry. Int J Environ Res 2:189–198
Banaee M, Sureda A, Mirvaghefi AR, Ahmadi K (2011) Effects of diazinon on biochemical parameters of blood in rainbow trout (Oncorhynchus mykiss). Pestic Biochem Physiol 99:1–6. doi:10.1016/j.pestbp.2010.09.001
Banks KE, Hunter DH, Wachal DJ (2005) Diazinon in surface waters before and after a federally-mandated ban. Sci Total Environ 350:86–93. doi:10.1016/j.scitotenv.2005.01.017
Box A, Sureda A, Galgani F, Pons A, Deudero S (2007) Assessment of environmental pollution at Balearic Islands applying oxidative stress biomarkers in the mussel Mytilus galloprovincialis. Comp Biochem Physiol C 146:531–539. doi:10.1016/j.cbpc.2007.06.006
Bulletin of Agriculture Ministry of Iran (2008) Annual report of performance of the Ministry of Agriculture of Iran (2006–2007). pp 1–301
Cattaneo R, Loro VL, Spanevello R, Silveira FA, Luz L, Miron DS, Fonseca MB, Moraes BS, Clasen B (2008) Metabolic and histological parameters of silver catfish (Rhamdia quelen) exposed to commercial formulation of 2,4-dichlorophenoxiacetic acid (2,4-D) herbicide. Pestic Biochem Physiol 92:133–137. doi:10.1016/j.pestbp.2008.07.004
Cengiz EI, Unlu E (2006) Sublethal effects of commercial deltamethrin on the structure of the gill, liver and gut tissues of mosquitofish, Gambusia affinis: a microscopic study. Environ Toxicol Pharmacol 21:246–253. doi:10.1016/j.etap.2005.08.005
Cong VN, Phuong NT, Bayley M (2008) Brain cholinesterase response in the snakehead fish (Channa striata) after field exposure to diazinon. Ecotoxicol Environ Saf 71:314–318. doi:10.1016/j.ecoenv.2008.04.005
de Menezes CC, Loro VL, da Fonseca MB, Cattaneo R, Pretto A, Miron DS, Santi A (2011) Oxidative parameters of Rhamdia quelen in response to commercial herbicide containing clomazone and recovery pattern. Pestic Biochem Physiol 100:145–150. doi:10.1016/j.pestbp.2011.03.002
Dubus I, Hollis J, Brown C (2000) Pesticides in rainfall in Europe. Environ Pollut 110:331–344 (PII: S0269-7491(99)00295-X)
U.S. EPA (2005) Aquatic life ambient water quality criteria Diazinon Final. Office of Science and Technology Washington, DC. (CAS Registry Number 333-41-5) 1-85
Ferreira D, da Motta AC, Kreutz LC, Toni C, Barcellos LJG, Loro VL (2010) Assessment of oxidative stress in Rhamdia quelen exposed to agrichemicals. Chemosphere 79:914–921. doi:10.1016/j.chemosphere.2010.03.024
Franco JL, Posser T, Mattos JJ, Sánchez-Chardi A, Trevisan R, Oliveira CS, Carvalho PSM, Leal RB, Marques MRF, Bainy ACD, Dafre AL (2008) Biochemical alterations in juvenile carp (Cyprinus carpio) exposed to zinc: glutathione reductase as a target. Mar Environ Res 66:88–89. doi:10.1016/j.marenvres.2008.02.031
Fujii Y, Asaka S (1982) Metabolism of diazinon and diazoxon in fish liver preparations. Bull Environ Contam Toxicol 29:455–460
Garcia S, Ake C, Clement B, Huebuer H, Donnelly K, Shalat S (2001) Initial results of environmental monitoring in the Texas Rio Grande valley. Environ Int 26:465–474. doi:10.1016/S0160-4120(01)00027-7
Ghassempour A, Mohammadkhah A, Najafie M, Rajabzadeh M (2002) Monitoring of the pesticide diazinon in soil, stem and surface water of rice fields. Anal Sci 18(7):779–783. doi:10.2116/analsci.18.779
Girón-Pérez MI, Santerre A, Gonzalez-Jaime F, Casas-Solis J, Hernández-Coronado M, Peregrina-Sandoval J, Takemura A, Zaitseva G (2007) Immunotoxicity and hepatic function evaluation in Nile tilapia (Oreochromis niloticus) exposed to diazinon. Fish Shellfish Immunol 23:760–769
Hai DQ, Varga SI, Matkovics B (1997) Organophosphate effects of antioxidant system of carp (Cyprinus carpio) and catfish (Ictalurus nebulosus). Comp Biochem Physiol 117:83–88 PMID: 9206589
Hamm JT, Wilson BW, Hinton DE (2001) Increasing uptake and bioactivation with development positively modulate diazinon toxicity in early life stage medaka (Oryzias latipes). Toxicol Sci 61:304–313 PMID: 11353139
Iris F, Benzie F, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of ‘‘antioxidant power’’: the FRAP assay. Anal Biochem 239:70–76
Isik I, Celik I (2008) Acute effects of methyl parathion and diazinon as inducers for oxidative stress on certain biomarkers in various tissues of rainbow trout (Oncorhynchus mykiss). Pestic Biochem Physiol 92:38–42. doi:10.1016/j.pestbp.2008.06.001
Jos A, Pichardo S, Prieto AI, Repetto G, Vazquez CM, Moreno I, Camean AM (2005) Toxic cyanobacterial cells containing microcystins induce oxidative stress in exposed tilapia fish (Oreochromis sp.) under laboratory conditions. Aquat Toxicol 72:261–271. doi:10.1016/j.aquatox.2005.01.003
Keizer J, D’Agostino G, Nagel R, Gramenzi F, Vittozzi L (1993) Comparative diazinon toxicity in guppy and zebra fish: different role of oxidative metabolism. Environ Toxicol Chem 12:1243–1250. doi:10.1002/etc.560120713
Keizer J, D’Agostino G, Nagel R, Volpe T, Gnemid P, Vittozzi L (1995) Enzymological differences of AChE and diazinon hepatic metabolism: correlation of in vitro data with the selective toxicity of diazinon to fish species. Sci Total Environ 171:213–220 (SSDI: 0048-9697(95) 04687-V)
Khara H, Salar Amoli J, Mazloumi H, Nezami SHA, Zolfinezhad K, Khodaparast SH, Hasan J, Akbarzadeh A, Mohammadi S, Gholipour S, Gholipour Z, Taghizadeh M (2008) Survey and seasonal measurement of pesticide (hinosan, machete and diazinon) in water of Oshmak River (east of Guilan). J Biol Sci 2(4):29–43
Khazaei SH (2007) Study of nitrate and diazinon residues in groundwater of Mahmoudabad area and verifying with one dimensional model. M.Sc. thesis, University of Tehran, Natural Resources Faculty, Department Fisheries and Environmental sciences, p 87
Khazaei SH, Khorasani N, Talebi KH, Ehteshami M (2010) Investigation of the groundwater contamination due to the use of diazinon insecticide in Mazandaran Province (Case study: Mahmoud Abad City). J Natur Environ (Iranian J Natur Resour) 63(1):23–32
Lartiges SB, Garrigues PP (1995) Degradation kinetics of organophosphorus and organonitrogen pesticides in different waters under various environmental conditions. Environ Sci Technol 29(5):1246–1254
Li PCH, Swanson EJ, Gobas FAPC (2002) Diazinon and its degradation products in agricultural water courses in British Columbia, Canada. Bull Environ Contam Toxicol 69:59–65. doi:10.1007/S00128-002-0010-0
Li ZH, Li P, Randak T (2010a) Ecotoxocological effects of short-term exposure to a human pharmaceutical Verapamil in juvenile rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol C 152:385–391. doi:10.1016/j.cbpc.2010.06.007
Li ZH, Zlabek V, Grabic R, Li P, Randak T (2010b) Modulation of glutathione-related antioxidant defense system of fish chronically treated by the fungicide propiconazole. Comp Biochem Physiol C 152:392–398. doi:10.1016/j.cbpc.2010.06.006
Li ZH, Zlabek V, Velíšek J, Grabic R, Machová J, Kolařová J, Li P, Randák T (2010c) Hepatic antioxidant status and hematological parameters in rainbow trout, Oncorhynchus mykiss, after chronic exposure to carbamazepine. Chem Biol Interact 183:98–104. doi:10.1016/j.cbi.2009.09.009
Li ZH, Zlabek V, Velíšek J, Grabic R, Machová J, Kolařová J, Li P, Randák T (2010d) Biochemical and physiological responses in liver and muscle of rainbow trout after long-term exposure to propiconazole. Ecotoxicol Environ Saf 73(2010):1391–1396. doi:10.1016/j.ecoenv.2010.05.017
Li ZH, Zlabek V, Velíšek J, Grabic R, Machová J, Kolařová J, Li P, Randák T (2010e) Chronic toxicity of verapamil on juvenile rainbow trout (Oncorhynchus mykiss): effects on morphological indices, hematological parameters and antioxidant responses. J Hazard Mater 185(2011):870–880. doi:10.1016/j.jhazmat.2010.09.102
Li ZH, Velisek J, Zlabek V, Grabic R, Machova J, Li P, Randak T (2011a) Chronic toxicity of verapamil on juvenile rainbow trout (Oncorhynchus mykiss): effects on morphological indices, hematological parameters and antioxidant responses. J Hazard Mater 185:870–880. doi:10.1016/j.jhazmat.2010.09.102
Li ZH, Zlabek V, Velíšek J, Grabic R, Machová J, Kolařová J, Li P, Randák T (2011b) Antioxidant responses and plasma biochemical characteristics in the freshwater rainbow trout, Oncorhynchus mykiss, after acute exposure to the fungicide propiconazole. Czech J Anim Sci 56(2):61–69
Luo Y, Su Y, Lin R, Shi H, Wang X (2006) 2-Chlorophenol induced ROS generation in fish Carassius auratus based on the EPR method. Chemosphere 65:1064–1073. doi:10.1016/j.chemosphere.2006.02.054
Mansour M, Feicht EA, Behechti A, Schramm K, Kettrup A (1999) Determination photo-stability of selected agrochemicals in water and soil. Chemosphere 39:575–585. doi:10.1016/S0045-6535(99)00123-X
Matos P, Fontaínhas-Fernandes A, Peixoto F, Carrola J, Rocha E (2007) Biochemical and histological hepatic changes of Nile tilapia Oreochromis niloticus exposed to carbaryl. Pestic Biochem Physiol 89:73–80. doi:10.1016/j.pestbp.2007.03.002
Mishra AK, Mohanty B (2008) Acute toxicity impacts of hexavalent chromium on behavior and histopathology of gill, kidney and liver of the freshwater fish, Channa punctatus (Bloch). Environ Toxicol Pharmacol 26:136–141. doi:10.1016/j.etap.2008.02.010
Mobasher M, Aramesh K, Aldavoud SJ, Ashrafganjooei N, Divsalar K, Phillips CJC, Larijani B (2008) Proposing a national ethical framework for animal research in Iran. Iranian J Publ Health 37(1):39–46
Monteiro DA, de Almeid JA, Rantin FT, Kalinin AL (2006) Oxidative stress biomarkers in the freshwater characid fish, Brycon cephalus, exposed to organophosphorus insecticide Folisuper 600 (methyl parathion). Comp Biochem Physiol C 143:141–149. doi:10.1016/j.cbpc.2006.01.004
Oruҫ EÖ, Usta D (2007) Evaluation of oxidative stress responses and neurotoxicity potential of diazinon in different tissues of Cyprinus carpio. Environ Toxicol Pharmacol 23:48–55. doi:10.1016/j.etap.2006.06.005
Ozmen I, Bayir A, Cengiz M, Sirkecioglu AN, Atamanalp M (2004) Effects of water reuse system on antioxidant enzymes of rainbow trout (Oncorhynchus mykiss W., 1792). Vet Med Czech 49(10):373–378
Parvez S, Pandey S, Ali M, Raisuddin S (2006) Biomarkers of oxidative stress in Wallago attu (Bl. and Sch.) during and after a fish-kill episode at Panipat, India. Sci Total Environ 368:627–636. doi:10.1016/j.scitotenv.2006.04.011
Peña-Llopis S, Ferrando MD, Peña JB (2003) Fish tolerance to organophosphate-induced oxidative stress is dependent on the glutathione metabolism and enhanced by N-acetylcysteine. Aquat Toxicol 65:337–360. doi:10.1016/S0166-445X(03)00148-6
Rahiminezhhad M, Shahtaheri SJ, Ganjalim R, Rahimi Foroushani A, Golbabaei F (2009) Moleculary imprinted solid phase extraction for trace analysis of diazinon in drinking water. IJEHSE 6:97–106
Robbins SL (2007) Liver, biliary tract, and pancreas. In: Cortan RS, Kumar V, Robbins SL (eds) Robbins pathologic basis of disease, 5th edn. Saunders, Philadelphia, pp 941–949
Schlenk D (2005) Pesticide biotransformation in fish. In: Mommsen TP, Moon TW (eds) Biochemistry and molecular biology of fishes, vol 6. Elsevier, Amsterdam, pp 171–190
Seguchi K, Asaka S (1981) Intake and excretion of diazinon in freshwater fishes. Bull Environ Contam Toxicol 27:244–249. doi:0007-4861/81/0027-0244
Sepici-Dinçel A, Cağlan Karasu Benli A, Selvi M, Sarıkaya R, Şahin D, Özkul IA, Erkoç F (2009) Sublethal cyfluthrin toxicity to carp (Cyprinus carpio L.) fingerlings: biochemical, hematological, histopathological alterations. Ecotoxicol Environ Saf 72:1433–1439. doi:10.1016/j.ecoenv.2009.01.008
Shayeghi M, Javadian EA (2001) Study of the residue of lindin and diazinon in the rice fields in Tonkabon City (Mazandaran Province) 1999. J Environ Sci Technol 9:51–58
Shayeghi M, Darabi H, Abtahi H, Sadeghi M, Pakbaz F, Golestaneh SR (2007) Assessment of persistence and residue of diazinon and malathion in three Rivers (Mond, Shahpour and Dalaky) of Bushehr province in 2004–2005 years. Iranian South Med J 10(1):54–60
Shayeghi M, Khoidel M, Bageri F, Abtahi M (2008) Azinphos methyl and diazinon residues in rivers of Qara-Su River and Gorgan-rud River in Golestan Province. J Publ Health Health Res Inst 6(1):75–82
Sureda A, Box A, Ensenat M, Alou E, Tauler P, Deudero S, Pons A (2006) Enzymatic antioxidant response of a labrid fish (Coris julis) liver to environmental caulerpenyne. Comp Biochem Physiol C 144:191–196. doi:10.1016/j.cbpc.2006.08.001
Sureda A, Box A, Deudero S, Pons A (2009) Reciprocal effects of caulerpenyne and intense herbivorism on the antioxidant response of Bittium reticulatum and Caulerpa taxifolia. Ecotoxicol Environ Saf 72:795–801. doi:10.1016/j.ecoenv.2007.12.007
Talebi K (1998) Diazinon residues in the basins of Anzali Lagoon, Iran. Bull Environ Contam Toxicol 61:477–483
Tarahi Tabrizi S (2001) Study of pesticide residues (diazinon, malathion, metasytoux) in the Tabriz Nahand River, M.Sc. thesis, Tehran University of Medical Science, Tehran, Iran, pp 1–88
Tavakol M (2007) Environmental impact assessment of diazinon in rice fields (a Case Study on Amol Township Rice Fields), M.Sc. thesis, Science and Research Branch, Islamic Azad University, Tehran, Iran, pp 1–95
Tejada S, Sureda A, Roca C, Gamundí A, Esteban S (2007) Antioxidant response and oxidative damage in brain cortex after high dose of pilocarpine. Brain Res Bullet 71:372–375. doi:10.1016/j.brainresbull.2006.10.005
Trenzado C, Hidalgo MC, García-Gallego M, Morales AE, Furné M, Domezain A, Domezain J, Sanz A (2006) Antioxidant enzymes and lipid peroxidation in sturgeon Acipenser naccarii and trout Oncorhynchus mykiss. A comparative study. Aquaculture 254:758–767. doi:10.1016/j.aquaculture.2005.11.020
Üner N, Oruç EÖ, Sevgiler Y, Şahin N, Durmaz H, Usta D (2006) Effects of diazinon on acetylcholinesterase activity and lipid peroxidation in the brain of Oreochromis niloticus. Environ Toxicol Pharmacol 21:241–245. doi:10.1016/j.etap.2005.08.007
van Dyk JC, Pieterse GM, van Vuren JHJ (2007) Histological changes in the liver of Oreochromis mossambicus (Cichlidae) after exposure to cadmium and zinc. Ecotoxicol Environ Saf 66:432–440. doi:10.1016/j.ecoenv.2005.10.012
Vinodhini R, Narayanan M (2009) Heavy metal induced histopathological alterations in selected organs of the Cyprinus carpio L. (Common Carp). Int J Environ Res 3(1):95–100
Wang TC, Hoffman ME (1991) Degradation of organophosphorus pesticides in coastal water. J Assoc Off Anal Chem 74(5):883–886
Zaruk D, Comba M, Struger J, Young S (2001) Comparison of immunoassay with a conventional method for the determination of Diazinon in surface waters. Anal Chim Acta 444:163–168 (PII: S0003-2670(01)01158-8)
Acknowledgments
The authors gratefully acknowledge the support offered from the Natural Resource Faculty, Tehran University. The authors are also grateful to Mr. Reza Ashori and Mrs. Maryam Mossavei, laboratory technicians, for their cooperation and assistance throughout the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banaee, M., Sureda, A., Mirvaghefi, A.R. et al. Biochemical and histological changes in the liver tissue of rainbow trout (Oncorhynchus mykiss) exposed to sub-lethal concentrations of diazinon. Fish Physiol Biochem 39, 489–501 (2013). https://doi.org/10.1007/s10695-012-9714-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-012-9714-1