Abstract
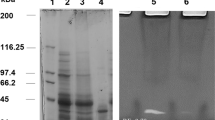
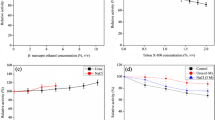
Trypsin from the viscera of Bogue (Boops boops) was purified to homogeneity by precipitation with ammonium sulphate, Sephadex G-100 gel filtration and Mono Q-Sepharose anion exchange chromatography, with an 8.5-fold increase in specific activity and 36% recovery. The molecular weight of the purified enzyme was estimated to be 23 kDa by SDS–PAGE and size exclusion chromatography. The purified trypsin appeared as a single band on native-PAGE and zymography staining. The purified enzyme showed esterase-specific activity on N-α-benzoyl-l-arginine ethyl ester (BAEE) and amidase activity on N-α-benzoyl-dl-arginine-p-nitroanilide (BAPNA). The optimum pH and temperature for the enzyme activity, after 10 min incubation, were pH 9.0 and 55°C, respectively, using BAPNA as a substrate. The trypsin kinetic constants K m and k cat on BAPNA were 0.13 mM and 1.56 s−1, respectively, while the catalytic efficiency k cat /K m was 12 s−1 mM−1. Biochemical characterisation of B. boops trypsin showed that this enzyme can be used as a possible biotechnological tool in the fish processing and food industries.






Similar content being viewed by others
References
Balti R, Barkia A, Bougatef A, Ktari N, Nasri M (2009) Heat-stable trypsin from cuttlefish (Sepia officinalis) hepatopancreas: purification and characterization. Food Chem 113:146–154
Ben Khaled H, Bougatef A, Balti R, Triki-Ellouz Y, Souissi N, Nasri M (2008) Isolation and characterization of trypsin from sardinelle (Sardinella aurita) viscera. J Sci Food Agric 88:2654–2662
Benjakul S, Visessanguan W, Thummaratwasik P (2000) Isolation and characterization of trypsin inhibitors from some Thai legume seeds. J. Food Biochem 24:107–127
Blanco RM, Guisan JM (1988) Protecting effects of competitive inhibitors during very intense insolubilized enzyme-activated support multipoint attachments: trypsin (amine)-agarose (aldehyde) system. Enzyme Microb Technol 10:227–232
Bougatef A, Souissi N, Fakhfakh N, Ellouz-Triki Y, Nasri M (2007) Purification and characterization of trypsin from the viscera of sardine (Sardina pilchardus). Food Chem 102:343–350
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Castillo-Yanez FJ, Pacheco-Aguilar R, Garcia-Carreno FL, Navarrete-Del T (2005) Isolation and characterization of trypsin from pyloric caeca of Monterey sardine Sardinops sagax caerulea. Comp Biochem Physiol 140B:91–98
Cherif M, Zarrad R, Gharbi H, Missaoui H, Jarboui O (2008) Length-weight relationships for 11 fish species from the Gulf of Tunis (SW Mediterranean Sea, Tunisia). Pan-Am J Aquat Sci 3:1–5
Cohen T, Gertler A (1981) Pancreatic proteolytic enzymes from Carp (Cyprinus carpio) I. Purification and physical properties of trypsin, chymosin, elastase and carboxypeptidase B. Comp Biochem Physiol B98:517–521
Erlanger BF, Kokowsky N, Cohen W (1961) The preparation and properties of two new chromogenic substrates of trypsin. Arch Biochem Biophys 95:271–278
Fuchise T, Kishimura H, Sekizaki H, Nonami Y, Kanno G, Klomklao S, Benjakul S, Chun BS (2009) Purification and characteristics of cold-zone fish trypsin. Food Chem 116:611–616
Gaamour A, Ben Abdallah L, Khemiri S, Mili S (2005) Study of the biology and the exploitation of small pelagics in Tunisia. MedSudMed 2003. Report of the MedSudMed Expert consultation on Marine Protected Areas and Fisheries Management. GCP/RER/010/ITA/MSM-TD-03, MedSudMed Technical Documents 5:56–74
Garcia-Carreno FL, Dimes LE, Haard NF (1993) Substrate-gel electrophoresis for composition and molecular weight of proteinases or proteinaceous proteinases inhibitors. Anal Biochem 214:65–69
Gates BJ, Travis J (1969) Isolation and comparative properties of shrimp trypsin. Biochemistry 8:4483–4489
Gupta R, Beg Q, Lorenz P (2002) Bacterial alkaline proteases: molecular approaches and industrial applications. Appl Microbiol Biotechnol 59:15–32
Haard NF (1998) Specialty enzymes from marine organisms. Food Technol 52:64–67
Jellouli K, Bougatef A, Daassi D, Barkia A, Nasri M (2009) New alkaline trypsin from intestine of Grey triggerfish (Balistes capriscus) with high activity at low temperature: isolation and characterization. Food Chem 116:644–650
Kishimura H, Hayashi K (2002) Isolation and characteristics of trypsin from pyloric ceca of the starfish Asterina pectinifera. Comp Biochem Physiol 132B:485–490
Kishimura H, Hayashi K, Miyashita Y, Nonami Y (2006) Characteristics of trypsins from the viscera of true sardine (Sardinops melanostictus) and the pyloric ceca of arabesque greenling (Pleuroprammus azonus). Food Chem 97:65–70
Kishimura H, Klomklao S, Benjakul S, Chun BS (2008) Characteristics of trypsin from the pyloric ceca of walleye pollock (Theragra chalcogramma). Food Chem 106:194–199
Klomklao S, Benjakul S, Visessanguan W, Kishimura H, Simpson BK, Saeki H (2006) Trypsins from yellowfin tuna (Thunnus albacores) spleen: purification and characterization. Comp Biochem Physiol 144B:47–56
Klomklao S, Benjakul S, Visessanguan W, Kishimura H, Simpson BK (2007) Purification and characterisation of trypsins from the spleen of skipjack tuna (Katsuwonus pelamis). Food Chem 100:1580–1589
Kurtovic I, Marshall SN, Simpson BK (2006) Isolation and characterization of a trypsin fraction from the pyloric ceca of chinook salmon (Oncorhynchus tshawytscha). Comp Biochem Physiol 143B:432–440
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:665–666
Martinez A, Olsen RL, Serra JL (1988) Purification and characterization of two trypsin-like enzymes from the digestive tract of anchovy Engraulis encrasicholus. Comp Biochem Physiol B91:677–684
North MJ (1982) Comparative biochemistry of the proteinases of eukaryotic microorganisms. Microbiol Rev 46:308–340
Rao MB, Tanksala AM, Ghatge MS, Deshpande VV (1998) Molecular and biotechnological aspects of microbial proteases. Microbiol Mol Biol Rev 62:597–635
Sekizaki H, Itoh K, Murakami M, Toyota E, Tanizawa K (2000) Anionic trypsin from chum salmon: activity with p-aminophenyl ester and comparison with bovine and Streptomyces griseus trypsins. Comp Biochem Physiol B127:337–346
Shahidi F, Kamil JYVA (2001) Enzymes from fish and aquatic invertebrates and their application in the food industry. Trends Food Sci Technol 12:435–464
Simpson BK (2000) Digestive proteinases from marine animals. In: Haard NF, Simpson BK (eds) Seafood enzymes: utilization and influence on postharvest seafood quality. Marcel Dekker, New York, pp 531–540
Zukowski MM (1992) Production of commercially valuable products. In: Doi RH, McGloughlin M (eds) biology of Bacilli: application to Industry. Butterworth-Heinemann, London, pp 311–337
Acknowledgments
This work was funded by «Ministry of Higher Education, Scientific Research and Technology-Tunisia».
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barkia, A., Bougatef, A., Nasri, R. et al. Trypsin from the viscera of Bogue (Boops boops): isolation and characterisation. Fish Physiol Biochem 36, 893–902 (2010). https://doi.org/10.1007/s10695-009-9365-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-009-9365-z