Abstract
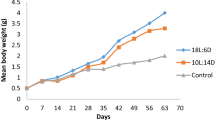
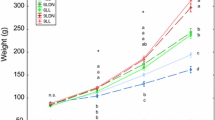
The effects of artificial photoperiod regimes on reproductive patterns have been studied in several species, as have haematological parameters. However, information on how artificial photoperiods may affect blood components is scarce, especially under field conditions. We have assessed the effects of constant light [long day (LD) photoperiod: 24 h (light):0 h (dark)] on haematological parameters of cultured rainbow trout in Chile (Southern Hemisphere). In the initial stage (March up to June), two groups of fish were maintained under similar conditions and under the ambient (natural) photoperiod (NP). One group was then placed under the LD photoperiod regime for 2 months (June/July), following which it was returned to␣the NP (August–January); the control group remained under the NP throughout the experiment (March–January). All fish were assessed for haematological parameters and growth performance at various times during the experiment. During the initial stage, no differences were found between groups. However, at the end of the LD 24:0 period, the LD fish had increased haematocrit values and erythrocyte numbers and diminished mean corpuscular haemoglobin. After the LD fish had been returned to the NP, they developed secondary sexual characteristics and had a 40–44% decrease in the number of leukocytes.
Similar content being viewed by others
References
Baggerman B (1980) Photoperiodic and endogenous control of the annual reproductive cycle in teleost fishes. In: Ali MA (eds) Environmental physiology of fishes. Plenum Press, New York, pp 553–556
Beagley KW, Gockel CM (2003) Regulation of innate and adaptive immunity by the female sex hormones oestradiol and progesterone. FEMS Immunol Med Microbiol 38:13–22
Bilbo SD, Dhabhar FS, Viswanathan K, Saul A, Yellon SM, Nelson RJ (2002) Short day lengths augment stress-induced leukocyte trafficking and stress-induced enhancement of skin immune function. Proc Natl Acad Sci USA 99:4067–4072
Bromage N, Porter M, Randall C (2001) The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 197:63–98
Bromage NR, Randall CF, Davies B, Porter M (1994) Photoperiod, melatonin and finfish reproduction. In high performance fish. Am Fish Soc 137–142
Biswas AK, Seoka M, Takii K, Maita M, Kumai H (2006) Stress response of red sea bream Pagrus major to␣acute handling and chronic photoperiod manipulation. Aquaculture (DOI:10.1016/j.aqu aculture. 2005.06.043) (available online 7 April 2006)
Bye V (1984) The role of environmental factors in the timing of reproductive cycles. In: Potts GW, Wiitton RJ (eds) Fish reproduction: strategies and tactics. Academic Press, London, pp 187–205
Campbell TW, Murru F (1990) An introduction to fish hematology. Comp Cont Ed Vet Sci 12:525–533
Carrillo M, Bromage N, Zanuy S, Prat F, Serrano R (1989) The effects of modifications in photoperiod on spawning time, ovarian development, and egg quality in the sea bass. Aquaculture 81:351–365
Collazos ME, Barriga C, Ortega E (1995) Seasonal variations in the immune system of the cyprinid Tinca tinca. Phagocytic function. Comp Immunol Microbiol Infect Dis 18:105–113
Collazos ME, Ortega E, Barriga C, Rodríguez B (1998) Seasonal variation in haematological parameters in male and female Tecn Tinca tinca. Mol Cell Biochem 114:432–435
De Vlaming VL (1972) Environmental control of teleost reproductive cycles; a brief review. J Fish Biol 4:131–140
Ellsaesser CF, Clem LW (1987) Cortisol-induced hematologic and immunologic changes in channel catfish (Ictalurus punctatus). Comp Biochem Physiol 87A:405–408
Espelid S, Lokken GB, Steiro K, Bogwald J (1996) Effects of cortisol and stress on the immune system in Atlantic salmon (Salmo salar). Fish Shellfish Immunol 6:95–110
Guijarro AI, Lopez-Patiño MA, Pinillos ML, Isorna E, De Pedro N, Alonzo-Gomez AL, Alonzo-Bedate M, Delgado MJ (2003) Seasonal changes in haematolgy and metabolic resources in the Tench. J Fish Biol 62:803–8015
Hemre GI, Taranger GL, Hansen T (2002) Gonadal development influences nutrient utilisation in cod (Gadus morhua). Aquaculture 214:201–209
Hoover E, Hubbard H (1937) Modification of the sexual cycle in trout by control of light. Copeia 4:206–210
Hoover EE (1937) Experimental modification of the sexual cycle in trout by control of light. Science 86:425–426
Johnson RW, Arkins S, Dantzer R, Kelley KW (1997) Hormones, lymphohemopoietic cytokines and the neuroimmune axis. Comp Biochem Physiol 116A:183–201
Kacem NH, Aldrin JF, Romestand B (1987) Immediate effects of a rapid temperature increase on some blood parameters of European sea bass Dicentrarchus labrax (Linne, 1758). Aquaculture 64:325–331
Kita J, Itazawa Y (1989) Release of erythrocytes from the spleen during exercise and splenic constriction by adrenaline infusion in the Rainbow trout. Jpn J Ichthyol 36:48–52
Kita J, Itazawa Y (1990) Effects of adrenaline on the blood flow through the spleen of rainbow trout (Salmo gairdneri). Comp Biochem Physiol 95A:591–595
Klontz GW (1994) Fish hematology. In: Stolen JD, Rowley AF, Zelikoff JT, Kaatari SL, Smith SA (eds) Techniques in fish immunology FITC 3. SOS Publications, Fair Haven, pp 121–131
Koger CS, The SJ, Hinton DE (1999) Variations of light and temperature regimes and resulting effects on reproductive parameters in Medaka (Oryzias latipes). Biol Reprod 61:1287–1293
Kumari J, Sahoo PK, Swain T, Sahoo SK, Sahu AK, Mohanty BR (2006) Seasonal variation in the innate immune parameters of the Asian catfish Clarias batrachus. Aquaculture 252:121–127
Leonardi M, Klempau A (2003) Artificial photoperiod influence on the immune system of juvenile rainbow trout (Oncorhynchus mykiss) in the Southern Hemisphere. Aquaculture 221:581–591
Leu SL, Shiah IS, Yatham LN, Cheu YM, Lam RW (2001) Immune-inflammatory markers in patients with seasonal affective disorder: effects of light therapy. J␣Affect Disord 63:27–34
Oppedal F, Juell JE, Taranger GL, Hansen T (2001) Artificial light and season affect vertical distribution and swimming behaviour of post-smolt Atlantic salmon in sea cages. J Fish Biol 58:1570–1584
Oppedal F, Taranger TL, Hansen T (2003) Growth performance and sexual maturation in diploid and triploid Atlantic salmon (Salmo salar L.) in seawater tanks exposed to continuous light or simulated natural photoperiod. Aquaculture 215:145–162
Pankhurst NW, Porter MJR (2003) Cold and dark or warm and light: variations on the theme of environmental control of reproduction. Fish Physiol Biochem 28:385–389
Pierson PM, Lamers A, Flik G, Mayer-Gostan N (2004) The stress axis, stanniocalcin, and ion balance in rainbow trout. Gen Comp Endocrinol 137:263–271
Porter MJR, Duncan NJ, Mitchell D, Bromage NR (1999) The use of cage lighting to reduce plasma melatonin in Atlantic salmon (Salmo salar) and its effects on the inhibition of grilsing. Aquaculture 176:237–244
Prendergast BJ, Nelson RJ, Zucker I (2002) Mammalian seasonal rhythms: behavior and neuroendocrine substrates. In: Ptaff DW (ed) Hormones, brain, and behavior. Academic Press, San Diego, pp 93–156
Randall D, Burggren W, French K (1998) Fisiología animal. McGraw-Hill/Interamericana de España, S.A.U. Madrid
Saha NL, Usami T, Suzuki Y (2004) In vitro effects of steroid hormones on IgM-secreting cells and IgM secretion in common carp (Cyprinus carpio). Fish Shellfish Immunol 17:149–158
Slater CH, Fitzpatrick MS, Schreck CB (1995) Characterization of an androgen receptor in salmonid lymphocytes: possible link to androgen-induced immunosuppression. Gen Comp Endocrinol 100:218–225
Takafumi Y, Watanuki H, Sakai M (2001) Effects of estradiol, progesterone and testosterone on the function of carp, Cyprinus carpio, phagocytes in vitro. Comp Biochem Physiol C 129:49–55
Valenzuela AE, Silva VM, Klempau AE (2006) Qualitative and quantitative effects of constant light photoperiod on rainbow trout (Oncorhynchus mykiss) peripheral blood erythrocytes. Aquaculture 251:596– 602
Vanecek J (1998) Cellular mechanisms of melatonin action. Physiol Rev 78:687–711
Wedemeyer GA, Barton BA, McLeay DJ (1990) Stress and acclimation. In: Schreck CB, Moyle PB (eds) Methods for fish biology. American Fisheries Society, Bethesda, Md., pp 451–489
Wira C, Fahey JV, Abrahams VM, Rossoll RM (2003) Influence of stage of the reproductive cycle and estradiol on thymus cell antigen presentation. J Steroid Biochem Mol Biol 84:79–87
Wojtaszek J, Dziewulska-Szwajkowska D, Lozinska-Gabska M, Adamowicz A, Dzugaj A (2002) Hematological effects of high dose of cortisol on the carp (Cyprinus carpio L.): cortisol effect on the carp blood. Gen Comp Endocrinol 125:176–183
Yellon SM, Fagoaga OR, Nehlsen-Cannarella SL (1999) Influence of photoperiod on immune cell functions in the male Siberian hamster. Am J Physiol 276:R97–R102
Zapata G, Varas A, Torroba M (1992) Seasonal variations in the immune system of lower vertebrates. Immunol Today 13:142–147
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valenzuela, A.E., Silva, V.M. & Klempau, A.E. Effects of constant light on haematological parameters of cultured rainbow trout (Oncorhynchus mykiss) in the Southern Hemisphere. Fish Physiol Biochem 32, 113–120 (2006). https://doi.org/10.1007/s10695-006-9103-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-006-9103-8