Abstract
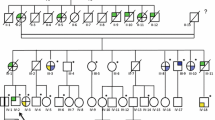
Hereditary leiomyomatosis and renal cell cancer (HLRCC) is a tumor predisposition syndrome caused by heterozygous germline mutations in the fumarate hydratase (FH) gene. Cutaneous and uterine leiomyomas are the most common clinical manifestations of HLRCC, whereas only approximately 20% of the families display renal cell cancer (RCC). The number of RCC cases in these families varies from one to five. Interestingly, families with multiple RCC cases are mainly found in Finland and the USA. Such aggregation of RCC in only some families and populations has led to the hypothesis that besides FH mutations also other inherited genetic and/or environmental factors may contribute to the malignant kidney tumor formation. To search for such a genetic modifier we have performed a genome-wide linkage analysis in two and an identical by descent analysis in four Finnish HLRCC families with several RCC patients. Additional Finnish and French families were used in fine-mapping and haplotype analyses. The only region compatible with linkage was the locus surrounding the FH gene itself in chromosome 1q43. The genes in the putative candidate region were screened, but no potentially pathogenic alterations were observed. Although these data do not rule out the existence of a genetic modifier, they emphasize the contribution of the FH genotype in HLRCC related RCC. Therefore, as all FH mutation carriers may have an increased risk for developing renal cancer, counseling and genetic testing should be offered for all HLRCC family members and clinical follow-up should be organized for the mutation carriers.


Similar content being viewed by others
Abbreviations
- CHRM3 :
-
Muscarinic acetylcholine receptor M3
- cM:
-
Centimorgan
- FH :
-
Fumarate hydratase (gene)
- FH:
-
Fumarase (enzyme)
- FMN2 :
-
Formin 2
- GREM2 :
-
Gremlin-2 precursor
- HIF:
-
Hypoxia-inducible factor
- HLRCC:
-
Hereditary leiomyomatosis and renal cell cancer
- IBD:
-
Identical by descent
- LOD:
-
Logarithm of odds
- Mb:
-
Mega base pairs
- MRI:
-
Magnetic resonance imaging
- RCC:
-
Renal cell cancer
- SDH:
-
Succinate dehydrogenase complex
- SNP:
-
Single nucleotide polymorphism
- TCAC:
-
Tricarboxylic acid cycle
References
Launonen V, Vierimaa O, Kiuru M et al (2001) Inherited susceptibility to uterine leiomyomas and renal cell cancer. Proc Natl Acad Sci USA 98(6):3387–3392
Alam NA, Bevan S, Churchman M et al (2001) Localization of a gene (MCUL1) for multiple cutaneous leiomyomata and uterine fibroids to chromosome 1q42.3–q43. Am J Hum Genet 68(5):1264–1269
Tomlinson IP, Alam NA, Rowan AJ et al (2002) Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat Genet 30(4):406–410
Lehtonen HJ, Kiuru M, Ylisaukko-Oja SK et al (2006) Increased risk of cancer in patients with fumarate hydratase germline mutation. J Med Genet 43(6):523–526
Merino MJ, TorresCabala C, Pinto P et al (2007) The morphologic spectrum of kidney tumors in hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome. Am J Surg Pathol 31(10):1578–1585
Kiuru M, Launonen V, Hietala M et al (2001) Familial cutaneous leiomyomatosis is a two-hit condition associated with renal cell cancer of characteristic histopathology. Am J Pathol 159(3):825–829
Alam NA, Rowan AJ, Wortham NC et al (2003) Genetic and functional analyses of FH mutations in multiple cutaneous and uterine leiomyomatosis, hereditary leiomyomatosis and renal cancer, and fumarate hydratase deficiency. Hum Mol Genet 12(11):1241–1252
Toro JR, Nickerson ML, Wei MH et al (2003) Mutations in the fumarate hydratase gene cause hereditary leiomyomatosis and renal cell cancer in families in North America. Am J Hum Genet 73(1):95–106
Alam NA, Olpin S, Rowan A et al (2005) Missense mutations in fumarate hydratase in multiple cutaneous and uterine leiomyomatosis and renal cell cancer. J Mol Diagn 7(4):437–443
Chan I, Wong T, Martinez-Mir A et al (2005) Familial multiple cutaneous and uterine leiomyomas associated with papillary renal cell cancer. Clin Exp Dermatol 30(1):75–78
Chuang GS, Martinez-Mir A, Geyer A et al (2005) Germline fumarate hydratase mutations and evidence for a founder mutation underlying multiple cutaneous and uterine leiomyomata. J Am Acad Dermatol 52(3 Pt 1):410–416
Chuang GS, Martinez-Mir A, Engler DE et al (2006) Multiple cutaneous and uterine leiomyomata resulting from missense mutations in the fumarate hydratase gene. Clin Exp Dermatol 31(1):118–121
Badeloe S, van Geel M, van Steensel MA et al (2006) Diffuse and segmental variants of cutaneous leiomyomatosis: novel mutations in the fumarate hydratase gene and review of the literature. Exp Dermatol 15(9):735–741
Wei MH, Toure O, Glenn GM et al (2006) Novel mutations in FH and expansion of the spectrum of phenotypes expressed in families with hereditary leiomyomatosis and renal cell cancer. J Med Genet 43(1):18–27
Refae MA, Wong N, Patenaude F et al (2007) Hereditary leiomyomatosis and renal cell cancer: an unusual and aggressive form of hereditary renal carcinoma. Nat Clin Pract Oncol 4(4):256–261
Alam NA, Olpin S, Leigh IM et al (2005) Fumarate hydratase mutations and predisposition to cutaneous leiomyomas, uterine leiomyomas and renal cancer. Br J Dermatol 153(1):11–17
Lehtonen HJ, Blanco I, Piulats JM et al (2007) Conventional renal cancer in a patient with fumarate hydratase mutation. Hum Pathol 38(5):793–796
Grubb RL 3rd, Franks ME, Toro J et al (2007) Hereditary leiomyomatosis and renal cell cancer: a syndrome associated with an aggressive form of inherited renal cancer. J Urol 177(6):2074–2079
Alam NA, Barclay E, Rowan AJ et al (2005) Clinical features of multiple cutaneous and uterine leiomyomatosis: an underdiagnosed tumor syndrome. Arch Dermatol 141(2):199–206
Bayley JP, Launonen V, Tomlinson IP et al (2008) The FH mutation database: an online database of fumarate hydratase mutations involved in the MCUL (HLRCC) tumor syndrome and congenital fumarase deficiency. BMC Med Genet 9:20
Ahvenainen T, Lehtonen HJ, Lehtonen R et al (2008) Mutation screening of fumarate hydratase by multiplex ligation-dependent probe amplification: detection of exonic deletion in a patient with leiomyomatosis and renal cell cancer. Cancer Genet Cytogenet 183(2):83–88
Gudbjartsson DF, Jonasson K, Frigge ML et al (2000) Allegro, a new computer program for multipoint linkage analysis. Nat Genet 25(1):12–13
Thiele H, Nurnberg P (2005) HaploPainter: a tool for drawing pedigrees with complex haplotypes. Bioinformatics 21(8):1730–1732
Sobel E, Lange K (1996) Descent graphs in pedigree analysis: applications to haplotyping, location scores, and marker-sharing statistics. Am J Hum Genet 58(6):1323–1337
Abecasis GR, Cherny SS, Cookson WO et al (2002) Merlin–rapid analysis of dense genetic maps using sparse gene flow trees. Nat Genet 30(1):97–101
Pollard PJ, Spencer-Dene B, Shukla D et al (2007) Targeted inactivation of fh1 causes proliferative renal cyst development and activation of the hypoxia pathway. Cancer Cell 11(4):311–319
Isaacs JS, Jung YJ, Mole DR et al (2005) HIF overexpression correlates with biallelic loss of fumarate hydratase in renal cancer: novel role of fumarate in regulation of HIF stability. Cancer Cell 8(2):143–153
Pollard PJ, Briere JJ, Alam NA et al (2005) Accumulation of Krebs cycle intermediates and over-expression of HIF1alpha in tumours which result from germline FH and SDH mutations. Hum Mol Genet 14(15):2231–2239
Selak MA, Armour SM, MacKenzie ED et al (2005) Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-alpha prolyl hydroxylase. Cancer Cell 7(1):77–85
Bayley JP, Devilee P, Taschner PE et al (2005) The SDH mutation database: an online resource for succinate dehydrogenase sequence variants involved in pheochromocytoma, paraganglioma and mitochondrial complex II deficiency. BMC Med Genet 6:39
Vanharanta S, Buchta M, McWhinney SR et al (2004) Early-onset renal cell carcinoma as a novel extraparaganglial component of SDHB-associated heritable paraganglioma. Am J Hum Genet 74(1):153–159
Neumann HP, Pawlu C, Peczkowska M et al (2004) Distinct clinical features of paraganglioma syndromes associated with SDHB and SDHD gene mutations. JAMA 292(8):943–951
Srirangalingam U, Walker L, Khoo B et al (2008) Clinical manifestations of familial paraganglioma and phaeochromocytomas in succinate dehydrogenase B (SDH-B) gene mutation carriers. Clin Endocrinol (Oxf) 69(4):587–596
Ricketts C, Woodward ER, Killick P et al (2008) Germline SDHB mutations and familial renal cell carcinoma. J Natl Cancer Inst 100(17):1260–1262
Acknowledgments
Sini Marttinen, Iina Vuoristo, Inga-Lill Svedberg, Mikko Aho, and Anniina Raitila are warmly thanked for excellent technical assistance. The study has been supported by grants from European Commission (LSHC-CT-2005-518200), Academy of Finland (Center of excellence in Translational Genome-Scale Biology and grants 212901, 214323, 213183, 214268), Finnish Cancer Society, the French National Cancer Institute (Kidney PNES, INCa) and the French League against Cancer (Comités du Cher et de l’Indre).
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors Pia Vahteristo and Taru A. Koski have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Vahteristo, P., Koski, T.A., Näätsaari, L. et al. No evidence for a genetic modifier for renal cell cancer risk in HLRCC syndrome. Familial Cancer 9, 245–251 (2010). https://doi.org/10.1007/s10689-009-9312-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-009-9312-2