Abstract
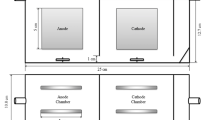
Two-chamber microbial fuel cells (MFCs) were used to study the applicability of MFCs for hexavalent chromium (Cr(VI)) detection in water. The microbial acetate oxidation in the anode and the Cr(VI) reduction in the cathode together generated voltages, which were used to indicate the change in Cr(VI) concentrations of the cathode under varying conditions of pH, ionic strength, co-existing Fe(II) concentration, and organic matter concentration. The MFC-based Cr(VI) detector showed a significant change in voltage with increasing Cr(VI) concentration at pH 1 and 2, but not at higher pH conditions. The detector also successfully measured the changes in Cr(VI) concentration at a range of ionic strength (i.e., 10–300 mM), and in the presence of different concentrations of fulvic acid (0–50 mg/L), which was used as a surrogate of organic matters, without interference. The Cr(VI) detection was not interfered by the presence of Fe(II) at the Cr(VI)/Fe(II) ratio of 1:1 and 1:15, but it was interfered at higher ratios (i.e., 1:164 and 1:848). The detector could measure the Cr(VI) concentration from 0.1 to 15 mg/L at pH 2. Overall, the MFC-based Cr(VI) detector may be applied to meet the growing need of real-time Cr(VI) monitoring in water.






Similar content being viewed by others
References
Ayyaru, S., & Dharmalingam, S. (2014). Enhanced response of microbial fuel cell using sulfonated poly ether ether ketone membrane as a biochemical oxygen demand sensor. Analytica Chimica Acta, 818, 15–22. doi:10.1016/j.aca.2014.01.059.
Balasubramanian, S., & Pugalenthi, V. (1999). Determination of total chromium in tannery waste water by inductively coupled plasma-atomic emission spectrometry, flame atomic absorption spectrometry and UV-visible spectrophotometric methods. Talanta, 50(3), 457–467. doi:10.1016/S0039-9140(99)00135-6.
Barceloux, D. G. (1999). Chromium. Journal of Toxicology. Clinical Toxicology, 37(2), 173–194, doi:10.1081/CLT-100102418.
Buerge, I. J., & Hug, S. J. (1997). Kinetics and pH dependence of chromium(VI) reduction by iron(II). Environmental Science and Technology, 31(5), 1426–1432. doi:10.1021/es960672i.
Cervantes, C., Campos-García, J., Devars, S., Gutiérrez-Corona, F., Loza-Tavera, H., Torres-Guzmán, J. C., et al. (2001). Interactions of chromium with microorganisms and plants. FEMS Microbiology Reviews, 25(3), 335–347. doi:10.1016/S0168-6445(01)00057-2.
Chang, I. S., Jang, J. K., Gil, G. C., Kim, M., Kim, H. J., Cho, B. W., et al. (2004). Continuous determination of biochemical oxygen demand using microbial fuel cell type biosensor. Biosensors and Bioelectronics, 19(6), 607–613. doi:10.1016/S0956-5663(03)00272-0.
Choi, S. (2015). Microscale microbial fuel cells: advances and challenges. Biosensors and Bioelectronics, 69, 8–25. doi:10.1016/j.bios.2015.02.021.
Di Lorenzo, M., Thomson, A. R., Schneider, K., Cameron, P. J., & Ieropoulos, I. (2014). A small-scale air-cathode microbial fuel cell for on-line monitoring of water quality. Biosensors and Bioelectronics, 62, 182–188. doi:10.1016/j.bios.2014.06.050.
Eckert, J. M., Stewart, J. J., Waite, T. D., Szymczak, R., & Williams, K. L. (1990). Reduction of chromium(VI) at sub-μg l−1 levels by fulvic acid. Analytica Chimica Acta, 236, 357–362. doi:10.1016/S0003-2670(00)83334-6.
Fukushima, M., Nakayasu, K., Tanaka, S., & Nakamura, H. (1997). Speciation analysis of chromium after reduction of chromium (VI) by humic acid. Toxicological and Environmental Chemistry, 62(1–4), 207–215.
Gangadharan, P., & Nambi, I. M. (2015). Hexavalent chromium reduction and energy recovery by using dual-chambered microbial fuel cell. Water Science and Technology, 71(3), 353–358. doi:10.2166/wst.2014.524.
Heijne, A. T., Liu, F., Weijden, R. V. D., Weijma, J., Buisman, C. J. N., & Hamelers, H. V. M. (2010). Copper recovery combined with electricity production in a microbial fuel cell. Environmental Science and Technology, 44(11), 4376–4381. doi:10.1021/es100526g.
Jenkins, V. L. S. D. (1980). Water chemistry. United States of America: Wiley.
Johnson, D. B. (2012). Geomicrobiology of extremely acidic subsurface environments. FEMS Microbiology Ecology, 81(1), 2–12. doi:10.1111/j.1574-6941.2011.01293.x.
Kotaś, J., & Stasicka, Z. (2000). Chromium occurrence in the environment and methods of its speciation. Environmental Pollution, 107(3), 263–283. doi:10.1016/S0269-7491(99)00168-2.
Kyziol, J., Twardowska, I., & Schmitt-Kopplin, P. (2006). The role of humic substances in chromium sorption onto natural organic matter (peat). Chemosphere, 63(11), 1974–1982. doi:10.1016/j.chemosphere.2005.09.042.
Li, Y., Yue, Q., Gao, B., Li, Q., & Li, C. (2008a). Adsorption thermodynamic and kinetic studies of dissolved chromium onto humic acids. Colloids and Surfaces B: Biointerfaces, 65(1), 25–29. doi:10.1016/j.colsurfb.2008.02.014.
Li, Z., Zhang, X., & Lei, L. (2008b). Electricity production during the treatment of real electroplating wastewater containing Cr6+ using microbial fuel cell. Process Biochemistry, 43(12), 1352–1358. doi:10.1016/j.procbio.2008.08.005.
Liu, H., Cheng, S., & Logan, B. E. (2005). Power generation in fed-batch microbial fuel cells as a function of ionic strength, temperature, and reactor configuration. Environmental Science and Technology, 39(14), 5488–5493. doi:10.1021/es050316c.
Osaki, S., Osaki, T., Hirashima, N., & Takashima, Y. (1983). The effect of organic matter and colloidal particles on the determination of chromium(VI) in natural waters. Talanta, 30(7), 523–526. doi:10.1016/0039-9140(83)80122-2.
Peiffer, S., Walton-Day, K., & Macalady, D. L. (1999). The interaction of natural organic matter with iron in a wetland (Tennessee Park, Colorado) receiving acid mine drainage. Aquatic Geochemistry, 5(2), 207–223. doi:10.1023/A:1009617925959.
Qafoku, N. P., Evan Dresel, P., Ilton, E., McKinley, J. P., & Resch, C. T. (2010). Chromium transport in an acidic waste contaminated subsurface medium: the role of reduction. Chemosphere, 81(11), 1492–1500. doi:10.1016/j.chemosphere.2010.08.043.
Remoundaki, E., Hatzikioseyian, A., & Tsezos, M. (2007). A systematic study of chromium solubility in the presence of organic matter: consequences for the treatment of chromium-containing wastewater. Journal of Chemical Technology and Biotechnology, 82(9), 802–808. doi:10.1002/jctb.1742.
Sedlak, D. L., & Chan, P. G. (1997). Reduction of hexavalent chromium by ferrous iron. Geochimica et Cosmochimica Acta, 61(11), 2185–2192.
Shanker, A. K., Cervantes, C., Loza-Tavera, H., & Avudainayagam, S. (2005). Chromium toxicity in plants. Environment International, 31(5), 739–753. doi:10.1016/j.envint.2005.02.003.
Stein, N. E., Hamelers, H. V. M., van Straten, G., & Keesman, K. J. (2012). Effect of toxic components on microbial fuel cell-polarization curves and estimation of the type of toxic inhibition. Biosensors, 2(3), 255–268. doi:10.3390/bios2030255.
Tandukar, M., Huber, S. J., Onodera, T., & Pavlostathis, S. G. (2009). Biological chromium(VI) reduction in the cathode of a microbial fuel cell. Environmental Science and Technology, 43(21), 8159–8165. doi:10.1021/es9014184.
Wang, H. Y., Bernarda, A., Huang, C. Y., Lee, D. J., & Chang, J. S. (2011). Micro-sized microbial fuel cell: A mini-review. Bioresource Technology, 102(1), 235–243. doi:10.1016/j.biortech.2010.07.007.
Wittbrodt, P. R., & Palmer, C. D. (1995). Reduction of Cr(VI) in the presence of excess soil fulvic acid. Environmental Science & Technology, 29(1), 255–263.
Zhang, Y., & Angelidaki, I. (2012). A simple and rapid method for monitoring dissolved oxygen in water with a submersible microbial fuel cell (SBMFC). Biosensors and Bioelectronics, 38(1), 189–194. doi:10.1016/j.bios.2012.05.032.
Zhang, B., Feng, C., Ni, J., Zhang, J., & Huang, W. (2012). Simultaneous reduction of vanadium (V) and chromium (VI) with enhanced energy recovery based on microbial fuel cell technology. Journal of Power Sources, 204, 34–39. doi:10.1016/j.jpowsour.2012.01.013.
Zhao, F., Harnisch, F., Schröder, U., Scholz, F., Bogdanoff, P., & Herrmann, I. (2006). Challenges and constraints of using oxygen cathodes in microbial fuel cells. Environmental Science and Technology, 40(17), 5193–5199. doi:10.1021/es060332p.
Acknowledgments
This research was mainly supported by Korea Ministry of Environment as GAIA (Geo-Advanced Innovative Action) project, and received an additional support by Basic Science Research Program through National Research Foundation of Korea (NRF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, H., Ju, W.J., Jho, E.H. et al. Applicability of a submersible microbial fuel cell for Cr(VI) detection in water. Environ Monit Assess 188, 613 (2016). https://doi.org/10.1007/s10661-016-5625-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-016-5625-4