Abstract
Analysis of survey data from 804 spring-oat fields divided into five climatic regions suggested that low inoculum level of Fusarium langsethiae was a major limiter of T-2 + HT-2. A 30-year climate with a cool and rainy 10-day period preceding estimated mid-anthesis and a warm 3-week period following anthesis were positively associated with T-2 + HT-2 contamination. In 12 survey years, warm weather from 4 weeks before GS65 until harvesting increased T-2 + HT-2, except in the 1–2 weeks preceding mid-anthesis, when the requirement for high humidity dominated. F. langsethiae and F. sporotrichioides were consistently promoted by high temperature from mid-anthesis onwards. A positive response of T-2 + HT-2 to high humidity peaked 5–10 days earlier and was shorter and weaker than the responses of DON and F. graminearum. The probability of high concentration and regional mean concentration of T-2 + HT-2 tended to be lower under ploughing than under non-ploughing, and they tended to increase with increasing cereal intensity. T-2 + HT-2 was positively associated with zero tillage when compared with minimum tillage. The responses of F. langsethiae and F. sporotrichioides to ploughing and cereal intensity varied by region. In contrast to T-2 + HT-2, DON was the same or higher under ploughing than under non-ploughing, not consistently affected by cereal intensity and not associated with zero tillage. F. graminearum was consistently more common under ploughing than under non-ploughing, and decreased with increasing cereal intensity in four regions. Also, F. culmorum and F. poae tended to be more common under ploughing. F. culmorum increased with cereal intensity in three regions.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
In Europe, Fusarium langsethiae Torp & Nirenberg (FL) is considered the primary producer of T-2 and HT-2 mycotoxins (T-2 + HT-2) in oat (Yli-Mattila et al. 2008; Fredlund et al. 2010; Edwards et al. 2012; Opoku et al. 2013; Imathiu et al. 2013; Hietaniemi et al. 2016; Hofgaard et al. 2016a; Schöneberg et al. 2018). F. sporotrichioides Sherb (FS) strains isolated from cereals in Europe are potent producers of T-2 + HT-2 in vitro (Langseth et al. 1998; Jestoi et al. 2008; Kokkonen et al. 2012a; Nazari et al. 2016), but they have not been shown to be important producers of T-2 + HT-2 in oat fields. Another important mycotoxin in European oat is deoxynivalenol (DON) with its derivatives, which in north-western Europe are primarily produced by F. graminearum Schwabe (FG) and to a lesser extent by F. culmorum (W.G.Smith) Sacc. (FC) (Yli-Mattila et al. 2008; Edwards et al. 2012; Nielsen et al. 2011; Fredlund et al. 2013; Hietaniemi et al. 2016; Hofgaard et al. 2016a). Nivalenol (NIV), currently less important, yet considered a notable mycotoxin, which is mainly produced by F. poae (Peck) Wollenw. (FP) (Pettersson et al. 1995; Yli-Mattila et al. 2008; Bernhoft et al. 2010; Fredlund et al. 2013; Martin et al. 2018; Schöneberg et al. 2018). The most comprehensive data on toxin contamination in cereals has been collected by the cereal industry, but is not publicly available. According to published survey results (Edwards 2009; Hietaniemi et al. 2016; Hofgaard et al. 2016a, Edwards 2017, Schöneberg et al. 2018), average T-2 + HT-2 contamination in Finnish spring oat (regional means over ten years 173–263 μg kg−1) does not seem to differ much from contamination observed in Switzerland, south-eastern Norway, Sweden, and in the UK. The contamination is higher in winter oat than in spring oat in Switzerland and the UK. In the UK, regional annual means have reached 400–800 μg kg−1 in winter oats.
Nazari et al. (2014) found in vitro that the growth of FL isolates from Italian durum wheat was possible between 10 and 35 °C. The optimal temperature range for infection of durum wheat spikes was 15–20 °C and for growth 20–30 °C, peaking at 24–25 °C. In experiments by Schöneberg et al. (2019) in Austria a lower optimum for infection of oat by FL was observed: inoculations at the beginning of anthesis (GS61) and at mid-anthesis (GS65) under 99% relative humidity (RH) at 10 °C led to clearly higher DNA content of FL than inoculations at 15 and 20 °C; 4 h of 99% RH after spraying of spores were enough for successful infection. The optimum for sporulation of FL was 25 °C (Nazari et al. 2016). For FS isolates in USA from wheat and maize the optimums were higher (Nazari et al. 2014, 2016). Toxin production by FL strains occurred over 10–35 °C with an optimal range of 20–30 °C (Nazari et al. 2014). For isolates from oat in north-western Europe, the optimal range was found to be the same (Medina and Magan 2011; Kokkonen et al. 2012b; Imathiu et al. 2016). The growth and toxin production by FL were reduced by water activity (aw) when it fell below 0.98, and at 0.90–0.93 the growth rate was very low (Medina and Magan 2011; and Kokkonen et al. 2012b). The temperature response of the toxin production was strongest at high aw, 0.98–0.99, and weak at 0.95. During the active growing period of oat in northern Europe, afternoon temperatures (ECA&D 2018) reach daily the steady state optimal range for growth and toxin production by FL in June to August, yet for most hours of the day temperatures in the air and in soil surface layers are below the optimum (Børresen and Njøs 1990; Mikkola 2014); only the optimum temperature for the infection by spores is reached 24 h a day during the expected period of primary infections. In living oat tissues aw remains high until ripening, when it falls initially below 0.95 (gravimetric water content approximately 25%) and then further under dry weather, but it varies strongly with air humidity and rain. Key information for timing the weather effects is the observed phenological phase when primary infections by FL take place. Injecting spores at boot stage, Divon et al. (2012) could create head infections, but Imathiu et al. (2013) and Opoku et al. (2013) found that the primary infections that later lead to toxin accumulation occurred largely at head emergence and anthesis. Schöneberg et al. (2019) found that inoculation with spores at GS61 and GS65 led to high DNA content of FL in oat grains, whereas earlier inoculations at GS51 and GS55 led to very low content.
These observations provide a basis for hypotheses of limitation of FL by weather. The hypotheses state that temperature limits growth of FL and T-2 + HT-2 production by FL throughout the season, but that low temperature favours primary infection. Humidity effects are expected to be seen near anthesis, when humidity limits primary infections by FL, and near ripening when under dry weather aw in grains falls below the demand for FL growth. Annual weather effects on FL and other Fusarium species may accumulate over years to become climatic effects.
The inoculum potential of FL is affected by agronomic factors, which is indicated by observed higher average T-2 + HT-2 contamination under non-inversion than inversion tillage in oat (Parikka et al. 2007; Bernhoft et al. 2010; Kuzdraliński et al. 2013; Hofgaard et al. 2016b; Edwards 2017; Imathiu et al. 2017; Schöneberg et al. 2018) and barley (Orlando et al. 2010; Schöneberg et al. 2016). Oat residue may be carrying more FL to the next season than the residues from other cereals because oat is the most susceptible cereal species to FL (Opoku et al. 2018) and the residue of oat turned to the soil surface has been found to increase T-2 + HT-2 contamination (Edwards 2017). However, in Europe the geographic distribution of the contamination does not seem to be well explained by the proportion of oat in rotation and tillage methods. The average contamination is lower in Finland than in the UK (Edwards 2009, Hietaniemi 2016, Edwards 2017), even though the proportion of arable land covered by oat is regionally five to fifteen times higher in Finland than in the UK (Eurostat 2017); the cereal intensity of arable land and the use of non-inversion tillage are currently approximately equally high in Finland and the UK (Eurostat 2013, 2017).
In this study, data collected over twelve years in 804 oat fields were analysed. The primary goals were to estimate the timing of weather effects on T-2 + HT-2, FL and FS oat and to assess the relative importance of agronomic, weather and climatic factors. Climatic factors refer to long-term cumulative weather factors, but may also reflect climate-dependent cropping systems. Weather and climate are correlated by definition, but in 1- to 2-week periods tied to oat phenology variation of weather around climatic means is high. It is caused by variation in weather and in annual variation of up to 20 days in the dates of sowing, anthesis and harvest in survey sites. There are two major difficulties in interpreting survey data on toxins and Fusarium species. Firstly, climatic and weather effects are confounded with the effects of agronomic practices since farm types, soils and crop species are geographically clustered. Secondly, the range of weather in a survey domain determines how well weather effects are detected. Some effects may be hidden, while others are more dominant than in a wider geographic domain. This has an unknown influence on the validity of the detected effects outside the survey domain and complicates comparison with results obtained elsewhere. The approach chosen for reducing the confounding was to stratify the data with respect of climate. For assessing the validity of the found effects on T-2 + HT-2, FL and FS, they were compared with prior hypotheses that were derived from lower system studies, and they were benchmarked with analogous effects on DON, FG, FC, FP and other Fusarium species.
Methods
Survey, climatic and weather data
Field data consisted of T-2, HT-2 and DON concentrations and percentage incidences of Fusarium species (percentage of contaminated grains) that had been determined from grain samples in the Natural Resources Institute Finland (Luke, formerly MTT AgriFood Research Finland). Toxin concentration was available for 804 fields in 2003–2014 and the incidences of Fusarium species in 2007–2014 for 606 fields, which coincided with the toxin samples (Table 2, part 4). These data were a subset of larger data of which sampling methods, toxin analysis and the microbiological determination of Fusarium species have been described by Hietaniemi et al. (2016). The observations selected to the subset had the same sampling procedure, equal methods for toxin and Fusarium determination and data were owned by Luke. Agronomic practices were available for most fields. They included sowing and harvest dates (Number of observations N = 803), moldboard ploughing (typically 25 cm depth, ploughed N = 662, non-ploughed N = 142), secondary tillage in non-ploughed fields (zero tillage N = 34, minimum tillage with chisel plough or disc harrow once or twice N = 108), and cereal intensity, defined as the number of cereal crops in four years preceding a sampling year (0 cereals crops (recoded to 1) N = 2,; 1 N = 257; 2 N = 348; 3 N = 66; 4 N = 129). Non-cereal pre-crop species and their sequences were not known, but typically both under non-inversion and inversion tillage they were broad-leaved species (rapeseed, potato, caraway, sugar beet) or fallow crops (annual grasses and broad-leaved species); under ploughing also perennial grasses.
Climate (daily mean temperature °C and rainfall mm in 1981–2010) and weather variables (three-hourly mean temperature °C, relative humidity % (RH) and rainfall (mm)) were derived from spatially gridded (10 × 10 km) data provided by the Finnish Meteorological Institute. The data had been built using observations from weather stations and rain radars (Vajda et al. 2013; Aalto et al. 2016). Since coordinates of sampled fields were not recorded and they were within a few kilometres from farm houses, the coordinates of farm addresses served as field coordinates.
Moving correlations with weather
Computing moving correlations (SAS proc corr) of the natural logarithm transformed concentrations of T-2 + HT-2 and DON and the incidences of Fusarium species with weather variables was the first step for timing weather effects and potential climate effects on toxins and Fusarium species. They also provided profiles for visual comparisons of the responses of the toxins and Fusarium species. The weather variables computed from 3-hourly data were means and sums over a seven-day window (−3 to +3 days from the window centre point). The window was moved one day at a time from 42 db-GS65 (days before mid-anthesis, see Abbreviations) to harvest. From 42 db-GS65 to 14 days before harvest the position of the window was expressed as a time distance to GS65; after that as a distance to the date of harvest. The dates of GS65 in surveyed fields were estimated with cultivar-specific linear temperature sum above 5 °C (ETS) from sowing date to GS65. ETS requirements of cultivars were derived from standard cultivar trials (Laine et al. 2015) where sowing and harvest dates and major phenological stages were recorded (unpublished). GS65 was set to 50 degree-days after panicle emergence, since anthesis lasts approximately 100 degree-days (Rajala and Peltonen-Sainio 2011). The ETS method may not have provided exact timing of anthesis, but the error was the same for T-2 + HT-2, DON and all Fusarium species, which allows comparing of the timing of their weather responses.
The weather variables were: mean temperature of a seven day period (Tm) (note that periods are not marked in variable names), sum of rainfall over seven days (Rainfall), weighted duration of high relative humidity over seven days (RHhours) [RHhours = ∑t1/(1 + exp(−s ∗ (RHt − m)))] and the interaction of Tm with RHhours (RHhours×Tm) [RHhours × Tm = ∑tTm ∗ 1/(1 + exp(−s ∗ (RHt − m)))], where t was hours over seven days, s = 0.25 and m = 80. s and m were constants that changed a step function (value of hourly duration rises from 0 to 1 as RH exceeds 80%) to a sigmoid function (value of hourly duration increases sigmoidally from 0 to 1 as RH increases from 65 to 95%, passing 0.5 at m = 80% RH). The purpose of using a sigmoidal threshold was to reduce the sensitivity of RHhours to possible small systematic spatial variation of the error of gridded RH.
Relative effects of climate, weather and agronomic factors
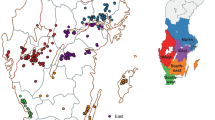
Cropping systems of the surveyed fields were known to be correlated to long-term climate – in warmer and drier regions cereal intensity was higher, ploughing less common and there were less grass break-crops (Table 2, parts 5, 6). Therefore, the effects of cereal intensity and ploughing were expected to be better detected if the data were stratified with respect to climate. Five disjoint regions (Fig. 4) were created with cluster analysis (SAS proc fastclus) on the basis of 30-year mean values of climatic variables and the proportion of arable land sown with cereals at the grid cells of the surveyed fields. The cereal areas to grid cells were taken from 192 municipalities in which the cells were located (Luke 2017). All input variables were standardized to mean zero and variance one to remove the influence of measuring scales. The climatic variables were constructed by using gridded daily data from the years 1980–2009 and GS65 dates predicted with ETS from grid cell specific mean sowing dates. Possible climatic variables for the cluster analysis were suggested by the moving correlation analysis (see above) and before cluster analysis their usefulness was assessed with linear regression log(ToxinConcentration) = α + β’x, where x were observations of climTm, climRainfall and their interaction. Since 30-year data at lower time resolution than a day was not available, the duration of high RH could not be used. The variables chosen to assign cells to regions in the cluster analysis were climRainfall (climatic rainfall) 35–26 db-GS65, climTm (climatic mean temperature) 10–1 db-GS65, climRainfall×climTm 10–1 db-GS65, climTm 5–24 da-GS65 (days after mid-anthesis, see Abbreviations).
Options for modelling the relative effects of environmental and agronomic factors on T-2 + HT-2 were limited by the large proportion of observations where T-2 + HT-2 concentration was below the detection limit (Table 2, part 2). The data met the requirements of logistic regression, which however did not provide interesting regional mean concentrations of toxins under ploughing and non-ploughing. They were computed with analysis of covariance (ancova) using cereal intensity (Cereal intensity) (number of preceding cereal crops 1 to 4) as a covariate.
Model 1
The probabilities of T-2 + HT-2 concentration quantiles (0, 0–75%, 75–100%) were predicted by logistic regressions, which treated all effects as fixed (SAS proc logistic). Four regressions were computed. First, using pooled observations of climatic variables with a model Prob(T-2 + HT-2 < 75% limit |x) = α + β’x, where x consisted of observations of variables climTm, climRainfall and their interactions. In the second stage pooled values of weather variables Tm, Rainfall, RHhours and their interactions were added as predictors. In the third stage ploughing (Ploughing) and Cereal intensity were added as predictors; they were first tried in a one-level model where all data were pooled, then in a two-level model where Ploughing and Cereal intensity were nested in the regions (A–E). Finally in the fourth stage, Inoculum presence of FL (FL = 0 or FL > 0) was added as a predictor. Inoculum presence was obviously partly associated with the other predictors, but it provided useful information for understanding the factors that determine T-2 + HT-2 contamination.
Potential climatic and weather variables for the models were derived from the moving correlation analysis (see above). They were concatenated over time when it improved or simplified the models. To describe the degree of collinearity of climatic and weather variables, their Pearson correlations were computed (Correlation 1). The regions had different average toxin contaminations (Table 2, parts 1, 2), but region effects themselves were not of interest since they were described with the climatic variables and regional differences in break-crop species in surveyed fields were not known. To show the relative effect sizes of the predictors, final parameter values were calculated using data where the predictors were standardized to mean zero and variance one. This removed intercepts from the models.
Model 2
Regional (A–E) mean effects of ploughing (ploughed or non-ploughed) on T-2 + HT-2 and DON contaminations were computed with ancova by region (A –E) using cereal intensity (1 to 4) as a covariate. The model was log(ToxinConcentration) = α + β’x, where x consisted of observations of ploughing and cereal intensity (SAS proc glm). Because even within regions local climate could have influenced the choice of tillage method and thus tillage effects might represent partly climatic effects, two-sided t-test was computed by region to see if Tm, RHhours, Rainfall and interaction RHhours×Tm in the periods used in the regression model 1 were different in ploughed and non-ploughed fields (t-test 1). The independence of ploughing from cereal intensity was tested by computing Kendall rank correlation of Ploughing with Cereal intensity (Correlation 2). To test if in non-ploughed fields toxin concentrations were affected by zero tillage vs. minimum tillage, Kendall rank correlation of zero vs. minimum tillage with toxin concentrations was computed using pooled data (Correlation 3) (SAS proc corr).
Model 3
To show the effects of ploughing (ploughed or non-ploughed) and cereal intensity (1 to 4) on the probability of high toxin concentrations (>75% quantile limit) without weather effects, binary logistic regressions (logit link) computed by region (A–E) were used (Prob(T-2 + HT-2 < 75% limit |x) = α + β’x, where x consisted of observations of Ploughing and Cereal intensity (SAS proc logistic).
Model 4
The effects of ploughing (ploughed or non-ploughed) and cereal intensity (1 to 4) on incidences of Fusarium species were predicted with Poisson regression computed by region (A–E) (log(Incidence%) = α + β’x, where x consists of observations of Ploughing and CerealIntensity) (SAS proc genmod). All effects were fixed.
Relative importance of Fusarium species
Model 5
Relative importance of Fusarium species for producing T2 + HT2 and DON were assessed by computing multiple linear regressions by region (A–E) where natural logarithm transformed T2 + HT2 and DON were predicted by FL, FS and FG, FC, respectively (SAS proc reg).
Results
Moving correlations with weather
At 30–20 db-GS65, T-2 + HT-2 concentration was positively correlated with Tm and negatively correlated with the variables that indicated wet weather (Rainfall and RHhours) (Fig. 1). Then, the correlation signs changed. A peak positive correlation with humid weather and negative correlation with cool weather occurred at 12–7 db-GS65. From a few db-GS65 until harvest, T-2 + HT-2 concentration was continuously positively correlated with Tm regardless of humidity. For most post-GS65 days, especially in the two weeks preceding harvest, T-2 + HT-2 was positively correlated with interaction RHhours×Tm. Until 14 da-GS65, T-2 + HT-2 was negatively correlated to Rainfall. Tm and Rainfall are mostly negatively associated in summer, which means that a correlation of the toxin with Tm may tell of a correlation of opposite sign with humidity or rainfall, unless otherwise suggested by a correlation of toxin with RHhours×Tm.
Moving correlations (vertical axes) of 7-day weather variables with concentrations of total T-2 and HT-2 (solid line), DON (dashed line). Correlations above and below gray area are significant at <0.05 level. Horizontal axis is day relative to mid-anthesis (0) and after discontinuation at day 43, day preceding harvest. Weather variables are: (a) average temperature °C (Tm), (b) rainfall mm (Rainfall), (c) duration of rain (Rainhours), (d) weighted duration of high relative humidity (RHhours), (e) interaction of weighted duration of high relative humidity × temperature (RHhours×Tm). Number of obs. For every day 804
Compared with T-2 + HT-2, DON responded more positively to Tm 20–30 db-GS65 when daily mean temperatures were 13–14 °C, and DON was more positively associated with Rainfall, except during the two weeks preceding harvest when it was positively related to cool and dry weather (Fig. 1). Two weeks before GS65, the response of DON to Rainfall and RHhours rose at the same time as the response of T-2 + HT-2, but the peak positive response of DON was approximately five days later and the response continued much longer, until 7–10 da-GS65.
The incidence of FL responded to weather very much like T-2 + HT-2 concentration (Fig. 2). The correlation profile of FS (Fig. 2) resembled that of FL, having similar peak response to Rainfall and other humidity variables at 10 db-GS65, and being constantly positively correlated with Tm after GS65. In 30–14 db-GS65, when Tm ranged from 11 to 19 °C, FS was more strongly than FL related to warm and dry weather. Based on the temperature and rainfall profiles, FP (Fig. 2) belonged clearly to the same group as FL and FS although FP was consistently favoured by drier conditions. FG responded to weather like DON (Fig. 3). FC (Fig. 3) was favoured by drier pre-GS65 weather than FG and had a very weak humidity response during anthesis. Weather types positively affecting F. avenaceum (Fr.:Fr.) Sacc. (FA) were a warm early season, rainy and humid 15–25 db-GS65, warm anthesis phase, cool and dry 10–25 da-GS65, increasingly rainy and humid towards ripening, and cool and dry near harvest (results not shown). F. tricinctum (Corda) Sacc. (FT) was correlated with a cool and dry week around GS65 and with increasingly warm weather after GS65 (results not shown). Other Fusarium species did not correlate with weather.
Moving correlations (vertical axes) of 7-day weather variables with percentage incidence of F. langsethiae (FL, solid line), F. sporotrichioides (FS, dashed line) and F. poae (FP, dotted line). FL and FS adjusted for cereal intensity and moldboard ploughing. Significance, horizontal axes and weather variables as in Fig. 1. Number of obs. For every day 606
Moving correlations (vertical axes) of 7-day weather variables with percentage incidence of F. graminearum (FG, solid line), F. culmorum (FC, dashed line). Significance, horizontal axes and weather variables as in Fig. 1. Number of obs. For every day 606
Relative effects of climate, weather and agronomic factors
The cluster analysis defined five regions largely by their climate. In Fig. 4 they are labelled A–E. The proportion sown with cereals could have been left out, but it joined a few spatial outliers to a surrounding region. In a one-level logistic model, Cereal intensity was not significant and Ploughing was only weakly significant. In a two-level nested model (Model 1), where Cereal intensity and Ploughing effects were nested in regions, their effects were significant in regions C to E (Table 1). Estimates of the generalized coefficient of determination (R2) indicated that the strongest predictor of T-2 + HT-2 contamination was Inoculum presence (Table 1). Climate and weather were roughly equally important predictors, and together they explained more than Ploughing and Cereal intensity. Significant climatic variables were Tm, its interaction with Rainfall in 10–1 db-GS65 and Tm in three weeks after GS65 (Table 1, part 1). Of the weather variables, T) from 5 db-GS65 to 39 da-GS65 was the most important predictor (Table 1, part 2).
Collinearity between the climatic and the weather variables was not higher than among the weather variables: absolute values of correlations of weather variables with climatic variables (Correlation 1) ranged from 0.01 to 0.33 (Table 1, part 5), while absolute values of correlations between weather variables ranged from 0.01 to 0.34 (result not shown). Within the regions, ploughing acted independently of weather: in two-sided t-tests (t-test 1) Tm, RHhours, Rainfall and interaction RHhours×Tm were not significantly different (p < 0.05) in ploughed and non-ploughed fields, except in region A, where early season temperature (Tm 40–21 db-GS65) was on average 0.6 °C lower in ploughed fields (results not shown). T-2 + HT-2 was correlated with zero (N = 34) vs. minimum tillage (N = 108) (Correlation 3, Kendall rank correlation tau = 0.15, p = 0.03). Median concentrations were 196 μg kg−1 and 133 μg kg−1 in zero and minimum tilled fields, respectively. Cereal intensity was not correlated with zero vs. minimum tillage (Correlation 2, tau = −0.017, p = 0.82), which indicates that the zero tillage effect was real and not caused by cereal intensity. DON was not correlated with zero vs. minimum tillage (tau = −0.02, p = 0.82).
Ancova (Model 2) showed that mean T-2 + HT-2 concentration was significantly lower in ploughed than in non-ploughed fields in regions C to E (Table 2, part 1). Cereal intensity was significantly positively associated with T-2 + HT-2 in region D, and did not significantly interact with ploughing (results not shown). DON was significantly higher in ploughed fields in regions A and B (Table 2, part 1).
The probability of high T-2 + HT-2 concentration (> Q75) (Model 3) was lower in ploughed than non-ploughed fields in regions C to E, whereas DON was significantly affected by ploughing in any (Fig. 5; Table 3, part 1). In region D the probability rose with increasing cereal intensity.
Probability of high toxin concentration (concentration > 75% quantile limit (Q75)) predicted with separate logistic regressions in regions A to E. Predictors cereal intensity (CI) (values on horizontal axes) and moldboard ploughing (MP) (ploughed = dashed line, non-ploughed = solid line). On upper pane row probability of T-2 + HT-2 > Q75, on lower pane row probability DON > Q75. Number of observations and significances of predictors CI and MP and their interactions CI×MP are shown in Table 3. When CI×MP were not significant (p ≤ 0.06), only CI and MP were used in prediction
The incidence of FL was lower in ploughed fields in three regions B, C and E, but higher in A and D (Model 4) (Fig. 6; Table 3, part 2); the incidence of FS was lower in ploughed fields in region A (Fig. 6, Table 3, part 2). The incidence of FG was higher in ploughed fields in three regions, and it decreased as cereal intensity increased in four regions (Model 4) (Table 3, part 2). The incidence of FC was clearly higher in ploughed than non-ploughed fields in four regions (Fig. 6; Table 3, part 2). The incidence of FP was higher in ploughed fields in regions A, B and D (Fig. 6; Table 3, part 2). FA was lower in ploughed fields in region D but decreased in C (Fig. 6; Table 3, part 2). F. tricinctum (FT) was significantly lower in ploughed fields in regions A, B, D, E and it was reduced in B and increased in D with increasing cereal intensity (results not shown). F. arthrosporioides Sherb. (FR) was significantly higher in ploughed fields in region C but lower in E; it decreased with increasing cereal intensity in C and E but increased in B (results not shown). Other Fusarium species were not significantly affected by the agronomic factors. Note that the incidences of Fusarium species varied by region and 76–90% of fields were ploughed. Therefore, the magnitude of the effects should be interpreted with caution.
Percentage incidence of Fusarium species predicted with separate Poisson regressions in regions A to E. Predictors cereal intensity (CI) (values on horizontal axes) and moldboard ploughing (MP) (ploughed = dashed line, non-ploughed = solid line). On pane rows FL = F. langsethiae %, FS = F. sporotrichioides %, FG = F. graminearum %, FC = F. culmorum %, FP = F.poae % and FA = F. avenaceum %. Number of observations and significances of predictors CI and MP and their interactions CI×MP are shown in Table 3. When CI×MP were not significant (p ≤ 0.06), only CI and MP were used in prediction
Relative importance of Fusarium species
T2 + HT2 was predicted largely by the incidence of FL (Table 4, part 1), but FS was a minor co-predictor in regions B, D and E increasing R2 less than by one fifth (Table 4, part 2) (Model 5). DON was predicted only by the incidence of FG in south-western region A (Table 4, part 3). In other regions FC increased R2 by about one tenth (Table 4, part 4).
Discussion
T-2 + HT-2 and DON contaminations were largely attributed to FL and FG, respectively, while FS and FC were minor toxin producers. To T-2 + HT-2 contamination contributed weather, agronomic factors and climate, but their overall explanation power (R2) was only 0.20. A major reason for the low explanation power was likely the low inoculum level of FL in a large part of fields, which prevented weather effects from becoming manifested. Observed presence of FL was used as an additional predictor for correcting the lack of knowledge of inoculum presence. For better explanation levels other predictors that would enable the use of zero inflated models would be needed. The10 × 10 km resolution of the weather data provides generally rather good temperature estimates for fields within grid cells, but it smooths out local peaks of RH and especially convective rainfall. Because they were important near anthesis, any errors in the timing of anthesis further reduced their accuracy. However, comparisons between T-2 + HT-2, DON and Fusarium species were less affected because the timing errors were the same for all of them. Separately from weather variables, climatic variables captured information of grid cell means. Since the significant climatic variables were theoretically meaningful in the same way as the weather variables, a plausible explanation is that they represent the cumulative influence of past weather and climate dependent cropping systems on the inoculum potential of FL.
Ploughing, using minimum tillage instead of zero tillage and having low cereal intensity of rotation mostly reduced T-2 + HT-2 contamination, but had contradictory effects on FL. By contrast, DON was mostly increased by ploughing, not affected by zero tillage and generally not affected by cereal intensity. Corroborating the tillage effect on DON, FG and FC were mostly more common under ploughing than under non-ploughing, and FG decreased with increasing cereal intensity in four regions out of five. On the other hand, FC incidence increased with cereal intensity in the three most northern regions.
The detected effects of cereal intensity and ploughing on T-2 + HT-2 fit the general pattern found in previous surveys and experiments on oat (Parikka et al. 2007; Bernhoft et al. 2012; Kuzdraliński et al. 2013; Hofgaard et al. 2016b; Edwards 2017; Imathiu et al. 2017; Schöneberg et al. 2018) and on barley (Orlando et al. 2010; Schöneberg et al. 2016). According to Edwards (2017), the pre-crop effect is cumulative, depends on the sequence of the pre-crops, is stronger with grass than other non-cereal pre-crops and interacts with ploughing. Regarding DON and its producers, the results of this study are counterintuitive but not entirely contrary to previous results. For DON in wheat, the effects of tillage methods have been found to be quite variable (e.g. Dill-Macky and Jones 2000; Schaafsma et al. 2001; Munger et al. 2014). The survival and inoculum potential of FG in wheat and oat fields have been found to be lower under moldboard ploughing than under zero or minimum tillage (Guo et al. 2010; Hofgaard et al. 2016b). However, non-inversion tillage can also bury a large proportion of crop residue, which is considered to reduce inoculum potential of FG (Khonga and Sutton 1988; Pereyra et al. 2004; Koch et al. 2006). Fernandez et al. (2005, 2007, 2008) found in Saskatchewan on wheat and barley that head infections by FG were occasionally increased by broad-leaved non-cereal pre-crops; tillage interacted with rotation; FG contamination in heads and crop residue tended to be the same or higher under minimum tillage than under ploughing, but under zero tillage FG contamination was lower than under ploughing. Tillman et al. (2017) in Germany found that FG was more common in winter wheat grains following sugar beet than following wheat. According to Rasiukevičiūtė et al. (2018), faba bean, pea, sugar beet, fodder beet, oilseed rape and potato plants may serve as alternative or reservoir hosts for FG isolates that can infect wheat. In an experiment by Supronienė et al. (2012), ploughing reduced T-2 + HT-2 in winter wheat, but tended to increase DON. Importantly, the conclusions from this study are locally supported by the results of the tillage experiments reported by Parikka et al. (2018): in 19 oat and barley experiments in multiple locations and years in Finland, FL incidence was on average higher, but the incidences of FG and FC were lower under zero tillage than under ploughing.
Tillage and rotation effects on Fusarium species may be modified by soil fauna. When not disturbed by ploughing, population densities of epigeic and especially anenic earthworms increase over years (Briones and Schmidt 2017). The most important anecic species in Europe and Finland is Lumbricus terrestris L. (Nieminen et al. 2011), which creates valuable deep macropores in soil and incorporates and breaks down straw, thereby potentially reducing Fusarium density on the soil surface. The reduction is enhanced by the preference of L. terrestris for Fusarium infested straw over non-infested straw (Wolfarth et al. 2016).
In their responses to weather, FL and T-2 + HT-2 differed from FG and DON mainly by having a shorter, 5–10 days earlier and weaker positive response to humidity near anthesis and a strikingly consistent positive response to warm weather in the post-anthesis period, when in first 4 weeks the range of daily mean temperatures is 15–23 °C and maximum temperatures are 19–30 °C. If GS65 dates in fields were on average correctly estimated, the positive humidity response peak and the negative temperature response peak of T-2 + HT-2 at 5–12 db-GS65 correspond to panicle emergence or early anthesis. The peaks coincide approximately with the phase when floret infections by FL have been determined to take place in the UK (Imathiu et al. 2013; Opoku et al. 2013) and FL has been isolated from panicles in Finland (Parikka et al. 2007). According to Tekle et al. (2012), the peak period of primary infections of oat by FG is later, after mid-anthesis. Over the post-GS65 period, humidity was of secondary importance for T-2 + HT-2, but not without influence, as was shown by the positive RHhours×Tm interaction. It is obvious that responses of Fusarium species to weather are conditional to crop phenology onwards from a few weeks before anthesis, but earlier responses might depend on crop status only through its influence on microclimate.
A comparison of the weather responses observed in Finland, the UK (Xu et al. 2014) and Norway (Hjelkrem et al. 2018) shows that the responses are qualitatively remarkably similar with only minor differences. Early in the season, temperature response was weaker in Finland than in the UK, but due to late sowing in Finland this period is warmer and drier in Finland than in the UK (ECA&D 2018). In the post-anthesis period, the response to daily mean temperature was as strong and consistent as in the UK, even though daily mean temperatures in the first four weeks after anthesis are 2–4 °C higher in Finland than in the UK (ECA&D 2018). In Norway, the response to daily mean temperature was weak, but the duration of temperature and relative humidity exceeding minimum thresholds was a significant predictor. At ripening, when daily temperature and RH are not negatively linked under Finnish climate, temperature and duration of high RH interacted positively, unlike under warmer autumn climate in Norway. In the UK, where oat is mostly harvested under warmer and drier conditions than in Finland, the humidity effect was not detected.
Based on their environmental responses, FG and FC form one group and FL, FS and FP form another. These groups correspond loosely to two phylogenetic groups of Fusarium species that infect small grains in northern Europe (Yli-Mattila 2010). Unlike FG and FC, FL, FS and FP were strongly favoured by warm post-anthesis weather. Most striking in the weather response of FG was an early positive response to warm weather followed by a three-week long strong positive response to duration of high humidity, which may reflect the temperature and humidity requirements of the growth and maturation of perithecia and ascospores (e.g. Manstretta and Rossi 2016). FC has in vitro a lower temperature optimum and is less sensitive to low aw than FG (Brennan et al. 2003; Hope et al. 2005). These traits fit with the observed higher tolerance of FC to low temperature at early season and the negative response of FC to humid pre-anthesis weather. FC did not significantly respond to humidity during anthesis, which suggests that at this stage FC could infect oat at lower humidity than FG, as shown by Rossi et al. (2001) on wheat, or a large part of recorded FC infections occurred later than at anthesis. In the third phylogenetic group consisting of FA, FR and FT (Yli-Mattila 2010), FA and FR were climate and cropping system generalists, whereas FT incidence was clearly lower under ploughing than under non-ploughing.
In conclusion, T-2 + HT-2 and DON contaminations were largely attributed to FL and FG, respectively, and only small proportions were attributed to FS and FC. The results suggest that T-2 + HT-2 contamination was influenced both by weather and factors that were represented by the 30-year climatic variables. The responses to high humidity and temperature indicated that before or during anthesis, infections by FL leading to T-2 + HT-2 accumulation occurred earlier and over a much shorter period than the infections by FG that led to accumulation of DON. T-2 + HT-2 contamination was favoured by seasons with humid and cool weather one to two weeks before anthesis and warm weather in other stages. The effect of temperature on T-2 + HT-2, FL and FS was especially consistent after anthesis. The validity of the observed weather responses is not strictly limited to the surveyed domain, which is shown by the matching of the observed responses with the hypotheses derived from in vitro studies and with the observations made in oceanic climates. This implies that it may be possible to construct a model that would describe the quantitative theory of the infection pressure of FL and the risk of T-2 + HT-2 in Northern Europe. Average T-2 + HT-2 contamination and the risk of high contamination were reduced by ploughing, minimum tillage instead of zero tillage and rotation with non-cereal crops. However, they were inefficient for controlling DON, FG, FC, FP and FA or even increased them. Since common rotation practices and tillage methods of cereal farms are not efficient for controlling the total toxin risk, further research should aim to assess the influence of specific hypothetically effective crops and increasingly used novel crops, inter-crops, green manure crops and fertilizers of organic origin.
Availability of data
The original data analysed in the current study are not publicly available because of data protection of farmers, the policies of institutes involved in data collection and excessive amount of data. Please contact corresponding author for data requests.
Abbreviations
- FL:
-
F. langsethiae
FS
F. sporotrichioides
FP
F. poae
FG
F. graminearum
FC
F. culmorum
FA
F. avenaceum
FT
F. tricinctum
FR
F. arthrosporioides
T-2 + HT-2
sum of T-2 and HT-2 concentrations (μg/kg)
Tm
mean temperature °C
RHhours
weighted duration of relative humidity h
climTm
30-year mean temperature °C
climRainfall
30-year rainfall mm. GS65
mid-anthesis
db-GS65
days before mid-anthesis
da-GS65
days after mid-anthesis.
References
Aalto, J., Pirinen, P., & Jylhä, K. (2016). New gridded daily climatology of Finland: Permutation-based uncertainty estimates and temporal trends in climate. Journal of Geophysical Research – Atmospheres, 121, 3807–3823.
Bernhoft, A., Clasen, P. E., Kristoffersen, A. B., & Torp, M. (2010). Less Fusarium infestation and mycotoxin contamination in organic than in conventional cereals. Food Additives and Contaminants: Part A, 27, 842–852.
Bernhoft, A., Torp, M., Clasen, P., Løes, A., & Kristoffersen, A. (2012). DON content in oat grains in Norway related to weather conditions at different growth stages. Influence of agronomic and climatic factors on Fusarium infestation and mycotoxin contamination of cereals in Norway. Food Additives and Contaminants: Part A, 29, 1129–1140.
Børresen, T., & Njøs, A. (1990). The effects of three tillage systems combined with different compaction and mulching treatments on soil temperature and soil thermal properties. Norwegian Journal of Agricultural Sciences, 4, 363–371.
Brennan, J. M., Fagan, B., van Maanen, M., Cooke, B. M., & Doohan, F. M. (2003). Studies on in vitro growth and pathogenity of European Fusarium fungi. European Journal of Plant Pathology, 109, 577–587.
Briones, M.J. & Schmidt, O. (2017). Conventional tillage decreases the abundance and biomass of earthworms andalters their community structure in a global meta-analysis. Global Change Biology, 23, 4396–4419.
Dill-Macky, R., & Jones, R. K. (2000). The effect of previous crop residues and tillage on Fusarium head blight of wheat. Plant Disease, 84, 71–76.
Divon, H. H., Razzaghian, J., Udnes-Aamot, H., & Sletner Klemsdal, S. S. (2012). Fusarium langsethiae (Torp and Nirenberg), investigation of alternative infection routes in oat. European Journal of Plant Pathology, 132, 147–161.
ECA&D (2018). https://www.ecad.eu/. Accessed 15 March 2018.
Edwards, S. G. (2009). Fusarium mycotoxin content in UK organic and conventional oat. Food Additives and Contaminants: Part A, 26, 1063–1069.
Edwards, D. G. (2017). Impact of agronomic and climatic factors on the mycotoxin content of harvested oat in the UK. Food Additives & Contaminants: Part A, 34, 2230–2241.
Edwards, S. G., Imathiu, S. M., Ray, R. V., Back, M., & Hare, M. C. (2012). Molecular studies to identify the Fusarium species responsible for HT-2 and T-2 mycotoxins in UK oat. International Journal of Food Microbiology, 156, 168–175.
EuroStat (2013). http://ec.europa.eu/eurostat/statistics-explained/index.php/Agri-environmental_indicators . Accessed 5 April 2018.
Eurostat (2017). Crop statistics by NUTS 2 regions (from 2000 onwards). http://appsso.eurostat.ec.europa.eu. Accessed 5 April 2018.
Fernandez, M. R., Selles, F., Gehl, D., DePauw, R. M., & Zentner, R. P. (2005). Crop production factors associated with Fusarium head blight in spring wheat in eastern Saskatchewan. Crop Science, 45, 1908–1916.
Fernandez, M. R., Huber, D., Basnyat, P., & Zentner, R. P. (2008). Impact of agronomic practices on populations of Fusarium and other fungi in cereal and noncereal crop residues on the Canadian prairies. Soil and Tillage Research, 100, 60–71.
Fredlund, E., Gidlund, A., Pettersson, H., Olsen, M., & Börjesson, T. (2010). Realtime PCR detection of Fusarium species in Swedish oat and correlation to T-2 and HT-2 toxin content. World Mycotoxin Journal, 3, 77–88.
Fredlund, E., Gidlund, A., Sulyok, M., Börjesson, T., Krska, R., Olsen, M., & Lindblad, M. (2013). Deoxynivalenol and other selected Fusarium toxins in Swedish oat – Occurrence and correlation to specific Fusarium species. International Journal of Food Microbiology, 167, 276–283.
Guo, X. W., Fernando, W. G. D., Bullock, P., & Sapirstein, H. (2010). Quantifying cropping practices in relation to inoculum levels of Fusarium graminearum on crop stubble. Plant Pathology, 59, 1107–1113.
Hietaniemi, V., Rämö, S., Yli-Mattila, T., Jestoi, M., Peltonen, S., Kartio, M., Sieviläinen, E., Koivisto, T., & Parikka, P. (2016). Updated survey of the Fusarium species and toxins in Finnish cereal grains. Food Additives and Contaminants: Part A, 33, 831–848.
Hjelkrem, A.-G., Aamot, H., Brodal, G., Strand, E., Torp, T., Edwards, S. G., Dill-Macky, R., & Hofgaard, I. (2018). HT-2 and T-2 toxins in Norwegian oat grains related to weather conditions at different growth stages. European Journal of Plant Pathology, 151, 501–514.
Hofgaard, I. S., Aamot, H. U., Torp, T., Jestoi, M., Lattanzio, V. M. T., Klemsdal, S. S., Waalwijk, C., Van der Lee, T., & Brodal, G. (2016a). Associations between Fusarium species and mycotoxins in oat and spring wheat from farmers’ fields in Norway over a six-year period. World Mycotoxin Journal, 9, 365–378.
Hofgaard, I. S., Seehusen, T., Aamot, H. U., Riley, H., Razzaghian, J., Le, V. H., Hjelkrem, A.-G., Dill-Macky, R., & Brodal, G. (2016b). Inoculum potential of Fusarium spp. relates to tillage and straw management in Norwegian fields of spring oat. Frontiers in Microbiology, 7, 556–571.
Hope, R., Aldred, D., & Magan, N. (2005). Comparison of environmental profiles for growth and deoxynivalenol production by Fusarium culmorum and F. graminearum on wheat grain. Letters in Applied Microbiology, 40, 295–300.
Imathiu, S., Ray, R., Back, M., Hare, M., & Edwards, S. (2013). A survey investigating the infection of Fusarium langsethiae and production of HT-2 and T-2 mycotoxins in UK oat fields. Journal of Phytopathology, 161, 553–561.
Imathiu, S. M., Ray, R. V., Back, M., Hare, M., & Edwards, S. G. (2016). In vitro growth characteristics of Fusarium langsethiae isolates recovered from oat and wheat grain in the UK. Acta Phytopathologica et Entomologica Hungarica, 51, 159–169.
Imathiu, S., Ray, R., Back, M., Hare, M., & Edwards, S. (2017). Agronomic practices influence the infection of an oat cultivar with Fusarium langsethiae. Acta Phytopathologica et Entomologica Hungarica, 52, 15–27.
Jestoi, M. N., Paavanen-Huhtala, S., Parikka, P., & Yli-Mattila, T. (2008). In vitro and in vivo mycotoxin production of Fusarium species isolated from Finnish grains. Archives of Phytopathology and Plant Protection, 41, 545–558.
Khonga, E. B., & Sutton, J. C. (1988). Inoculum production and survival of Gibberella zeae in maize and wheat residues. Canadian Journal of Plant Pathology, 10, 232–239.
Koch, H. J., Pringas, C., & Maerlaender, B. (2006). Evaluation of environmental and management effects on Fusarium head blight infection and deoxynivalenol concentration in the grain of winter wheat. European Journal of Agronomy, 24, 357–366.
Kokkonen, M., Jestoi, M., & Laitila, A. (2012a). Mycotoxin production of Fusarium langsethiae and Fusarium sporotrichioides on cereal-based substrates. Mycotoxin Research, 28, 25–35.
Kokkonen, M., Medina, A., & Magan, N. (2012b). Comparative study of water and temperature relations of growth and T-2/HT-2 toxin production by strains of Fusarium sporotrichioides and Fusarium langsethiae. World Mycotoxin Journal, 5, 365–372.
Kuzdraliński, A., Solarska, E., & Mazurkiewicz, J. (2013). Mycotoxin content of organic and conventional oat from southeastern Poland. Food Control, 33, 68–72.
Laine, A., Högnäsbacka, M., Kujala, M., Niskanen, M., Jauhiainen, L., & Nikander, H. (2015). Virallisten lajikekokeiden tulokset 2007–2014. Luonnonvara- ja biotalouden tutkimus (p. 3/2015). http://urn.fi/URN:ISBN:978-952-326-002-3.
Langseth, W., Bernhoft, A., Rundberget, T., Kosiak, B., & Gareis, M. (1998). Mycotoxin production and cytotoxicity of Fusarium strains isolated from Norwegian cereals. Mycopathologia, 144, 103–113.
Luke (2017). Luke statistics database. Utilised agricultural area. http://statdb.luke.fi/PXWeb/pxweb/en/LUKE/LUKE__02%20Maatalous__04%20Tuotanto__22%20Kaytossa%20oleva%20maatalousmaa/?tablelist=true&rxid=0be16ebf-d6a8-444d-8a57-093f982b1465 [Table] Accessed 5 April 2018.
Manstretta, V., & Rossi, V. (2016). Effects of temperature and moisture on development of Fusarium graminearum perithecia in maize stalk residues. Applied and Environmental Microbiology, 82, 184–191.
Martin, C., Schöneberg, T., Vogelgsang, S., Mendes Ferreira, C. S., Morisoli, R., Bertossa, M., Bucheli, T. D., Mauch-Mani, B., & Mascher, F. (2018). Responses of oat grains to Fusarium poae and F. langsethiae infections and mycotoxin contaminations. Toxins, 2018, 10. https://doi.org/10.3390/toxins10010047.
Medina, A., & Magan, N. (2011). Temperature and water activity effects on production of T-2 and HT-2 by Fusarium langsethiae strains from north European countries. Food Microbiology, 28, 392–398.
Mikkola, J. (2014). Temperature of clay soil as affected by tillage methods. University of Helsinki, master’s thesis. https://helda.helsinki.fi/bitstream/handle/10138/135972/Maisterintutkielma_Jaakko_Mikkola.pdf?sequence=1. Accessed 5 April 2018.
Munger, H., Vanasse, A., Rioux, S. & Légère, A. (2014). Bread wheat performance, fusarium head blight incidence and weed infestation response to low-input conservation tillage systems in eastern Canada. Canadian Journal of Plant Science, 94, 193–201.
Nazari, L., Pattori, E., Terzi, V., Morcia, C., & Rossi, V. (2014). Influence of temperature on infection, growth, and mycotoxin production by Fusarium langsethiae and F. sporotrichioides in durum wheat. Food Microbiology, 39, 19–26.
Nazari, L., Manstretta, V., & Rossi, V. (2016). A non-linear model for temperature-dependent sporulation and T-2 and HT-2 production of Fusarium langsethiae and Fusarium sporotrichioides. Fungal Biology, 120, 562–571.
Nielsen, L. K., Jensen, J. D., Nielsen, G. C., Jensen, J. E., Spliid, N. H., Thomsen, I. K., Justesen, A. F., Collinge, D. B., & Jørgensen, L. N. (2011). Fusarium head blight of cereals in Denmark: Species complex and related mycotoxins. Phytopathology, 101, 960–969.
Nieminen, M., Ketoja, E., Mikola, J., Terhivuo, J., Sirén, T., & Nuutinen, V. (2011). Local land use effects and regional environmental limits on earthworm communities in Finnish arable landscapes. Ecological Applications, 21, 3162–3177.
Opoku, N., Back, M. A., & Edwards, S. G. (2013). Development of Fusarium langsethiae in commercial cereal production. European Journal of Plant Pathology, 136, 159–170.
Opoku, N., Back, M. A., & Edwards, S. G. (2018). Susceptibility of cereal species to Fusarium langsethiae under identical field conditions. European Journal of Plant Pathology, 150, 869–879.
Orlando, B., Barrier-Guillot, B., Gourdain, E. & Maumené., C. (2010). Identification of agronomic factors that influence the levels of T-2 and HT-2 toxins in barley grown in France. World Mycotoxin Journal, 3, 169–174.
Parikka, P., Hietaniemi, V., Rämö, S., & Jalli, H. (2007). The effect of cultivation practices on Fusarium langsethiae infection of oat and barley. Fusarium diseases in cereals – Potential impact from sustainable cropping systems. In S. Vogelsang, M. Jalli, G. Kovacs, & G. Vida (Eds.), Proceedings of the COST SUSVAR Fusarium Workshop, 1–2 June 2007. Valence (Hungary) (pp. 15–18).
Parikka, P., Kaukoranta, T. & Rämö, S. (2018) Does zero tillage increase mycotoxins in oat and barley in cool climate? 14 th European Fusarium seminar, Tulln, Austria. Book of abstracts, p. 59.
Pereyra, S. A., Dill-Macky, R., & Sims, A. L. (2004). Survival and inoculum production of Gibberella zeae in wheat residue. Plant Disease, 88, 724–730.
Pettersson, H., Hedman, R., Engström, B., Elwinger, K., & Fossum, O. (1995). Nivalenol in Swedish cereals – Occurrence, production and toxicity towards chickens. Food Additives and Contaminants, 12, 373–376.
Rajala, A., & Peltonen-Sainio, P. (2011). Pollination dynamics, grain weight and grain cell number within the inflorescence and spikelet in oat and wheat. Agricultural Sciences, 2, 283–290.
Rasiukevičiūtė, N., Supronienė, S., Kelpšienė, J., Svegzda, P., Kadžienė, G., Šneideris, D., Ivanauskas, A., & Treikale, O. (2018). Susceptibility of non-cereal crops to Fusarium graminearum complex and their role within cereal crop rotation as a source of inoculum for Fusarium head blight. Spanish Journal of Agricultural Research, 16. https://doi.org/10.5424/sjar/2018164-13952.
Rossi, V., Ravanetti, A., Pattori, E., & Giosuè, S. (2001). Influence of temperature and humidity on the infection of wheat spikes by some fungi causing Fusarium head blight. Journal of Plant Pathology, 83, 189–198.
Schaafsma, A. W., Tamburic-Ilinic, L., Miller, J. D., & Hooker, D. C. (2001). Agronomic considerations for reducing deoxynivalenol in wheat grain. Canadian Journal of Plant Pathology, 23, 279–285.
Schöneberg, T., Martin, C., Wettstein, F. E., Bucheli, T. D., Mascher, F., Bertossa, M., Musa, T., Keller, B., & Vogelgsang, S. (2016). Fusarium and mycotoxin spectra in Swiss barley are affected by various cropping techniques. Food Additives & Contaminants: Part A, 33, 1608–1619.
Schöneberg, T., Jenny, E., Wettstein, F. E., Bucheli, T. D., Mascher, F., Bertossa, M., Musa, T., Gräfenhan, T., Keller, B., & Vogelsang, S. (2018). Occurrence of Fusarium species and mycotoxins in Swiss oat – Impact of cropping factors. European Journal of Agronomy, 92, 123–132.
Schöneberg, T., Kibler, K., Wettstein, F. E., Bucheli, T. D., Forrer, H. R., Musa, T., Mascher, F., Bertossa, M., Keller, B., & Vogelgsang, S. (2019). Influence of temperature, humidity duration and growth stage on the infection and mycotoxin production by Fusarium langsethiae and Fusarium poae in oats. Plant Pathology, 68, 173–184.
Supronienė, S., Mankovičienė, A., Kadžienė, G., Kačergius, A., Feiza, V., Feizienė, D., Semaškienė, R., Dabkevičius, Z., & Tamošiūnas, K. (2012). The impact of tillage and fertilization on Fusarium infection and mycotoxin production in wheat grains. Žemdirbystė=Agriculture, 99, 265–272.
Tekle, S., Dill-Macky, R., Skinnes, H., Tronsmo, A. M., & Bjørnstad, Å. (2012). Infection process of Fusarium graminearum in oat (Avena sativaL.). European Journal of Plant Pathology, 132, 431–442.
Tillman, M., von Tiedemann, A., & Winter, M. (2017). Crop rotation effects on incidence and diversity of Fusarium species colonizing stem bases and grains of winter wheat. Journal of Plant Diseases and Protection, 124, 121–130.
Vajda, A., Venäläinen, A., Suomi, I., Junila, P., & Mäkelä, H. P. (2013). Assessment of forest fire danger in a boreal forest environment: Description and evaluation of the operational system applied in Finland. Meteorological Applications, 21, 879–887.
Wolfarth, F., Schrader, S., Oldenburg, E., & Brunotte, J. (2016). Mycotoxin contamination and its regulation by the earthworm species Lumbricus terrestris in presence of other soil fauna in an agroecosystem. Plant and Soil, 402, 331–342.
Xu, X., Madden, L. V., & Edwards, S. G. (2014). Modeling the effects of environmental conditions on HT-2 and T-2 toxin accumulation in field oat grains. Phytopathology, 104, 57–66.
Yli-Mattila, T. (2010). Ecology and evolution of toxigenic Fusarium species in cereals in northern Europe and Asia. Journal of Plant Pathology, 92, 7–18.
Yli-Mattila, T., Paavanen-Huhtala, S., Parikka, P., Hietaniemi, V., Jestoi, M., Gagkaeva, T., Sarlin, T., Haikara, A., Laaksonen, S., & Rizzo, A. (2008). Real-time PCR detection and quantification of Fusarium poae, F. graminearum, F. sporotrichioides and F. langsethiaea compared to mycotoxin production in grains in Finland and Russia. Archives of Phytopathology and Plant Protection, 41, 243–260.
Acknowledgements
Open access funding provided by Natural Resources Institute Finland (LUKE). We thank Dr. Visa Nuutinen for advising on the ecology of earthworms in cropping systems. The collection of original data has been supported by the Ministry of Agriculture and Forestry and The Finnish Cereal Committee (VYR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human participants or animals
Data on people and animals were not collected or analysed.
Informed consent
The data analysed in this study are based on cereal samples collected from farms with the consent of farmers for the purpose of assessing the temporal and geographic distribution of quality of cereals.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kaukoranta, T., Hietaniemi, V., Rämö, S. et al. Contrasting responses of T-2, HT-2 and DON mycotoxins and Fusarium species in oat to climate, weather, tillage and cereal intensity. Eur J Plant Pathol 155, 93–110 (2019). https://doi.org/10.1007/s10658-019-01752-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-019-01752-9