Abstract
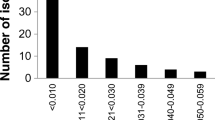
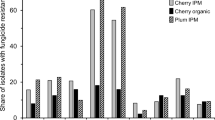
Botrytis cinerea is a complex species prone to fungicide resistance and characterized by enormous genetic diversity. During 2013, 220 B. cinerea isolates causing gray mold were collected from greenhouse-grown crops in the regions of Ammochostos, Larnaca, and Limassol (Cyprus). Sensitivities of the sampled populations to seven botryticides with different modes of action were screened in vitro. The results of this in vitro screening highlighted the widespread phenomenon of fungicide resistance in greenhouses, since only 8.6 % of the isolates were sensitive to all botryticides. Resistance to thiophanate-methyl was the most prevalent, with frequencies ranging from 53.8 % to 80 %. Similarly, high resistance frequencies were observed for pyraclostrobin (27.1 to 78.9 %) and boscalid (28.2 to 66.2 %). Multiple fungicide resistant phenotypes were predominant, covering 67.3 % of the population, with frequencies of 80.0, 37.5, 53.8, 83.1, and 60.2 % in cucumber, eggplant, green bean, strawberry, and tomato, respectively. No fludioxonil-resistant isolates were observed. Botrytis cinerea and Botrytis group S genotypes comprised the gray mold population. B. cinerea was predominant within cucumber, eggplant and strawberry, whereas both genotypes were in equilibrium in green bean and tomato. However, Botrytis group S was found in all hosts. B. cinerea was the most prevalent in the majority of fungicide resistance phenotypes from strawberry, while genotype distributions within tomato were generally more balanced. B. pseudocinerea was not detected in the sampled population. Overall, frequency of the mating type allele MAT1–1 was higher to MAT1–2, underlying their unequal distribution in the population. However, cases of 1:1 distribution were apparent within particular subpopulations, suggesting that mating in the field cannot be excluded.



Similar content being viewed by others
References
Adjebli, A., Leyronas, C., Aissat, K., & Nicot, P. C. (2015). Comparison of Botrytis cinerea populations collected from tomato greenhouses in northern Algeria. . Journal of Phytopathology, 163, 124–132.
Amiri, A., Heath, S. M., & Peres, N. A. (2013). Phenotypic characterization of multidrug resistance in Botrytis cinerea isolates from strawberry in Florida. Plant Disease, 97, 393–410.
Angelini, d. R. M. M., Rotolo, C., Pollastro, S., & Faretra, F. (2016). Molecular analysis of the mating type (MAT1) locus in strains of the heterothallic ascomycete Botrytis cinerea. Plant Pathology, (Early view). doi:10.111/ppa.12509.
Banno, S., Fukumori, F., Ichiishi, A., Okada, K., Uekusa, H., Kimura, M., et al. (2008). Genotyping of benzimidazole-resistant and dicarboximide-resistant mutations in Botrytis cinerea using real-time polymerase chain reaction assays. Phytopathology, 98, 397–404.
Brent, K. J., & Hollomon, D. W. (1998). Fungicide resistance: the assessment of risk. FRAC Monograph No. 2 (pp 1–48). Global crop protection federation, Brussels, Belgium.
Cary, J. W., Ehrlich, K. C., Beltz, S. B., Harris-Coward, P., & Klich, M. A. (2009). Characterization of the Aspergillus ochraceoroseus aflatoxin/sterigmatocystin biosynthetic gene cluster. Mycologia, 101, 352–362.
Castilla, N., & Hernandez, J. (2005). The plastic greenhouse industry of Spain. Chronica Horticulturae, 45, 15–20.
Chatzidimopoulos, M., Papaevaggelou, D., & Pappas, A. C. (2013). Detection and characterization of fungicide resistant phenotypes of Botrytis cinerea in lettuce crops in Greece. European Journal of Plant Pathology, 137, 363–376.
Delcan, J., & Melgarejo, P. (2002). Mating behaviour and vegetative compatibility in Spanish populations of Botryotinia fuckeliana. European Journal of Plant Pathology, 108, 391–400.
Fernández-Ortuño, D., Chen, F., & Schnabel, G. (2012). Resistance to pyraclostrobin and boscalid in Botrytis cinerea isolates from strawberry fields in the Carolinas. Plant Disease, 96, 1198–1203.
Fernández-Ortuño, D., Chen, F., & Schnabel, G. (2013). Resistance to cyprodinil and lack of fludioxonil resistance in Botrytis cinerea isolates from strawberry in North and South Carolina. Plant Disease,, 97, 81–85.
Fernández-Ortuño, D., Grabke, A., Bryson, P. K., Amiri, A., Peres, N. A., & Schnabel, G. (2014). Fungicide resistance profiles in Botrytis cinerea from strawberry fields of seven Southern U.S. states. Plant Disease, 98, 825–833.
Fernández-Ortuño, D., Grabke, A., Li, X., & Schnabel, G. (2015). Independent emergence of resistance to seven chemical classes of fungicides in Botrytis cinerea. Phytopathology, 105, 424–432.
Fournier, E., Levis, C., Fortini, D., Leroux, P., Giraud, T., & Brygoo, Y. (2003). Characterization of Bc-hch, the Botrytis cinerea homolog of the Neurospora crassa het-c vegetative incompatibility locus, and its use as a population marker. Mycologia, 95, 251–261.
Fournier, E., Giraud, T., Albertini, C., & Brygoo, Y. (2005). Partition of the Botrytis cinerea complex in France using multiple gene genealogies. Mycologia, 97, 1251–1267.
Giraud, T., Fortini, D., Levis, C., Leroux, P., & Brygoo, Y. (1997). RFLP markers show genetic recombination in Botrytotinia fuckeliana (Botrytis cinerea) and transposable elements reveal two sympatric species. Molecular Biology and Evolution, 14, 1177–1185.
Grabke, A., & Stammler, G. (2015). A Botrytis cinerea population from a single strawberry field in Germany has a complex fungicide resistance pattern. Plant Disease, 99, 1078–1086.
Grabke, A., Fernández-Ortuño, D., & Schnabel, G. (2013). Fenhexamid resistance in Botrytis cinerea from strawberry fields in the Carolinas is associated with four target gene mutations. Plant Disease, 97, 271–276.
Grabke, A., Fernández-Ortuño, D., Amiri, A., Li, X., Peres, N. A., Smith, P., et al. (2014). Characterization of iprodione resistance in Botrytis cinerea from strawberry and blackberry. Phytopathology, 104, 396–402.
Hyde, K. D., Nilsson, R. H., Alias, S. A., Ariyawansa, H. A., Blair, J. E., Cai, L., et al. (2014). One stop shop: backbones trees for important phytopathogenic genera: I (2014. Fungal Diversity, 67, 21–125.
Johnston, P. R., Hoksbergen, K., Park, D., & Beever, R. E. (2014). Genetic diversity of Botrytis in New Zealand vineyards and the significance of its seasonal and regional variation. Plant Pathology, 63, 888–898.
Kanetis, L., Förster, H., Jones, C. A., Borkovich, K. A., & Adaskaveg, J. E. (2008). Characterization of genetic and biochemical mechanisms of fludioxonil and pyrimethanil resistance in field isolates of Penicillium digitatum. Phytopathology, 98, 205–214.
Kanetis, L., Förster, H., & Adaskaveg, J. E. (2010). Determination of natural resistance frequencies in Penicillium digitatum using a new air-sampling method and characterization of fludioxonil- and pyrimethanil resistant isolates. Phytopathology, 100, 38–746.
Karchani-Balma, S., Gautier, A., Raies, A., & Fournier, E. (2008). Geography, plants, and growing systems shape the genetic structure of Tunisian Botrytis cinerea populations. Phytopathology, 98, 1271–1279.
Kim, Y. K., & Xiao, C. L. (2011). Stability and fitness of pyraclostrobin- and boscalid-resistant phenotypes in field isolates of Botrytis cinerea from apple. Phytopathology,, 101, 1385–1391.
Konstantinou, S., Veloukas, T., Leroch, M., Menexes, G., Hahn, M., & Karaoglanidis, G. (2015). Population structure, fungicide resistance profile, and sdhB mutation frequency of Botrytis cinerea from strawberry and greenhouse-grown tomato in Greece. Plant Disease, 99, 240–248.
Kretschmer, M., & Hahn, M. (2008). Fungicide resistance and genetic diversity of Botrytis cinerea isolates from a vineyard in Germany. Journal of Plant Diseases and Protection, 115, 214–219.
Kretschmer, M., Leroch, M., Mosbach, A., Walker, A.-S., Fillinger, S., et al. (2009). Fungicide-driven evolution and molecular basis of multidrug resistance in field populations of the grey mould fungus Botrytis cinerea. PLoS Pathogens, 5, e1000696. doi:10.1371/joumal.ppat.l 000696.
Le Gac, M., Hood, M. E., Fournier, E., & Giraud, T. (2007). Phylogenetic evidence of host-specific cryptic species in the anther smut fungus. Evolution, 61, 15–26.
Leroch, M., Kretschmer, M., & Hahn, M. (2011). Fungicide resistance phenotypes of Botrytis cinerea isolates from commercial vineyards in South Germany. Journal of Phytopathology,, 159, 63–65..
Leroch, M., Plesken, C., Weber, R. W. S., Kauff, F., Scalliet, G., & Hahn, M. (2013). Gray mold populations in German strawberry fields are resistant to multiple fungicides and dominated by a novel clade closely related to Botrytis cinerea. Applied and Environmental Microbiology, 79, 159–167.
Leroux, P., Gredt, M., Leroch, M., & Walker, A.-S. (2010). Exploring mechanisms of resistance to respiratory inhibitors in field strains of Botrytis cinerea, the causal agent of gray mold. Applied and Environmental Microbiology, 76, 6615–6630.
Li, X., Fernández-Ortuño, D., Grabke, A., & Schnabel, G. (2014). Resistance to fludioxonil in Botrytis cinerea isolates from blackberry and strawberry. Phytopathology, 104, 724–732.
Li, N., Zhang, J., Yang, L., Wu, M. D., & Li, G. Q. (2015). First report of Botrytis pseudocinerea causing gray mold on tomato (Lycopersicon esculentum) in central China. Plant Disease, 99, 283.
Milgroom, M. G. (1996). Recombination and the multilocus structure of fungal populations. Annual Review of Phytopathology, 34, 457–477.
Moorman, G. W., & Lease, R. J. (1992). Benzimidazole- and dicarboximide-resistant Botrytis cinerea from Pennsylvania greenhouses. Plant Disease, 76, 477–480.
Moyano, C., Gomez, V., & Melgarejo, P. (2004). Resistance to pyrimethanil and other fungicides in Botrytis cinerea populations collected on vegetable crops in Spain. Journal of Phytopathology, 152, 484–490.
Myresiotis, C. K., Karaoglanidis, G. S., & Tzavella-Klonari, K. (2007). Resistance of Botrytis cinerea isolates from vegetable crops to anilinopyrimidine, phenylpyrrole, hydroxyanilide, benzimidazole, and dicarboximide fungicides. Plant Disease, 91, 407–413.
Plesken, C., Weber, R. W. S., Rupp, S., Leroch, M., & Hahn, M. (2015). Botrytis pseudocinerea is a significant pathogen of several crop plants but susceptible to displacement by fungicide-resistant B. cinerea strains. Applied and Environmental Microbiology, 81, 7048–7056.
Saito, S., Michailides, T. J., & Xiao, C. L. (2014). First report of Botrytis pseudocinerea causing gray mold on blueberry in North America. Plant Disease, 98, 1743.
Samuel, S., Papayiannis, L. C., Leroch, M., Veloukas, T., Hahn, M., & Karaoglanidis, G. S. (2011). Evaluation of the incidence of the G143 A mutation and cytb intron presence in the cytochrome bc-1 gene conferring QoI resistance in Botrytis cinerea populations from several hosts. Pest Management Science, 67, 1029–1036.
Staats, M., van Baarlen, P., & van Kan, J. A. L. (2005). Molecular phylogeny of the plant pathogenic genus Botrytis and the evolution of host specificity. Molecular Biology and Evolution, 22, 333–346.
Tuite, J. (1969). Plant Pathological Methods. Burgess Publishing Company, Minneapolis, MN, USA.
Veloukas, T., Markoglou, A. N., & Karaoglanidis, S. (2013). Differential effect of sdhB gene mutations of the sensitivity to SDHI fungicides in Botrytis cinerea. Plant Disease, 97, 118–122.
Walker, A.-S., Gautier, A., Confais, J., Martinho, D., Viaud, M., Le Pecheur, P., et al. (2011). Botrytis pseudocinerea, a new cryptic species causing gray mold in French vineyards in sympatry with Botrytis cinerea. Phytopathology, 101, 1433–1445.
Walker, A.-S., Gladieux, P., Decognet, V., Fermaud, M., Confais, J., Roudet, J., et al. (2015). Population structure and temporal maintenance of the multihost fungal pathogen Botrytis cinerea: causes and implications for disease management. Environmental Microbiology, 17, 1261–1274.
Weber, R. W. S. (2011). Resistance of Botrytis cinerea to multiple fungicides in northern German small-fruit production. Plant Disease, 95, 1263–1269.
Weber, R. W. S., & Hahn, M. (2011). A rapid simple method for determining fungicide resistance in Botrytis. Journal of Plant Diseases and Protection, 118, 17–25.
Wessels, B. A., Lamprecht, S. C., Linde, C. C., Fourie, P. H., & Mostert, L. (2013). Characterization of the genetic variation and fungicide resistance in Botrytis cinerea populations on rooibos seedlings in the Western Cape of South Africa. European Journal of Plant Pathology, 136, 407–417.
Yourman, L. F., & Jeffers, S. N. (1999). Resistance to benzimidazole and dicarboximide fungicides in greenhouse isolates of Botrytis cinerea. Plant Disease, 83, 569––575.
Acknowledgments
We are grateful to the agronomists of the Department of Agriculture, Ammochostos District Office, Mr. G. Hadjikonstantis, Mr. C. Hadjipetros and Mrs. T. Konstantinou for their contribution in the sampling process.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kanetis, L., Christodoulou, S. & Iacovides, T. Fungicide resistance profile and genetic structure of Botrytis cinerea from greenhouse crops in Cyprus. Eur J Plant Pathol 147, 527–540 (2017). https://doi.org/10.1007/s10658-016-1020-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-1020-9