Abstract
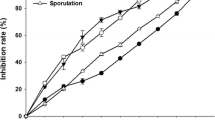
Four carboxylic acid amide (CAA) fungicides, mandipropamid (MPD), dimethomorph (DMM), benthiavalicarb (BENT) and iprovalicarb (IPRO) were examined for their effects on various developmental stages of Bremia lactucae, the causal agent of downy mildew in lettuce, in vitro and in planta. Spore germination in vitro or on leaf surfaces was inhibited by all CAA fungicides (technical or formulated). MPD was more effective in suppressing germination than DMM or BENT, whereas IPRO was least effective. CAA induced no disruption of F-actin microfilament organisation in germinating spores of B. lactucae. CAA applied to germinating spores in vitro prevented further extension of the germ tubes. When applied to germinated spores on the leaf surface they prevented penetration. Preventive application of CAA to intact plants inhibited infection. MPD was more effective in suppressing infection than DMM or BENT, whereas IPRO was least effective. Curative application was effective at ≤3 h post-inoculation (hpi) but not at ≥18 hpi. CAA (except IPRO) applied to upper leaf surfaces inhibited spore germination on the lower surface and hence reduced infection. CAA suppressed sporulation of B. lactucae on floating leaf discs and when sprayed onto infected plants two days before onset of sporulation. BENT and DMM were more effective in suppressing sporulation than MPD or IPRO. Epidemics of downy mildew in shade-house grown lettuce were suppressed by CAA. A single spray applied to five-leaf plants before transplanting controlled the disease for 50 days. The results suggest that CAA are effective inhibitors of spore germination and therefore should be used as preventive agents against downy mildew of lettuce caused by B. lactucae.











Similar content being viewed by others
References
Albert, G., Curtze, J., & Drandarevski, C. (1988). Dimethomorph (CME 151), a novel curative fungus fungicide. Brighton Crop Protection Conference—Pests and Diseases, pp. 17–22.
Albert, G., Thomas, A., & Guehne, M. (1991). Fungicidal activity of dimethomorph on different stages in the life cycle of Phytophthora infestans and Plasmopara viticola. In Third International Conference on Plant Diseases, Bordeaux pp. 887–894. Paris, France: ANPP.
Anon. (2006). http://www.frac.info/work/work_CAA.htm.
Bagirova, S. F., Li, A. Z., Dolgova, A. V., Elansky, S. N., Shaw, D. S., & Dyakov, Y. T. (2001). Mutants of Phytophthora infestans resistant to dimethomorph fungicide. Journal of Russian Phytopathology, 2, 19–24.
Cohen, Y., Baider, A., & Cohen, B. H. (1995). Dimethomorph activity against oomycete fungal plant pathogens. Phytopathology, 85, 1500–1506.
Cohen, Y., Eyal, H., & Hanania, J. (1990). Ultrastructure, autofluorescence, callose deposition and lignification in susceptible and resistant muskmelon leaves infected with the powdery mildew fungus Sphaerotheca fuliginea. Physiological and Molecular Plant Pathology, 36, 191–204.
Cohen, Y., Eyal, H., Hanania, J., & Malik, Z. (1989). Ultrastructure of Pseudoperonospora cubensis in muskmelon genotype susceptible and resistant to downy mildew. Physiological and Molecular Plant Pathology, 34, 27–40.
Cohen, Y., & Gisi, U. (1996). Systemic translocation of 14C-DL-3-amino-n-butanoic acids. Physiological and Molecular Plant Pathology, 45, 441–456.
Cohen, Y., & Gisi, U. (2007). Differential activity of carboxylic acid amides fungicides against various developmental stages of Phytophthora infestans. Phytopathology, 97, 1274–1283.
Dereviagina, M. K., Elanskij, S. N., & Diakov, Y. T. (1999). Resistance of Phytophthora infestans to the dimethomorph fungicide. Mikologiya I Fitopatologiya, 33, 208–213.
Gisi, U., Waldner, M., Kraus, N., Dubuis, P. H., & Sierotzki, H. (2006). Inheritance of resistance to carboxylic acid amide (CAA) fungicides in Plasmopara viticola. Plant Pathology, 56, 199–208.
Griffiths, R. G., Dancer, J., O’Neill, E., & Harwood, J. L. (2003). A mandelamide pesticide alters lipid metabolism in Phytophthora infestans. New Phytologist, 158, 345–353.
Harp, T., Cloud, G., Minton, B., & Cochran, A. (2006). Mandipropamid: A new fungicide for control of late blight and downy mildews. Phytopathology, 96, S185.
Harp, T., Cochran, A., Tory, D., Kuhn, P., Payan, L., Laird, D., et al. (2007). Development of Revus 2.09SC (a.i. mandipropamid) in the U.S. for control of downy mildew on leafy vegetables. Phytopathology, 97, S45.
Hermann, D., Bartlett, D. W., Fischer, W., & Kempf, H. J. (2005). The behaviour of mandipropamid on and in plants. Proceedings of the BCPC International Congress of Crop Science & Technology, Glasgow, UK, 1, 87–92.
Huggenberger, F., & Kuhn, P. J. (2006). Biological and physico-chemical properties of mandipropamid, a new fungicide for the control of oomycete pathogens. Phytopathology, 96, S51.
Huggenberger, F., Lamberth, C., & Iwanzik, W. (2005). Mandipropamid a new fungicide against Oomycete pathogens. Proceedings of the BCPC International Congress Crop Science & Technology, Glasgow, UK, 1, 93–98.
Jende, G., Steiner, U., & Dehne, H. -W. (1999). Effects of iprovalicarb (SZX 0722) on the development of Phytophthora infestans in tomato plants. Pflanzenchutz-Nachrichten Bayer, 52, 49–60.
Jende, G., Steiner, U., & Dehne, H.-W. (2001). Microscopical characterization of fungicidal effects on infection structures and cell wall formation of Phytophthora infestans. In H. W. Dehne, U. Gisi, K. H. Kuck, P. E. Russell, & H. Lyr (Eds.), Modern fungicides and antifungal compounds (pp. 83–90). Bonn: AgroConcept.
Lamberth, C., Cederbaum, F., Jeanguenat, A., Kempf, H. J., Zeller, M., & Zeun, R. (2006). Synthesis and fungicidal activity of N-2-(3-methoxy-4-propagyloxy) phenylethyl amides. Part II: Anti-oomycetic mandelamides. Pest Management Science, 62, 446–451.
Matheron, M. E., & Porchas, M. (2000). Impact of azoxystrobin, dimethomorph, fluazinam, fosetyl-Al, and metalaxyl on growth, sporulation, and zoospore cyst germination of three Phytophthora spp. Plant Disease, 84, 454–458.
Miyake, Y., Sakai, J., Miura, I., Nagayama, K., & Shibata, M. (2003). Effects of a novel fungicide benthiavalicarb-isopropyl against oomycete fungal diseases. The BCPC International Congress of Crop Science and Technology, Glasgow, Scotland, UK, Nov. 2003.
Reuveni, M. (2003). Activity of the new fungicide benthiavalicarb against Plasmopara viticola and its efficacy in controlling downy mildew in grapevines. European Journal of Plant Pathology, 109, 243–251.
Sharaf, K., Lewinsohn, D., Nevo, E., & Beharav, A. (2007). Virulence patterns of Bremia lactucae in Israel. Phytoparasitica, 35, 100–108.
Stenzel, K., Pontzen, R., Seitz, T., Tiemann, R., & Witzenberger, A. (1998). SZX 722: A novel systemic oomycete fungicide. Brighton Crop Protection Conference, pp. 367–374.
Young, D. H., Kemmit, G. M., & Owen, J. (2005). A comparative study of XR-539 and other oomycete fungicides: Similarity to dimethomorph and amino acid amides in its mechanism of action. In H. W. Dehne, U. Gisi, K. H. Kuch, P. E. Russell, & H. Lyr (Eds.), Modern fungicides and antifungal compounds IV (pp. 145–152). Alton: BCPC.
Zhu, S. S., Liu, X. L., Liu, P. F., Li, Y., Li, J. Q., Wang, H. M., et al. (2007). Flumorph is a novel fungicide that disrupts microfilament organization in Phytophthora melonis. Phytopathology, 97, 643–649.
Acknowledgement
We are grateful to Dr. Alex Perelman of Bar-Ilan University for his assistance with confocal microscopy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cohen, Y., Rubin, A.(. & Gotlieb, D. Activity of carboxylic acid amide (CAA) fungicides against Bremia lactucae . Eur J Plant Pathol 122, 169–183 (2008). https://doi.org/10.1007/s10658-008-9327-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-008-9327-9