Abstract
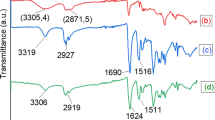
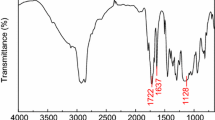
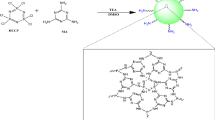
Polymeric microspheres with N-methyl-d-glucamine (NMDG) ligands have been tested in the adsorption–membrane filtration process for boron removal from aqueous solutions. The chelating resins were synthesized by reacting NMDG with the vinylbenzyl chloride–styrene–1,4-divinylbenzene (VBC/S/DVB) copolymer at the reflux temperature and in the microwave reactor. VBC/S/DVB spheres with a gel structure that contained 6 wt% DVB were obtained by membrane emulsification followed by suspension polymerization. By selecting the optimal emulsification and polymerization parameters, it was possible to obtain 25-μm-diameter particles with a narrow size distribution. Resins obtained by microwave modification showed the higher boron adsorption capacity.




Similar content being viewed by others
References
Badruk, M., Kabay, N., Demircioglu, M., Mordogan, H., & Ipekoglu, U. (1994). Removal of boron from wastewater of geothermal power plant by selective ion-exchange resin. I. Batch sorption-elution studies. Separation Science and Technology, 34, 2553–2569.
Bryjak, M., Wolska, J., & Kabay, N. (2008). Removal of boron from seawater by adsorption-membrane hybrid process: Implementation and challenges. Desalination, 223, 57–62.
Cakmak, I., & Römheld, V. (1997). Boron deficiency-induced impairments of cellular functions in plants. Plant and Soil, 193, 71–83.
Kabay, N., Yilmaz, I., Bryjak, M., & Yuksel, M. (2006). Removal of boron from aqueous solutions by a hybrid Ion exchange-membrane process. Desalination, 198, 158–165.
Melnyk, L., Goncharuk, V., Butnyk, I., & Tsapiuk, E. (2005). Boron removal from natural and wastewaters using combined sorption/membrane process. Desalination, 185, 147–157.
Rainey, C. J., Nyquist, L. A., Christensen, R. E., Strong, P. L., Dwight Culver, B., & Coughlin, J. R. (1999). Daily boron intake from the American diet. Journal of the American Dietetic Association, 99, 335–340.
Sah, R. N., & Brown, P. H. (1997). Techniques for boron determination and their application to the analysis of plant and soil samples. Plant and Soil, 193, 15–33.
Simonnat, M. O., Castel, C., Nicolai, M., Rosin, C., Sardin, M., & Jauffret, H. (2000). Boron removal from drinking water with a boron selective resin: Is the treatment really selective? Water Research, 34, 109–116.
Şimşek, A., Korkmaz, D., Velioğlu, Y. S., & Ataman, O. Y. (2003). Determination of boron in hazelnut (Corylus avellana L.) varieties by inductively coupled plasma optical emission spectrometry and spectrophotometry. Food Chemistry, 83, 293–296.
Sprague, R. W. (1972). The ecological significance of boron. Anaheim CA: US Borax Research Corporation.
Wolska, J. (2008). Membrane emulsification as a method for polymeric microspheres obtained. PhD thesis. Wroclaw University of Technology, Wroclaw.
Wyness, A. J., Parkman, R. H., & Neal, C. (2003). A summary of boron surface water quality data throughout the European Union. The Science of the total environment, 314–316, 255–269.
Yilmaz, I., Kabay, N., Bryjak, M., Yuksel, M., Wolska, J., & Koltuniewicz, A. (2006). A submerge membrane-ion-exchange hybrid process for boron removal. Desaliantion, 198, 310–315.
Zhu, X., & Alexandratos, S. D. (2005). Affinity and selectivity of immobilized N-methyl-d-glucamine for mercury (II) ions. Industrial & Engineering Chemistry Research, 44(19), 7490–7495.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wolska, J., Bryjak, M. & Kabay, N. Polymeric microspheres with N-methyl-d-glucamine ligands for boron removal from water solution by adsorption–membrane filtration process. Environ Geochem Health 32, 349–352 (2010). https://doi.org/10.1007/s10653-010-9291-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-010-9291-0