Summary
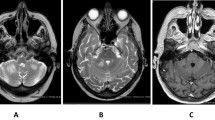
Immune checkpoint inhibitors (ICIs) have improved the overall survival of many patients with advanced cancers. However, unlike cytotoxic and targeted drugs, ICIs may cause various immune-related adverse events (irAEs). Among these irAEs, autoimmune meningitis is very rare. Here, we report a case of early-onset, atezolizumab-induced meningitis after administration of one dose of atezolizumab. A 56-year-old man with lung adenocarcinoma had received seventh-line treatment with atezolizumab when he experienced dysarthria. Blood examinations, including the measurement of electrolytes, glucose, and organ functions, were unremarkable, but enhanced head magnetic resonance imaging T1-weighted images showed meningeal enhancement. Although cerebral spinal fluid (CSF) examinations revealed elevated lymphocyte and protein levels, no cancer cells were detected in the CSF. CSF cultures and serological tests, including polymerase chain reaction for herpes simplex virus, were negative. The patient was therefore diagnosed with atezolizumab-triggered autoimmune meningitis. With steroid treatment, the patient’s clinical and neurological state improved immediately and he recovered to baseline conditions. Prompt diagnosis and therapeutic intervention are essential for the effective treatment of autoimmune meningitis.

Similar content being viewed by others
References
Rittmeyer A, Barlesi F, Waterkamp D et al (2017) Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet 389:255–265
Gordon R, Kasler MK, Stasi K et al (2017) Checkpoint Inhibitors: Common Immune-Related Adverse Events and Their Management. Clin J Oncol Nurs 21:45–52
Johnson DB, Manouchehri A, Haugh AM et al (2019) Neurologic toxicity associated with immune checkpoint inhibitors: a pharmacovigilance study. J Immunother Cancer 7:134
Sato K, Mano T, Iwata A et al (2019) Neurological and related adverse events in immune checkpoint inhibitors: a pharmacovigilance study from the Japanese Adverse Drug Event Report database. J Neurooncol 145:1–9
Brahmer J, Reckamp KL, Baas P et al (2015) Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N Engl J Med 373:123–135
Borghaei H, Paz-Ares L, Horn L et al (2015) Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N Engl J Med 373:1627–1639
Ramchandren R, Domingo-Domenech E, Rueda A et al: Nivolumab for Newly Diagnosed Advanced-Stage Classic Hodgkin Lymphoma: Safety and Efficacy in the Phase II CheckMate 205 Study. J Clin Oncol:Jco1900315, 2019
Ribas A, Puzanov I, Dummer R et al (2015) Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol 16:908–918
Schachter J, Ribas A, Long GV et al (2017) Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet 390:1853–1862
Reck M, Rodriguez-Abreu D, Robinson AG et al (2016) Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N Engl J Med 375:1823–1833
Mok TSK, Wu YL, Kudaba I et al (2019) Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 393:1819–1830
Chung HC, Ros W, Delord JP et al (2019) Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Cervical Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol 37:1470–1478
Marabelle A, Le DT, Ascierto PA et al: Efficacy of Pembrolizumab in Patients With Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol:Jco1902105, 2019
Le DT, Kim TW, Van Cutsem E et al: Phase II Open-Label Study of Pembrolizumab in Treatment-Refractory, Microsatellite Instability-High/Mismatch Repair-Deficient Metastatic Colorectal Cancer: KEYNOTE-164. J Clin Oncol:Jco1902107, 2019
Antonia SJ, Villegas A, Daniel D et al (2017) Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N Engl J Med 377:1919–1929
Garassino MC, Cho BC, Kim JH et al (2018) Durvalumab as third-line or later treatment for advanced non-small-cell lung cancer (ATLANTIC): an open-label, single-arm, phase 2 study. Lancet Oncol 19:521–536
Motzer RJ, Tannir NM, McDermott DF et al (2018) Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J Med 378:1277–1290
Wolchok JD, Chiarion-Sileni V, Gonzalez R et al (2017) Overall Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N Engl J Med 377:1345–1356
Paz-Ares L, Luft A, Vicente D et al (2018) Pembrolizumab plus Chemotherapy for Squamous Non-Small-Cell Lung Cancer. N Engl J Med 379:2040–2051
Gandhi L, Rodriguez-Abreu D, Gadgeel S et al (2018) Pembrolizumab plus Chemotherapy in Metastatic Non-Small-Cell Lung Cancer. N Engl J Med 378:2078–2092
Socinski MA, Jotte RM, Cappuzzo F et al (2018) Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N Engl J Med 378:2288–2301
Levine JJ, Somer RA, Hosoya H et al (2017) Atezolizumab-induced Encephalitis in Metastatic Bladder Cancer: A Case Report and Review of the Literature. Clin Genitourin Cancer 15:e847–e849
Arakawa M, Yamazaki M, Toda Y et al (2019) Atezolizumab-induced encephalitis in metastatic lung cancer: a case report and literature review. eNeurologicalSci 14:49–50
Laserna A, Tummala S, Patel N et al: Atezolizumab-related encephalitis in the intensive care unit: Case report and review of the literature, SAGE Open Med Case Rep. England, 2018, pp 2050313 × 18792422
Brahmer JR, Lacchetti C, Schneider BJ et al (2018) Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol 36:1714–1768
Astaras C, de Micheli R, Moura B et al (2018) Neurological Adverse Events Associated with Immune Checkpoint Inhibitors: Diagnosis and Management. Curr Neurol Neurosci Rep 18:3
Kim K, Ramiya N, Krajewski K et al (2013) Ipilimumab associated hepatitis: imaging and clinicopathologic findings. Invest New Drug 31:1071–1077
Beck KE, Blansfield JA, Tran KQ et al (2006) Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol 24:2283–2289
Johnson DB, McDonnell WJ, Gonzalez-Ericsson PI et al (2019) A case report of clonal EBV-like memory CD4(+) T cell activation in fatal checkpoint inhibitor-induced encephalitis. Nat Med 25:1243–1250
Author information
Authors and Affiliations
Contributions
Koichi Ogawa produced the first draft of the paper.; Koichi Ogawa, Hiroyasu Kaneda, Tamaki Kawamoto, Yoko Tani, Motohiro Izumi, Yoshiya Matsumoto, Kenji Sawa, Tomohiro Suzumura, Tetsuya Watanabe, Shigeki Mitsuoka, Kazuhisa Asai, and Tomoya Kawaguchi contributed and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Rights and permissions
About this article
Cite this article
Ogawa, K., Kaneda, H., Kawamoto, T. et al. Early-onset meningitis associated with atezolizumab treatment for non-small cell lung cancer: case report and literature review. Invest New Drugs 38, 1901–1905 (2020). https://doi.org/10.1007/s10637-020-00947-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-020-00947-w