Summary
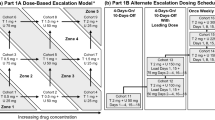
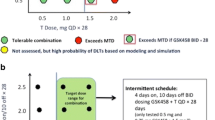
Aim AZD8931 is an oral equipotent inhibitor of EGFR (erbB1), HER2 (erbB2) and HER3 (erbB3) signaling. This Phase I, open-label study evaluated the safety, tolerability, and pharmacokinetics of multiple ascending doses of AZD8931 in patients with advanced solid tumors (NCT00637039). Methods Patients received AZD8931 as a single oral dose followed by 4 days of observation, then twice-daily dosing for 21 consecutive days. Using a standard 3 + 3 design, AZD8931 doses were escalated from 40 mg bid until the maximum tolerated dose (MTD) was established. Results Twenty-eight patients received AZD8931 (n = 5, 40 mg bid; n = 8, 80 mg bid; n = 6, 160 mg bid; n = 6, 240 mg bid; n = 3, 300 mg bid). Ovary (n = 8) and breast (n = 5) were the most common primary tumor types. The most frequent adverse events were treatment-emergent cutaneous (n = 27) and diarrhea (n = 21). Dose-limiting toxicities (DLTs) were identified in one patient in the 240 mg bid cohort (Grade 3 rash) and two patients in the 300 mg bid cohort (Grade 3 and 4 diarrhea). The pharmacokinetic profile of AZD8931 supported twice-daily dosing. AZD8931 was rapidly absorbed (median tmax 1–3 h), was well distributed and had moderate to high clearance with an elimination half-life of approximately 11 h. Exposure appeared to increase approximately proportionally with dose up to 160 mg. Of 21 patients evaluable for response at day 21, 12 had stable disease and nine had disease progression. Conclusion The MTD of AZD8931 determined from the 21-day DLT period was 240 mg bid, although more long-term data are needed to confirm a dose of AZD8931 suitable for chronic treatment.



Similar content being viewed by others
References
Alvarez JV, Greulich H, Sellers WR, Meyerson M, Frank DA (2006) Signal transducer and activator of transcription 3 is required for the oncogenic effects of non-small-cell lung cancer-associated mutations of the epidermal growth factor receptor. Cancer Res 66:3162–3168
Citri A, Yarden Y (2006) EGF-ERBB signalling: towards the systems level. Nat Rev Mol Cell Biol 7:505–516
Hubbard SR (2009) The juxtamembrane region of EGFR takes center stage. Cell 137:1181–1183
Hynes NE, Horsch K, Olayioye MA, Badache A (2001) The ErbB receptor tyrosine family as signal integrators. Endocr Relat Cancer 8:151–159
Hynes NE, Lane HA (2005) ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 5:341–354
Jorissen RN, Walker F, Pouliot N, Garrett TP, Ward CW, Burgess AW (2003) Epidermal growth factor receptor: mechanisms of activation and signalling. Exp Cell Res 284:31–53
Baselga J, Swain SM (2009) Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nat Rev Cancer 9:463–475
Ciardiello F, Tortora G (2008) EGFR antagonists in cancer treatment. N Engl J Med 358:1160–1174
DiGiovanna MP, Stern DF, Edgerton SM, Whalen SG, Moore D, Thor AD (2005) Relationship of epidermal growth factor receptor expression to ErbB-2 signaling activity and prognosis in breast cancer patients. J Clin Oncol 23:1152–1160
Nicholson RI, Gee JM, Harper ME (2001) EGFR and cancer prognosis. Eur J Cancer 37(Suppl 4):S9–S15
Salomon D, Gullick W (2001) The erbB family of receptors and their ligands: multiple targets for therapy. Signal 2:4–11
Sjogren S, Inganas M, Lindgren A, Holmberg L, Bergh J (1998) Prognostic and predictive value of c-erbB-2 overexpression in primary breast cancer, alone and in combination with other prognostic markers. J Clin Oncol 16:462–469
Hickinson DM, Klinowska T, Speake G, Vincent J, Trigwell C, Anderton J, Beck S, Marshall G, Davenport S, Callis R, Mills E, Grosios K, Smith P, Barlaam B, Wilkinson RW, Ogilvie D (2010) AZD8931, an equipotent, reversible inhibitor of signaling by epidermal growth factor receptor, ERBB2 (HER2), and ERBB3: a unique agent for simultaneous ERBB receptor blockade in cancer. Clin Cancer Res 16:1159–1169
Kosaka T, Yamaki E, Mogi A, Kuwano H (2011) Mechanisms of resistance to EGFR TKIs and development of a new generation of drugs in non-small-cell lung cancer. J Biomed Biotechnol 2011:165214
Vokes EE, Chu E (2006) Anti-EGFR therapies: clinical experience in colorectal, lung, and head and neck cancers. Oncology (Williston Park) 20:15–25
Medina PJ, Goodin S (2008) Lapatinib: a dual inhibitor of human epidermal growth factor receptor tyrosine kinases. Clin Ther 30:1426–1447
Cully M, You H, Levine AJ, Mak TW (2006) Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer 6:184–192
Kapitanovic S, Radosevic S, Slade N, Kapitanovic M, Andelinovic S, Ferencic Z, Tavassoli M, Spaventi S, Pavelic K, Spaventi R (2000) Expression of erbB-3 protein in colorectal adenocarcinoma: correlation with poor survival. J Cancer Res Clin Oncol 126:205–211
Tanner B, Hasenclever D, Stern K, Schormann W, Bezler M, Hermes M, Brulport M, Bauer A, Schiffer IB, Gebhard S, Schmidt M, Steiner E, Sehouli J, Edelmann J, Lauter J, Lessig R, Krishnamurthi K, Ullrich A, Hengstler JG (2006) ErbB-3 predicts survival in ovarian cancer. J Clin Oncol 24:4317–4323
Yi ES, Harclerode D, Gondo M, Stephenson M, Brown RW, Younes M, Cagle PT (1997) High c-erbB-3 protein expression is associated with shorter survival in advanced non-small cell lung carcinomas. Mod Pathol 10:142–148
Engelman JA, Cantley LC (2006) The role of the ErbB family members in non-small cell lung cancers sensitive to epidermal growth factor receptor kinase inhibitors. Clin Cancer Res 12:4372s–4376s
Engelman JA, Zejnullahu K, Mitsudomi T, Song Y, Hyland C, Park JO, Lindeman N, Gale CM, Zhao X, Christensen J, Kosaka T, Holmes AJ, Rogers AM, Cappuzzo F, Mok T, Lee C, Johnson BE, Cantley LC, Janne PA (2007) MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 316:1039–1043
Lee-Hoeflich ST, Crocker L, Yao E, Pham T, Munroe X, Hoeflich KP, Sliwkowski MX, Stern HM (2008) A central role for HER3 in HER2-amplified breast cancer: implications for targeted therapy. Cancer Res 68:5878–5887
Zhang XL, Yang YS, Xu DP, Qu JH, Guo MZ, Gong Y, Huang J (2009) Comparative study on overexpression of HER2/neu and HER3 in gastric cancer. World J Surg 33:2112–2118
AstraZeneca (2011) Global policy: bioethics. Available at: http://www.astrazeneca.com/Responsibility/Code-policies-standards/Our-global-policies
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Lopez Martin J, Vidal Losada M, Cortes J, Bermejo B, Lluch Fernandez A, Learoyd M, Saunders A, Stuart M, Baselga J (2011) Phase I, dose-finding study of AZD8931, an inhibitor of ErbB1, 2 and 3 receptor signaling, in combination with paclitaxel (P). J Clin Oncol 29(15S):abst 3105
Perez-Soler R (2003) Can rash associated with HER1/EGFR inhibition be used as a marker of treatment outcome? Oncology (Huntingt) 17:23–28
Fukuoka M, Yano S, Giaccone G, Tamura T, Nakagawa K, Douillard JY, Nishiwaki Y, Vansteenkiste J, Kudoh S, Rischin D, Eek R, Horai T, Noda K, Takata I, Smit E, Averbuch S, Macleod A, Feyereislova A, Dong RP, Baselga J (2003) Multi-institutional randomized phase II trial of gefitinib for previously treated patients with advanced non-small-cell lung cancer. J Clin Oncol 21:2237–2246
Kris MG, Natale RB, Herbst RS, Lynch TJ Jr, Prager D, Belani CP, Schiller JH, Kelly K, Spiridonidis H, Sandler A, Albain KS, Cella D, Wolf MK, Averbuch SD, Ochs JJ, Kay AC (2003) Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer: a randomized trial. JAMA 290:2149–2158
Forsythe B, Faulkner K (2004) Overview of the tolerability of gefitinib (IRESSA) monotherapy: clinical experience in non-small-cell lung cancer. Drug Saf 27:1081–1092
Acknowledgments
We wish to thank the patients who participated in this trial, and the study staff. We also wish to thank Quintiles Laboratories Europe, Livingston, Scotland for their analysis of biological samples.
This study was sponsored by AstraZeneca. We thank Zoё van Helmond PhD from Mudskipper Bioscience who provided medical writing support funded by AstraZeneca.
Conflict of interest disclosures
S Tjulandin has received honoraria from AstraZeneca as a lecturer. M Learoyd and M Stuart are employees of and stockholders of AstraZeneca. A Saunders is a paid consultant to AstraZeneca, as a project physician on AZD8931. All remaining authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tjulandin, S., Moiseyenko, V., Semiglazov, V. et al. Phase I, dose-finding study of AZD8931, an inhibitor of EGFR (erbB1), HER2 (erbB2) and HER3 (erbB3) signaling, in patients with advanced solid tumors. Invest New Drugs 32, 145–153 (2014). https://doi.org/10.1007/s10637-013-9963-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-013-9963-6