Summary
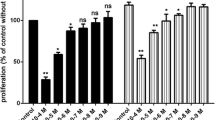
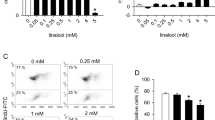
TGF-β is a multifunctional cytokine that regulates cell proliferation, differentiation, apoptosis and extracellular matrix production. Deregulation of TGF-β production or signaling has been associated with a variety of pathological processes such as cancer, metastasis, angiogenesis and fibrosis. Therefore, TGF-β signaling has emerged as an attractive target for the development of new cancer therapeutics. In a screening program of natural compounds from fungi inhibiting the TGF-β dependent expression of a reporter gene in HepG2 cells, we found that the flavone isoxanthohumol inhibited the binding of the activated Smad2/3 transcription factors to the DNA and antagonized the cellular effects of TGF-β including reporter gene activation and expression of TGF-β induced genes in HepG2 and MDA-MB-231 cells. In an in vitro angiogenesis assay, isoxanthohumol (56 μM) strongly decreased the formation of capillary-like tubules of MDA-MB-231 cells on Matrigel. In addition, we found that isoxanthohumol blocked IFN-γ, IL-4 and IL-6 dependent Jak/Stat signaling and strongly inhibited the induction of pro-inflammatory genes in MonoMac6 cells at the transcriptional level after LPS/TPA treatment.












Similar content being viewed by others
References
Siegel PM, Massague J (2003) Cytostatic and apoptotic actions of TGF-beta in homeostasis and cancer. Nat Rev Cancer 3:807–821
Li MO, Wan YY, Sanjabi S, Robertson A-KL, Flavell RA (2006) Transforming growth factor-β regulation of immune responses. Annu Rev Immunol 24:99–149
Massague J, Seoane J, Wotton D (2006) Smad transcription factors. Genes Dev 19:2783–2810
Gordon KJ, Blobe GC (2008) Role of transforming growth factor-β super family signaling pathways in human disease. Biochim Biophys Acta 1782:197–228
Feng X-H, Derynck R (2005) Specificity and versatility in TGF-β signaling through Smads. Annu Rev Cell Dev Biol 21:659–693
Moustakas A, Heldin CH (2005) Non-Smad TGF-β signals. J Cell Sci 118:3573–3584
Pardali K, Moustakas A (2007) Actions of TGF-β as tumor suppressor and pro-metastatic factor in human cancer. Biochim Biophys Acta 1775:21–62
Bierie B, Moses HL (2006) TGFβ: the molecular Jekyll and Hyde of cancer. Nat Rev Cancer 6:506–520
Leivonen S-K, Kähäri V-M (2007) Transforming growth factor-β signaling in cancer invasion and metastasis. Int J Cancer 121:2119–2124
Bertolino P, Deckers M, Lebrin F, ten Dijke P (2005) Transforming growth factor-β signal transduction in angiogenesis and vascular disorders. Chest 128:585S–590S
Wahl SM, Wen J, Moutsopoulos N (2006) TGF-β: a mobile purveyor of immune privilege. Immunol Rev 213:213–227
Yingling JM, Blanchard KL, Sawyer S (2004) Development of TGF-β signaling inhibitors for cancer therapy. Nat Rev Drug Discov 3:1011–1022
Iyer S, Wang Z-G, Akhtari M, Zhao W, Seth P (2005) Targeting TGFβ signaling for cancer therapy. Cancer Biol Ther 4:261–266
Pinkas J, Teicher BA (2006) TGF-β in cancer and as therapeutic target. Biochem Pharmacol 72:523–529
Mojzis J, Varinska L, Mojzisova G, Kostova I, Mirossay L (2008) Antiangiogenic effects of flavonoids and chalcones. Pharmacol Res 57:259–265
Gerhäuser C (2005) Beer constituents as potential cancer chemo preventive agents. Eur J Cancer 41:1941–1954
Juvvadi PR, Seshime Y, Kitamoto K (2005) Genomics reveals traces of fungal phenylpropanoid-flavonoid metabolic pathway in the filamentous fungus Aspergillus oryzae. J Microbiol 43:475–486
White TJ, Bruns T, Lee S, Taylor AW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, Inc, San Diego, pp 315–322
Stevens JF, Taylor AW, Deinzer ML (1999) Quantitative analysis of xanthohumol and related prenylflavonoids in hops and beer by liquid chromatography—tandem mass spectrometry. J Chromatogr A 832:97–107
Roehm NW, Rodgers H, Hatfield SM, Glasebrook AL (1991) An improved colorimetric assay for cell proliferation and viability utilizing the tetrazolium salt XTT. J Immunol Meth 142:257–265
Erkel G, Belahmer H, Serwe A, Anke T, Kunz H, Kolshorn H, Liermann J, Opatz T (2008) Oxacyclododecindione, a novel inhibitor of IL-4 signaling from Exserohilum rostratum. J Antibiot (Tokyo) 61:285–290
Dennler S, Itoh S, Vivien D, ten Dijke P, Huet S, Gauthier JM (1998) Direct binding of Smad3 and Smad4 to critical TGF beta-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J 17:3091–3100
Mikita T, Campbell D, Wu P, Williamson K, Schindler U (1996) Requirements for interleukin-4-induced gene expression and functional characterization of STAT6. Mol Cell Biol 16:5811–5820
Weidler M, Rether J, Anke T, Erkel G (2000) Inhibition of interleukin-6 signaling by galiellalactone. FEBS Lett 484:1–6
Rether J, Erkel G, Anke T, Sterner O (2004) Inhibition of inducible TNF-α expression by oxaspirodion, a novel spiro-compound from the ascomycete Chaetomium subspirale. Biol Chem 385:829–834
Spurrell JCL, Wiehler S, Zaheer RS, Sanders SP, Proud D (2005) Human airway epithelial cells produce IP-10 (CXCL10) in vitro and in vivo upon rhinovirus infection. Am J Physiol Lung Cell Mol Physiol 289:85–95
Ray S, Sherman CT, Lu M, Brasier AR (2002) Angiotensinogen gene expression is dependent on signal transducer and activator of transcription 3-mediated p300/cAMP response element binding protein-binding protein coactivator recruitment and histone acyltransferase activity. Mol Endocrinol 16:824–836
Pahl HL, Baeuerle PA (1995) A novel signal transduction pathway from the endoplasmatic reticulum to the nucleus is mediated by transcription factor NF-κB. EMBO J 14:2580–2588
Lokker NA, Sullivan CM, Hollenbach SJ, Israel MA, Giese NA (2002) Platelet-derived growth factor (PDGF) autocrine signaling regulates survival and mitogenic pathways in glioblastoma cells: evidence that the novel PDGF-C and PDGF-D ligands may play a role in the development of brain tumors. Cancer Res 62:3729–3735
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:2002–2007
Liao D, Johnson RS (2007) Hypoxia: a key regulator of angiogenesis in cancer. Cancer Metastasis Rev 26:281–290
Semenza GL (2010) Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 29:625–634
Melillo G (2007) Targeting hypoxia cell signaling for cancer therapy. Cancer Metastasis Rev 26:341–352
Abdollah S, Macias-Silva M, Tsukazaki T, Hayashi H, Attisano L, Wrana JL (1997) TßRI phosphorylation of Smad2 on Ser465 and Ser467 is required for Smad2-Smad4 complex formation and signaling. J Biol Chem 272:27678–27685
Souchelnytskyi S, Tamaki K, Engstrom U, Wernstedt C, ten Dijke P, Heldin CH (1997) Phosphorylation of Ser465 and Ser467 in the C terminus of Smad2 mediates interaction with Smad4 and is required for transforming growth factor-beta signaling. J Biol Chem 272:28107–28115
Liu X, Sun Y, Constantinescu SN, Karam E, Weinberg RA, Lodish HF (1997) Transforming growth factor beta-induced phosphorylation of Smad3 is required for growth inhibition and transcriptional induction in epithelial cells. Proc Natl Acad Sci USA 94:10669–10674
Chen CR, Kang Y, Massague J (2001) Defective repression of c-myc in breast cancer cells: a loss at the core of the transforming growth factor β growth arrest program. Proc Natl Acad Sci USA 98:992–999
Deng J, Grande F, Neamati N (2007) Small molecule inhibitors of Stat3 signaling pathway. Curr Cancer Drug Targets 7:91–107
Klampfer L (2006) Signal transducers and activators of transcription (STATs): novel targets of chemo preventive and chemotherapeutic drugs. Curr Cancer Drug Targets 6:107–121
Janknecht R, Wells NJ, Hunter T (1998) TGF-β-stimulated cooperation of Smad proteins with the coactivators CBP/p300. Genes Dev 12:2114–2119
Simonsson M, Kanduri M, Grönroos E, Heldin C-H, Ericsson J (2006) The DNA binding activities of Smad2 and Smad3 are regulated by coactivator-mediated acetylation. J Biol Chem 281:39870–39880
Hebenstreit D, Wirnsberger G, Horejs-Hoeck J, Duschl A (2006) Signaling mechanisms, interaction partners, and target genes of Stat6. Cytokine Growth Factor Rev 17:173–188
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454:436–444
Gaestel M, Kotlyarov A, Kracht M (2009) Targeting innate immunity protein kinase signalling in inflammation. Nat Rev Drug Discov 8:480–499
Wrzesinski SH, Wan YY, Flavell RA (2007) Transforming growth factor-β and the immune response: Implications for anticancer therapy. Clin Cancer Res 13:5262–5270
von Gersdorff G, Susztak K, Rezvani F, Bitzer M, Liang D, Boettinger EP (2000) Smad3 and Smad4 mediate transcriptional activation of the human Smad7 promoter by transforming growth factor β. J Biol Chem 275:11320–11326
Javelaud D, Mauviel A (2005) Crosstalk mechanisms between the mitogen-activated protein kinase pathways and Smad signaling downstream of TGF-β: implications for carcinogenesis. Oncogene 24:5742–5750
Zhang S, Zhang D, Sun B (2007) Vasculogenic mimicry: current status and future prospects. Cancer Lett 254:157–164
Paulis YWJ, Soetekouw PMMB, Verheul HMW, Tjan-Heijnen VCG, Griffioen AW (2010) Signalling pathways in vasculogenic mimicry. Biochim Biophys Acta 1806:18–28
Reich NC, Liu L (2006) Tracking Stat nuclear traffic. Nat Rev Immunol 6:602–612
Decker T, Kovarik P (2000) Serine phosphorylation of STATs. Oncogene 19:2628–2637
Inoue Y, Itoh Y, Abe K, Okamoto T, Daitoku H, Fukamizu A, Onozaki K, Hayashi H (2007) Smad3 is acetylated by p300/CBP to regulate its transactivation activity. Oncogene 26:500–508
Shankaranarayanan P, Chaitidis P, Kühn H, Nigam S (2001) Acetylation by histone acetyltransferase CREB-binding protein/p300 of Stat6 is required for transcriptional activation of the 15-lipoxygenase-1 gene. J Biol Chem 276:42753–42760
Yu H, Jove R (2004) The Stats of cancer-new molecular targets come of age. Nat Rev Cancer 4:97–105
Devarajan E, Huang S (2009) STAT3 as a central regulator of tumor metastases. Curr Mol Med 9:626–633
Chen Z, Han ZC (2008) Stat3: a critical transcription activator in angiogenesis. Med Res Rev 28:185–200
Berg T (2008) Signal transducers and activators of transcription as targets for small organic molecules. Chembiochem 9:2039–2044
Lin W-W, Karin M (2007) A cytokine-mediated link between innate immunity, inflammation and cancer. J Clin Invest 117:1175–1183
Aggarwal BB, Shishodia S, Sandur SK, Pandey MK, Sethi G (2006) Inflammation and cancer: how hot is the link? Biochem Pharmacol 72:1605–1621
Nakanishi C, Toi M (2005) Nuclear factor-κB inhibitors as sensitizers to anticancer drugs. Nat Rev Cancer 5:297–309
Gerhauser C, Alt A, Heiss E, Gamal-Eldeen A, Klimo K, Knauft J, Neumann I, Scherf H-R, Frank N, Bartsch H, Becker H (2002) Cancer chemo preventive activity of xanthohumol, a natural product derived from hop. Mol Cancer Ther 1:959–969
Albini A, Dell’Eva R, Vene R, Ferrari N, Buhler DR, Noonan DM, Fassina G (2006) Mechanisms of the antiangiogenic activity by the hop flavonoid xanthohumol: NF-kappaB and Akt as targets. FASEB J 20:527–529
Harikumar KB, Kunnumakkara AB, Ahn KS, Anand P, Krishnan S, Guha S, Aggarwal BB (2009) Modification of the cysteine residues in IκBα kinase and NF-κB (p65) by xanthohumol leads to suppression of NF-κB-regulated gene products and potentiation of apoptosis in leukemia cells. Blood 113:2003–2013
Vanhoecke B, Derycke L, Van Marck V, Depypere H, De Keukeleire D, Bracke M (2005) Antiinvasive effect of xanthohumol, a prenylated chalcone present in hops (Humulus lupulus L.) and beer. Int J Cancer 117:889–895
Monteiro R, Calhau C, e Silva AO, Pinheiro-Silva S, Guerreiro S, Gärtner F, Azevedo I, Soares R (2008) Xanthohumol Inhibits inflammatory factor production and angiogenesis in breast cancer xenografts. J Cell Biochem 04:1699–1707
Cho Y-C, Kim HJ, Kim Y-J, Lee KY, Choi HJ, Lee I-S, Kang BY (2008) Differential anti-inflammatory pathway by xanthohumol in IFN-γ and LPS-activated macrophages. Int Immunopharmacol 8:567–573
Bertl E, Becker H, Eicher T, Herhaus C, Kapadia G, Bartsch H, Gerhäuser C (2004) Inhibition of endothelial cell functions by novel potential cancer chemo preventive agents. Biochem Biophys Res Commun 325:287–295
Acknowledgements
This work was supported by a grant from the Stiftung Rheinland-Pfalz für Innovation. We are very thankful to Prof. H. Anke for providing the crude extracts for the screening as well as Trichoderma harzianum IBWF278b-95. We thank Prof. S. Dooley, Medical Faculty of Mannheim, for providing the (AGCCAGACA)9MLP-Luc reporter plasmid, Prof. B. Brüne, University of Frankfurt, for providing the HepG2-pH3SVL cells, and Prof. O. Sterner, University of Lund, for the structure elucidation.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Serwe, A., Rudolph, K., Anke, T. et al. Inhibition of TGF-β signaling, vasculogenic mimicry and proinflammatory gene expression by isoxanthohumol. Invest New Drugs 30, 898–915 (2012). https://doi.org/10.1007/s10637-011-9643-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-011-9643-3