Abstract
Purpose
To test a new 2-flash multifocal electroretinogram (mfERG) paradigm in glaucoma using a reduced light intensity of the m-frame flash as opposed to the global flash, as it has been suggested that this may increase the responses induced by the global flash, which has been the part of the mfERG response where most changes have been noted in glaucoma.
Methods
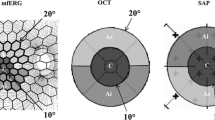
A mfERG was recorded from one eye of 22 primary open angle glaucoma (POAG) patients [16 normal tension glaucoma (NTG), 6 high tension glaucoma (HTG)] and 20 control subjects. A binary m-sequence (2^13-1, L max 100 cd/m2, L min <1 cd/m2), followed by two global flashes (L max 200 cd/m2) at an interval of 26 ms (VERIS 6.0™, FMSIII), was used. The stimulus array consisted of 103 hexagons. Retinal signals were amplified (gain = 50 K) and bandpass filtered at 1–300 Hz. For each focal response, the root mean square was calculated. We analyzed 5 larger response averages (central 15° and 4 adjoining quadrants) as well as 8 smaller response averages (central 10° and 7 surrounding response averages of approximately 7° radius each). Three epochs were analyzed: the direct component at 15–45 ms (DC) and the following two components induced by the effects of the preceding focal flash on the response to the global flashes at 45–75 ms (IC-1) and at 75–105 ms (IC-2). Statistical analysis was performed using linear mixed effects models adjusted for age.
Results
Responses differed significantly between POAG patients and controls in all central response averages. This difference was larger for the central 10° than for the response average of the central 15°. While these observations held true for all response epochs analyzed, the DC differed least and the IC-1 most when POAG was compared to control. For POAG, the most sensitive differential measure was IC-1 of the central 10° with an area under the ROC curve of 0.78. With a cutoff value of 12.52 nV/deg2, 80 % of the POAG patients (100 % HTG, 69 % NTG) were correctly classified as abnormal, while 77 % of the control subjects were correctly classified as normal. When the results of the mfERG were compared to the visual fields, there was a tendency for the mfERG to decrease as the mean defect increased. However, this correlation was only significant in the superior nasal quadrant when the IC-1 of the mfERG was compared to the corresponding area of the visual field.
Conclusion
When compared to findings from previous studies, reducing the luminance of the m-frame flash in the 2-global flash paradigm did not increase the sensitivity and specificity of the mfERG to detect glaucoma further.





Similar content being viewed by others
References
Quigley HA (2011) Glaucoma. Lancet 377:1367–1377
Heijl A, Leske MC, Hyman L, Yang Z, Bengtsson B (2010) Intraocular pressure reduction with a fixed treatment protocol in the Early Manifest Glaucoma Trial. Acta Ophthalmol 89(8):749–754
Hirneiss C, Niedermaier A, Kernt M, Kampik A, Neubauer AS (2010) Gesundheitsökonomische Aspekte des Glaukomscreenings. Ophthalmologe 107:143–149
Quigley HA, Addicks EM, Green WR (1982) Optic nerve damage in human glaucoma III. Quantitative correlation of nerve fiber loss and visual field defect in glaucoma, ischemic neuropathy, papilledema, and toxic neuropathy. Arch Ophtalmol 100(1):135–146
Kerrigan-Baumrind LA, Quigley HA, Pease ME, Kerrigan DF, Mitchell RS (2000) Number of ganglion cells in glaucoma eyes compared with threshold visual field tests in the same persons. Invest Ophthalmol Vis Sci 41(3):741–748
Hood DC, Kardon RH (2007) A framework for comparing structural and functional measures of glaucomatous damage. Prog Retin Eye Res 26(6):688–710
Palmowski AM, Allgayer R, Heinemann-Vernaleken B (2000) The multifocal ERG in open angle glaucoma—a comparison of high and low contrast recordings in high- and low-tension open angle glaucoma. Doc Ophthalmol 101(1):35–49
Palmowski AM, Ruprecht KW (2004) Follow up in open angle glaucoma. A comparison of static perimetry and the fast stimulation mfERG. Doc Ophthalmol 108:55–60
Hood DC, Greenstein VC, Holopigian K, Bauer R, Firoz B, Liebmann JM, Odel JG, Ritch R (2000) An attempt to detect glaucomatous damage to the inner retina with the multifocal ERG. Invest Ophthalmol Vis Sci 41(6):1570–1579
Palmowski AM, Allgayer R, Heinemann-Vernaleken B, Ruprecht KW (2002) Multifocal electroretinogram with a multiflash stimulation technique in open-angle glaucoma. Ophthalmic Res 34:83–89
Fortune B, Bearse MA Jr, Coiffi GA, Johnson CA (2002) Selective loss of an oscillatory component from temporal retinal multifocal ERG responses in glaucoma. Invest Ophthalmol Vis Sci 43:2638–2647
Palmowski-Wolfe AM, Todorova MG, Orguel S, Flammer J, Brigell M (2007) The two global flash mfERG in high and normal tension primary open-angle glaucoma. Doc Ophthalmol 114:9–19
Chu PH, Chan HH, Brown B (2006) Glaucoma detection is facilitated by luminance modulation of the global flash multifocal electroretinogram. Invest Ophthalmol Vis Sci 47(3):929–937
Shimada Y, Bearse MA Jr, Sutter EE (2005) Multifocal electroretinograms combined with periodic flashes: direct responses and induced components. Graefe’s Arch Clin Exp Ophthalmol 243:132–141
Hood DC, Bach M, Brigell M, Keating D, Kondo M, Lyons JS, Marmor MF, McCulloch DL, Palmowski-Wolfe AM, For the International Society For Clinical Electrophysiology of Vision (2012) ISCEV standard for clinical multifocal electroretinography (mfERG) (2011 edition). Doc Ophthalmol 124(1):1–13
Machida S, Toba Y, Ohtaki A, Gotoh Y, Kaneko M, Kurosaka D (2008) Photoptic negative response of focal electroretinograms in glaucomatous eyes. Invest Ophthalmol Vis Sci 49(12):5636–5644
Lou X, Patel NB, Harwerth RS, Frishman LJ (2011) Loss of the low-frequency component of the global-flash multifocal electroretinogram in primate eyes with experimental glaucoma. Invest Ophthalmol Vis Sci 52(6):3792–3804
R Development Core Team (2010) R, version 2.10.1, accessed on Dec 2009: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org/
Palmowski AM, Sutter EE, Bearse MA Jr, Fung W (1997) Mapping of retinal function in diabetic retinopathy using the multifocal electroretinogram. Invest Ophthalmol Vis Sci 38(12):2586–2596
Glovinsky Y, Quigley HA, Pease ME (1993) Foveal ganglion cell loss is size dependent in experimental glaucoma. Invest Ophthalmol Vis Sci 34:395–400
Kanadani FN, Hood DC, Grippo TM, Wangsupadilok B, Harizman N, Greenstein VC, Liebmann JM, Ritch R (2006) Structural and functional assessment of the macular region in patient with glaucoma. Br J Ophthalmol 90:1393–1397
Hood DC, Rasa AS, De Morales CGV, Oder JG, Greenstein VC, Liebmann JM, Ritch R (2011) Initial arcuate defects within the central 10 degrees in glaucoma. Invest Ophthalmol Vis Sci 52(2):940–946
Wang M, Hood DC, Cho HS, Ghadiali Q, De Morales CG, Zhang X, Ritch R, Liebmann JM (2009) Measurement of local retinal ganglion cell layer thickness in patients with glaucoma using frequency-domain optical coherence tomography. Arch Ophthalmol 127(7):875–881
Kaltwasser Ch, Horn FK, Kremers J, Juenemann A (2009) A comparison of the suitability of cathode ray tube (CRT) and liquid crystal display (LCD) monitors as visual stimulators in mfERG diagnostics. Doc Ophthalmol 118(3):179–189
Acknowledgments
This study was supported by the SNF (Swiss National Fund, 32003B-135219, PAM) and the LHW Stiftung Lichtenstein. The data were presented in part at ARVO 2011, Abstract number: 5482 A283.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kramer, S.A., Ledolter, A.A., Todorova, M.G. et al. The 2-global flash mfERG in glaucoma: attempting to increase sensitivity by reducing the focal flash luminance and changing filter settings. Doc Ophthalmol 126, 57–67 (2013). https://doi.org/10.1007/s10633-012-9360-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-012-9360-z