Abstract
Background
SLC9A8 has been shown to be involved in mucus layer formation, intestinal mucosal integrity, and hyperproliferation of colitis-associated tumor development. However, its effects on the epithelial–mesenchymal transition (EMT) and the metastasis of colorectal cancer (CRC) remain unknown.
Aims
To explore whether SLC9A8 participates in EMT and the metastasis of CRC.
Methods
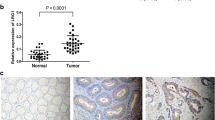
Western blotting and immunohistochemistry were performed to evaluate the expression of SLC9A8 in CRC patients. At the cellular level, the effect of SLC9A8 on proliferation, migration, and invasion was measured using cell viability analysis, flow cytometry analysis, and Transwell assays. Mouse tumor xenograft and metastasis models were established to analyze whether knockdown of SLC9A8 increased tumor volume, tumor weight, and metastasis. Moreover, whether downregulated expression of SLC9A8 promotes EMT via activation of the IL6-JAK1-STAT3 signaling pathway was investigated.
Results
SLC9A8 protein was downregulated in CRC tissues, and this downregulation was significantly associated with tumor size, lymph node status, pTNM stage, and poor prognosis. SLC9A8 overexpression markedly suppressed cell proliferation, migration, and invasion. Downregulation of SLC9A8 promoted CRC cell proliferation, migration, and invasion. Moreover, knockdown of SLC9A8 also increased tumor volume, tumor weight, and metastasis in vivo. Meanwhile, downregulation of SLC9A8 significantly promoted the in vitro migration of CRC cells via EMT by activating the IL6-JAK1/STAT3 signaling pathway.
Conclusions
Downregulation of SLC9A8 plays an important role in EMT and metastasis of CRC progression and may become a new potential therapeutic target for the treatment of CRC.






Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Paauwe M, Schoonderwoerd MJA, Helderman R et al. Endoglin Expression on Cancer-Associated Fibroblasts Regulates Invasion and Stimulates Colorectal Cancer Metastasis. Clin Cancer Res. 2018;24:6331–6344.
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. The Lancet. 2019;394:1467–1480.
Donowitz M, Ming Tse C, Fuster D. SLC9/NHE gene family, a plasma membrane and organellar family of Na(+)/H(+) exchangers. Mol Aspects Med. 2013;34:236–251.
Gurney MA, Laubitz D, Ghishan FK, Kiela PR. Pathophysiology of Intestinal Na(+)/H(+) exchange. Cell Mol Gastroenterol Hepatol. 2017;3:27–40.
Xu H, Ghishan FK, Kiela PR. SLC9 Gene Family: Function, Expression, and Regulation. Compr Physiol. 2018;8:555–583.
Ruhlemann MC, Degenhardt F, Thingholm LB et al. Application of the distance-based F test in an mGWAS investigating beta diversity of intestinal microbiota identifies variants in SLC9A8 (NHE8) and 3 other loci. Gut Microbes. 2018;9:68–75.
Wang A, Li J, Zhao Y, Johansson ME, Xu H, Ghishan FK. Loss of NHE8 expression impairs intestinal mucosal integrity. Am J Physiol Gastrointest Liver Physiol. 2015;309:855–864.
Xu H, Li J, Chen H, Wang C, Ghishan FK. NHE8 plays important roles in gastric mucosal protection. Am J Physiol Gastrointest Liver Physiol. 2013;304:257–261.
Xu H, Zhang B, Li J, Wang C, Chen H, Ghishan FK. Impaired mucin synthesis and bicarbonate secretion in the colon of NHE8 knockout mice. American Journal of Physiology-Gastrointestinal and Liver Physiology. 2012;303:335–343.
Xu H, Li J, Chen H, Ghishan FK. NHE8 Deficiency Promotes Colitis-Associated Cancer in Mice via Expansion of Lgr5-Expressing Cells. Cell Mol Gastroenterol Hepatol. 2019;7:19–31.
Saksena S, Dudeja PK. Role of Membrane Transporters in Intestinal Cancers. Cellular and Molecular Gastroenterology and Hepatology. 2019;7:241–242.
Li X, Cai L, Xu H et al. Somatostatin regulates NHE8 protein expression via the ERK1/2 MAPK pathway in DSS-induced colitis mice. Am J Physiol Gastrointest Liver Physiol. 2016;311:954–963.
Li X, Liu W, Geng C et al. Ginsenoside Rg3 Suppresses Epithelial-Mesenchymal Transition via Downregulating Notch-Hes1 Signaling in Colon Cancer Cells. Am J Chin Med. 2021;49:217–235.
Zhang X, Sun H, Chen W et al. Elevated expression of AGGF1 predicts poor prognosis and promotes the metastasis of colorectal cancer. BMC Cancer. 2019;19:1252.
Cao L, Yuan Z, Liu M, Stock C. (Patho-)Physiology of Na+/H+ Exchangers (NHEs) in the Digestive System. Frontiers in Physiology. 2020. https://doi.org/10.3389/fphys.2019.01566.
Deng S, Wang A, Chen X et al. HBD Inhibits the Development of Colitis-Associated Cancer in Mice via the IL-6R/STAT3 Signaling Pathway. Int J Mol Sci. 2019. https://doi.org/10.3390/ijms20051069.
Rokavec M, Oner MG, Li H et al. IL-6R/STAT3/miR-34a feedback loop promotes EMT-mediated colorectal cancer invasion and metastasis. J Clin Invest. 2014;124:1853–1867.
Ahmad R, Kumar B, Chen Z et al. Loss of claudin-3 expression induces IL6/gp130/Stat3 signaling to promote colon cancer malignancy by hyperactivating Wnt/beta-catenin signaling. Oncogene. 2017;36:6592–6604.
Jin W. Role of JAK/STAT3 Signaling in the Regulation of Metastasis, the Transition of Cancer Stem Cells, and Chemoresistance of Cancer by Epithelial-Mesenchymal Transition. Cells. 2020. https://doi.org/10.3390/cells9010217.
Chen X, Yang W, Deng X, Ye S, Xiao W. Interleukin-6 promotes proliferative vitreoretinopathy by inducing epithelial-mesenchymal transition via the JAK1/STAT3 signaling pathway. Molecular vision. 2020;26:517–529.
Xiong H, Hong J, Du W et al. Roles of STAT3 and ZEB1 proteins in E-cadherin down-regulation and human colorectal cancer epithelial-mesenchymal transition. J Biol Chem. 2012;287:5819–5832.
Bromberg J, Wang TC. Inflammation and cancer: IL-6 and STAT3 complete the link. Cancer Cell. 2009;15:79–80.
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Müller-Newen G, Schaper F. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J. 2003;374:1–20.
Haan C, Kreis S, Margue C, Behrmann I. Jaks and cytokine receptors–an intimate relationship. Biochemical pharmacology. 2006;72:1538–1546.
Lu R, Zhang YG, Sun J. STAT3 activation in infection and infection-associated cancer. Molecular and cellular endocrinology. 2017;451:80–87.
Fang X, Hong Y, Dai L et al. CRH promotes human colon cancer cell proliferation via IL-6/JAK2/STAT3 signaling pathway and VEGF-induced tumor angiogenesis. Molecular carcinogenesis. 2017;56:2434–2445.
Wen J, Chang X, Bai B, Gao Q, Zhao Y. Orexin A Suppresses the Expression of Exosomal PD-L1 in Colon Cancer and Promotes T Cell Activity by Inhibiting JAK2/STAT3 Signaling Pathway. Digestive diseases and sciences. 2021. https://doi.org/10.1007/s10620-021-07077-0.
Funding
The work was supported by the National Natural Science Foundation of China (82070544) and the Sichuan Science and Technology Program (2020YFS0239).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have declared that there is no conflict of interests regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, W., Geng, C., Li, X. et al. Downregulation of SLC9A8 Promotes Epithelial–Mesenchymal Transition and Metastasis in Colorectal Cancer Cells via the IL6-JAK1/STAT3 Signaling Pathway. Dig Dis Sci 68, 1873–1884 (2023). https://doi.org/10.1007/s10620-022-07805-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-022-07805-0