Abstract
Background
Intestinal fibrosis is a common complication of Crohn’s disease (CD). Adiponectin reportedly exerts anti-inflammatory effects in various disease models, including colitis models.
Aims
In this study, we aimed to determine the effects of adiponectin on intestinal fibrosis and the underlying mechanisms.
Methods
A murine model of intestinal fibrosis was established by administering increasing doses of 2,4,6-trinitrobenzene sulfonic acid to Balb/c mice via enema for 7 weeks. Primary human fibroblasts were isolated from the colon tissues of patients with CD. The fibroblasts were incubated with transforming growth factor (TGF)-β1 to establish a fibrosis model in vitro. Pathway inhibitors were used to verify the potential signaling pathways involved in the anti-fibrogenic effect of adiponectin.
Results
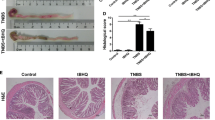
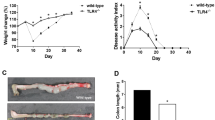
Compared with the normal mesentery, adiponectin expression was significantly increased in the hypertrophic mesentery of patients with CD. Intraperitoneal injection of adiponectin significantly decreased the activity of myeloperoxidase and the expression of pro-inflammatory cytokines (tumor necrosis factor α and interleukin 6) in the colon of fibrosis model mice, whereas the expression of the anti-inflammatory cytokine interleukin 10 was substantially increased. Moreover, adiponectin treatment inhibited colon shortening, decreased colon weight, and reduced fibrotic protein deposition in the model mice. Adiponectin reduced the phosphorylation of Smad2 and collagen deposition induced by TGF-β1 in primary human intestinal fibroblasts, with an increase in AMP-activated protein kinase (AMPK) phosphorylation. Furthermore, this phenomenon was reversed by the AMPK inhibitor.
Conclusions
Adiponectin can protect against intestinal fibrosis by enhancing the phosphorylation of AMPK and inhibiting the activity of the TGF-β1/Smad signaling pathway.






Similar content being viewed by others
References
Cosnes J, Cattan S, Blain A et al. Long-term evolution of disease behavior of Crohn’s disease. Inflamm Bowel Dis. 2002;8:244–250.
Cohen RD, Larson LR, Roth JM, Becker RV, Mummert LL. The cost of hospitalization in Crohn’s disease. Am J Gastroenterol. 2000;95:524–530.
Louis E, Collard A, Oger AF et al. Behaviour of Crohn’s disease according to the Vienna classification: changing pattern over the course of the disease. Gut. 2001;49:777–782.
Eder P, Adler M, Dobrowolska A, Kamhieh-Milz J, Witowski J. The role of adipose tissue in the pathogenesis and therapeutic outcomes of inflammatory bowel disease. Cells-Basel. 2019;8:628.
Bilski J, Mazur-Bialy A, Wojcik D et al. Role of obesity, mesenteric adipose tissue, and adipokines in inflammatory bowel diseases. Biomolecules. 2019;9:780.
Zhao Q, Liu Y, Tan L, Yan L, Zuo X. Adiponectin administration alleviates DSS-induced colonic inflammation in Caco-2 cells and mice. Inflamm Res. 2018;67:663–670.
Yang J, Lin SC, Chen G et al. Adiponectin promotes monocyte-to-fibroblast transition in renal fibrosis. J Am Soc Nephrol. 2013;24:1644–1659.
Handy JA, Fu PP, Kumar P et al. Adiponectin inhibits leptin signalling via multiple mechanisms to exert protective effects against hepatic fibrosis. Biochem J. 2011;440:385–395.
Weidinger C, Ziegler JF, Letizia M, Schmidt F, Siegmund B. Adipokines and their role in intestinal inflammation. Front Immunol. 2018;9:1974.
Konishi M, Haraguchi G, Ohigashi H et al. Adiponectin protects against doxorubicin-induced cardiomyopathy by anti-apoptotic effects through AMPK up-regulation. Cardiovasc Res. 2011;89:309–319.
Baranyi U, Winter B, Gugerell A et al. Primary human fibroblasts in culture switch to a Myofibroblast-Like phenotype independently of TGF beta. Cells-Basel. 2019;8:721.
Lian L, Huang Q, Zhang L et al. Anti-fibrogenic potential of mesenchymal stromal cells in treating fibrosis in Crohn’s disease. Dig Dis Sci. 2018;63:1821–1834. https://doi.org/10.1007/s10620-018-5082-8.
Shrout PE, Fleiss JL. Intraclass correlations: uses in assessing rater reliability. Psychol Bull. 1979;86:420–428.
Rieder F, Fiocchi C, Rogler G. Mechanisms, management, and treatment of fibrosis in patients with inflammatory bowel diseases. Gastroenterology. 2017;152:340–350.
Scheibe K, Kersten C, Schmied A et al. Inhibiting interleukin 36 receptor signaling reduces fibrosis in mice with chronic intestinal inflammation. Gastroenterology. 2019;156:1082–1097.
Holvoet T, Devriese S, Castermans K et al. Treatment of intestinal fibrosis in experimental inflammatory bowel disease by the pleiotropic actions of a local rho kinase inhibitor. Gastroenterology 2017;153:1054–1067.
Crohn BB, Ginzburg L, Oppenheimer GD. Regional ileitis: a pathologic and clinical entity. Am J Med. 1952;13:583–590.
Olivier I, Theodorou V, Valet P et al. Is Crohn’s creeping fat an adipose tissue? Inflamm Bowel Dis. 2011;17:747–757.
Rosen ED, Spiegelman BM. What we talk about when we talk about fat. Cell. 2014;156:20–44.
de Lima AR, Da CSAL, Costa C, Rufino R. Adiponectin in asthma and obesity: protective agent or risk factor for more severe disease? Lung. 2015;193:749–755.
Jin Z, Du Y, Schwaid AG et al. Maternal adiponectin controls milk composition to prevent neonatal inflammation. Endocrinology. 2015;156:1504–1513.
Louis E, Ribbens C, Godon A et al. Increased production of matrix metalloproteinase-3 and tissue inhibitor of metalloproteinase-1 by inflamed mucosa in inflammatory bowel disease. Clin Exp Immunol. 2000;120:241–246.
Heuschkel RB, MacDonald TT, Monteleone G et al. Imbalance of stromelysin-1 and TIMP-1 in the mucosal lesions of children with inflammatory bowel disease. Gut. 2000;47:57–62.
Neilson EG. Mechanisms of disease: fibroblasts: a new look at an old problem. Nat Clin Pract Nephrol. 2006;2:101–108.
Rodriguez P, Sassi Y, Troncone L et al. Deletion of delta-like 1 homologue accelerates fibroblast-myofibroblast differentiation and induces myocardial fibrosis. Eur Heart J. 2019;40:967–978.
Liu P, Miao K, Zhang L et al. Curdione ameliorates bleomycin-induced pulmonary fibrosis by repressing TGF-beta-induced fibroblast to myofibroblast differentiation. Respir Res. 2020;21:58.
Truffi M, Sorrentino L, Monieri M et al. Inhibition of fibroblast activation protein restores a balanced extracellular matrix and reduces fibrosis in Crohn’s disease strictures ex vivo. Inflamm Bowel Dis. 2018;24:332–345.
Kurahara LH, Sumiyoshi M, Aoyagi K et al. Intestinal myofibroblast TRPC6 channel may contribute to stenotic fibrosis in Crohn’s disease. Inflamm Bowel Dis. 2015;21:496–506.
de Bruyn JR, van den Brink GR, Steenkamer J et al. Fibrostenotic phenotype of myofibroblasts in crohn’s disease is dependent on tissue stiffness and reversed by LOX inhibition. J Crohns Colitis. 2018;12:849–859.
Wang Y, Jia L, Hu Z et al. AMP-activated protein kinase/myocardin-related transcription factor-A signaling regulates fibroblast activation and renal fibrosis. Kidney Int. 2018;93:81–94.
Yamamoto K, Kiyohara T, Murayama Y et al. Production of adiponectin, an anti-inflammatory protein, in mesenteric adipose tissue in Crohn’s disease. Gut. 2005;54:789–796.
Paul G, Schaffler A, Neumeier M et al. Profiling adipocytokine secretion from creeping fat in Crohn’s disease. Inflamm Bowel Dis. 2006;12:471–477.
Ha C, Martin A, Sepich-Poore GD et al. Translocation of viable gut microbiota to mesenteric adipose drives formation of creeping fat in humans. Cell. 2020;183:666–683.
Beagley KW, Fujihashi K, Lagoo AS et al. Differences in intraepithelial lymphocyte T cell subsets isolated from murine small versus large intestine. J Immunol. 1995;154:5611–5619.
Zhou C, Qiu Y, Yang H. CD4CD8alphaalpha IELs: they have something to say. Front Immunol. 2019;10:2269.
MacDonald TT. Breakdown of tolerance to the intestinal bacterial flora in inflammatory bowel disease (IBD). Clin Exp Immunol. 1995;102:445–447.
Funding
This study was supported by the National Natural Science Foundation of China (81770557 and 82070684) and Guangdong Natural Science Fund for Outstanding Youth Scholars (2020B151502067).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest regarding the content of this article.
Ethics approval and consent to participate
The experiments were carried out under a protocol approved by the Ethics Committee of the Sixth Affiliated Hospital of Sun Yat-sen University. The institutional and national guide for the care and use of laboratory animals was followed in the animal experiments, and it was approved by the Institutional Animal Care and Use Committee of the Sixth Affiliated Hospital of Sun Yat-Sen University.
Informed consent
All clinical samples collected and analyzed in this study were approved by the patients and all patients signed informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xie, M., Xiong, Z., Yin, S. et al. Adiponectin Alleviates Intestinal Fibrosis by Enhancing AMP-Activated Protein Kinase Phosphorylation. Dig Dis Sci 67, 2232–2243 (2022). https://doi.org/10.1007/s10620-021-07015-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-07015-0