Abstract
Objective
The role of TL1A in the intestinal mucosa barrier in inflammatory bowel disease (IBD) is still unclear. This study was aimed to investigate the expression levels of tight junction protein (TJ), myosin light chain kinase (MLCK), MyD88 and tumor necrosis factor (TNF) receptor-associated factor-6 (TRAF6) and how TL1A influences the intestinal barrier in IBD.
Methods
The mouse models of IBD were built using FMS-TL1A-GFP-transgenic mice and wild-type mice. The morphological and histopathological changes, bacterial translocation, permeability of colonic mucosa, and LPS level were assessed. Caco-2 cells were used to further investigate the association between TL1A and TNF-α and LPS. The protein level and mRNA changes of TJ proteins including ZO-1, occluding, JAMA, claudin-1, claudin-2, and claudin-3 were investigated using Western blot and real-time PCR. Protein changes of MLCK, MyD88 and TNF receptor-associated factor-6 (TRAF6), and TNF-α mRNA in the mouse colon were further assessed.
Results
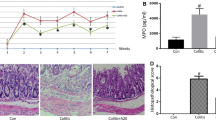
The IBD models were successfully built. Cooper HS score and histopathological score of the colon were higher in DSS/WT group than in control/WT group (P < 0.05), higher in DSS/Tg group than in control/Tg group (P < 0.05), and higher in DSS/Tg group than in DSS/WT group. PAS, colonic permeability of the colon, and FITC-D examination showed the similar results and trends. Compared with control/WT group, the levels of TL1A and claudin-2 were higher and the levels of ZO-1, occludin, JAMA, claudin-1, and claudin-3 were lower in DSS/WT group (P < 0.05). Compared with control/Tg group, the levels of TL1A and claudin-2 were higher and the levels of ZO-1, occludin, JAMA, claudin-1, and claudin-3 were lower in DSS/Tg group. Compared with Caco-2 + TNF-α group, the expression level of occludin and claudin-1 in Caco-2 + LV-TNFSF15 + TNF-α group was significantly lower (P < 0.05); p-MLC level was significantly higher. Compared with Caco-2 + LPS group, the expression level of occludin and claudin-1 significantly decreased in Caco-2 + LV-TNFSF15 + LPS group; MyD88 and TRAF6 expression level significantly increased.
Conclusion
The results suggested that TL1A could impair intestinal epithelial barrier in the mouse model of IBD and might regulate TJ expression via MLCK/p-MLC pathway and LPS-mediated MyD88/TRAF6 pathway.








Similar content being viewed by others
References
Baumgart DC, Carding SR. Inflammatory bowel disease: cause and immunobiology. Lancet. 2007;369:1627–1640.
Zhai H, Liu A, Huang W, et al. Increasing rate of inflammatory bowel disease: a 12-year retrospective study in NingXia, China. BMC Gastroenterol. 2016;16:1–7.
Ma TY, Iwamoto GK, Hoa NT, et al. TNF-alpha-induced increase in intestinal epithelial tight junction permeability requires NF-kappa B activation. Am J Physiol Gastrointest Liver Physiol. 2004;286:G367.
Fréour T, Jarry A, Bach-Ngohou K, et al. TACE inhibition amplifies TNF-alpha-mediated colonic epithelial barrier disruption. Int J Mol Med. 2009;23:41–48.
Migone TS, Zhang J, Luo X, et al. TL1A is a TNF-like ligand for DR3 and TR6/DcR3 and functions as a T cell costimulator. Immunity. 2002;16:479–492.
Cunningham KE, Turner JR. Myosin light chain kinase: pulling the strings of epithelial tight junction function. Ann N Y Acad Sci. 2012;1258:34–42.
Shih DQ, Robert B, Xiaolan Z, et al. Constitutive TL1A (TNFSF15) expression on lymphoid or myeloid cells leads to mild intestinal inflammation and fibrosis. PLoS ONE. 2011;6:e16090.
Jørgensen LG, Fredholm L, Hyltoft PP, Hey H, Munkholm P, Brandslund I. How accurate are clinical activity indices for scoring of disease activity in inflammatory bowel disease (IBD)? Clin Chem Lab Med CCLM. 2005;42:403–411.
Cooper HS, Murthy SN, Shah RS, Sedergran DJ. Clinicopathologic study of dextran sulfate sodium experimental murine colitis. Lab Invest. 1993;69:238–249.
Llamas MA, Aller MA, Marquina D, Nava MP, Arias J. Bacterial translocation to mesenteric lymph nodes increases in chronic portal hypertensive rats. Dig Dis Sci. 2010;55:2244–2254.
Shih DQ, Zheng L, Zhang X, et al. Inhibition of a novel fibrogenic factor Tl1a reverses established colonic fibrosis. Mucosal Immunol. 2014;7:1492–1503.
Buchheister S, Buettner M, Basic M, et al. CD14 plays a protective role in experimental inflammatory bowel disease by enhancing intestinal barrier function. Am J Pathol. 2017;187:1106.
Ye X, Sun M. AGR2 ameliorates tumor necrosis factor-α-induced epithelial barrier dysfunction via suppression of NF-κB p65-mediated MLCK/p-MLC pathway activation. Int J Mol Med. 2017;39:1206–1214.
Chang J, Leong RW, Wasinger V, Ip M, Yang M, Giang PT. Impaired intestinal permeability contributes to ongoing bowel symptoms in patients with inflammatory bowel disease and mucosal healing. Gastroenterology. 2017;153:723.
Takeda K, Akira S. Toll-like receptors. In: Current Protocols in Immunology. 2007; Chapter 14:Unit 14.12.
Taniguchi K, Wu LW, Grivennikov SI, et al. A gp130-Src-YAP module links inflammation to epithelial regeneration. Nature. 2015;519:57–62.
Amasheh M, Grotjohann I, Amasheh S, et al. Regulation of mucosal structure and barrier function in rat colon exposed to tumor necrosis factor alpha and interferon gamma in vitro: a novel model for studying the pathomechanisms of inflammatory bowel disease cytokines. Scand J Gastroenterol. 2009;44:1226–1235.
Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol. 2001;2:675–680.
Negroni A, Costanzo M, Vitali R, et al. Characterization of adherent-invasive Escherichia coli isolated from pediatric patients with inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:913–924.
Ahmad R, Chaturvedi R, Olivares-Villagómez D, et al. Targeted colonic claudin-2 expression renders resistance to epithelial injury, induces immune suppression, and protects from colitis. Mucosal Immunol. 2014;7:1340–1353.
Oshima T, Miwa H, Matsumoto T, Joh T. Changes in the expression of claudins in active ulcerative colitis. J Gastroenterol Hepatol. 2008;136:A-414-A-414.
Du J, Chen Y, Shi Y, et al. 1,25-Dihydroxyvitamin D protects intestinal epithelial barrier by regulating the myosin light chain kinase signaling pathway. Inflamm Bowel Dis. 2015;21:2495.
He F, Peng J, Deng XL, et al. Mechanisms of tumor necrosis factor-alpha-induced leaks in intestine epithelial barrier. Cytokine. 2012;59:264–272.
Boivin MA, Ye D, Kennedy JC, Al-Sadi R, Shepela C, Ma TY. Mechanism of glucocorticoid regulation of the intestinal tight junction barrier. Am J Physiol Gastrointest Liver Physiol. 2007;292:G590.
Barrett R, Zhang X, Koon HW, et al. Constitutive TL1A expression under colitogenic conditions modulates the severity and location of gut mucosal inflammation and induces fibrostenosis. Am J Pathol. 2012;180:636–649.
Negroni A, Costanzo M, Vitali R, et al. Characterization of adherent-invasive Escherichia coli isolated from pediatric patients with inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:913–924.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, M., Jia, W., Wang, D. et al. Effects and Mechanism of Constitutive TL1A Expression on Intestinal Mucosal Barrier in DSS-Induced Colitis. Dig Dis Sci 64, 1844–1856 (2019). https://doi.org/10.1007/s10620-019-05580-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-05580-z