Abstract
Background
Nausea and vomiting are commonly associated with medication use. Dopaminergic agonists have been associated with these symptoms, but their impact in patients without Parkinson’s disease, such as those with restless legs syndrome (RLS), is not well characterized.
Aims
We sought to determine whether the non-ergoline dopamine agonist ropinirole is associated with nausea and vomiting in adults with RLS.
Methods
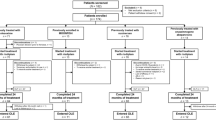
We conducted a systematic review using PUBMED, EMBASE, and clinical trial databases to identify placebo-controlled clinical trials of ropinirole for RLS treatment. We extracted data including dosing schedule and the proportion of patients reporting nausea and/or vomiting. We also determined hazard ratios (HR) using a random effects proportional hazard model.
Results
We extracted data from a pool of 13 studies. The prevalence of nausea in the ropinirole-treated RLS group (RLS-R; N = 1528) was 37.2% compared to 9.4% in the placebo-treated RLS group (RLS-P; N = 1395) (p < 0.0001). The prevalence of vomiting in the RLS-R group was 10.9% compared to 2.6% in the RLS-P group (p < 0.0001). Ropinirole use was associated with a higher risk of reporting nausea (HR 5.924 [4.410–7.959], p < 0.001) and experiencing vomiting (HR 4.628 [3.035–7.057], p < 0.0001). Nausea and vomiting represented nearly 50% of all adverse events reported.
Conclusions
Nausea and vomiting are quite common side effects in those using ropinirole for RLS. As RLS is more widely recognized and treated; the prevalence of ropinirole-induced nausea and vomiting could grow substantially. Ropinirole use should be considered as a cause of chronic nausea and vomiting.


Similar content being viewed by others
References
Haug TT, Mykletun A, Dahl AA. The prevalence of nausea in the community: psychological, social and somatic factors. Gen Hosp Psychiatry. 2002;24:81–86.
Hassid BG, Day LW, Awad MA, Sewell JL, Osterberg EC, Breyer BN. Using search engine query data to explore the epidemiology of common gastrointestinal symptoms. Dig Dis Sci. 2017;62:588–592.
Peery AF, Dellon ES, Lund J, et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012;143:1179–1187.e1.
Aurora RN, Kristo DA, Bista SR, et al. The treatment of restless legs syndrome and periodic limb movement disorder in adults—an update for 2012: practice parameters with an evidence-based systematic review and meta-analyses: an American Academy of Sleep Medicine Clinical Practice Guideline. Sleep. 2012;35:1039–1062.
Allen RP, Walters AS, Montplaisir J, et al. Restless legs syndrome prevalence and impact: REST general population study. Arch Intern Med. 2005;165:1286–1292.
Hening W, Walters AS, Allen RP, Montplaisir J, Myers A, Ferini-Strambi L. Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: the REST (RLS epidemiology, symptoms, and treatment) primary care study. Sleep Med. 2004;5:237–246.
Thorpy MJ. New paradigms in the treatment of restless legs syndrome. Neurology. 2005;64:S28–S33.
Zhou C-Q, Zhang J-W, Wang M, Peng G-G. Meta-analysis of the efficacy and safety of long-acting non-ergot dopamine agonists in Parkinson’s disease. J Clin Neurosci. 2014;21:1094–1101.
Singaram C, Ashraf W, Gaumnitz EA, et al. Dopaminergic defect of enteric nervous system in Parkinson’s disease patients with chronic constipation. The Lancet. 1995;346:861–864.
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17:1–12.
Walters AS, LeBrocq C, Dhar A, et al. Validation of the International Restless Legs Syndrome Study Group rating scale for restless legs syndrome. Sleep Med. 2003;4:121–132.
Kenward H, Pelligand L, Savary-Bataille K, Elliott J. Nausea: current knowledge of mechanisms, measurement and clinical impact. Vet J. 2015;203:36–43.
O’Brien C. Nausea and vomiting. Can Fam Physician. 2008;54:861–863.
Janelsins MC, Tejani MA, Kamen C, Peoples AR, Mustian KM, Morrow GR. Current pharmacotherapy for chemotherapy-induced nausea and vomiting in cancer patients. Expert Opin Pharmacother. 2013;14:757–766.
Giorgi L, Asgharian A, Hunter B. Ropinirole in patients with restless legs syndrome and baseline IRLS total scores ≥ 24: efficacy and tolerability in a 26-week, double-blind, parallel-group, placebo-controlled study followed by a 40-week open-label extension. Clin Ther. 2013;35:1321–1336.
Benes H, Mattern W, Peglau I, et al. Ropinirole improves depressive symptoms and restless legs syndrome severity in RLS patients: a multicentre, randomized, placebo-controlled study. J Neurol. 2011;258:1046–1054.
Trenkwalder C, Garcia-Borreguero D, Montagna P, et al. Ropinirole in the treatment of restless legs syndrome: results from the TREAT RLS 1 study, a 12 week, randomised, placebo controlled study in 10 European countries. J Neurol Neurosurg Psychiatr. 2004;75:92–97.
Allen R, Becker PM, Bogan R, et al. Ropinirole decreases periodic leg movements and improves sleep parameters in patients with restless legs syndrome. Sleep. 2004;27:907–914.
Bogan RK, Fry JM, Schmidt MH, Carson SW, Ritchie SY. TREAT RLS US Study Group. Ropinirole in the treatment of patients with restless legs syndrome: a US-based randomized, double-blind, placebo-controlled clinical trial. Mayo Clin Proc. 2006;81:17–27.
Walters AS, Ondo WG, Dreykluft T, et al. Ropinirole is effective in the treatment of restless legs syndrome. TREAT RLS 2: a 12-week, double-blind, randomized, parallel-group, placebo-controlled study. Mov Disord. 2004;19:1414–1423.
Montplaisir J, Karrasch J, Haan J, Volc D. Ropinirole is effective in the long-term management of restless legs syndrome: a randomized controlled trial. Mov Disord. 2006;21:1627–1635.
Tompson D, Della P. A single-blind, placebo-controlled, forced-titration study of ropinirole in RLS: pharmacokinetic-pharmacodynamic relationships. Boston: Associated Professional Sleep Societies; 2004:A298–A299.
A 12-week, double-blind, placebo controlled, parallel group study to assess the efficacy and safety of Ropinirole XR (extended release) in patients with restless legs syndrome. Glaxo Smith Klein Clinical Study Register. https://www.gsk-clinicalstudyregister.com/files2/gsk-101468-205-clinical-study-report-redact.pdf.
A 12-week, multi-center, double-blind, placebo-controlled, parallel group, flexible dose polysomnography study of ropinirole controlled release for restless legs syndrome (RLS) in RLS patients with sleep disturbance and periodic limb movements (PLM) during sleep. Glaxo Smith Kline Clinical Study Register. https://www.gsk-clinicalstudyregister.com/files2/gsk-rrl103660-clinical-study-report-redact.pdf.
A 12-week, double-blind, placebo-controlled, parallel-group study to assess the effectiveness of ropinirole in patients willing to take regular medication for their restless legs syndrome in a primary care setting. Glaxo Smith Kline Clinical Study Register. https://www.gsk-clinicalstudyregister.com/files2/23627.pdf.
Kushida CA, Geyer J, Tolson JM, Asgharian A. Patient- and physician-rated measures demonstrate the effectiveness of ropinirole in the treatment of restless legs syndrome. Clin Neuropharmacol. 2008;31:281–286.
A double-blind, randomized, placebo-controlled, parallel-group study to investigate the tolerability of a dose-escalating regimen of ropinirole in patients suffering from Restless Legs Syndrome (RLS). Glaxo Smith Kline Clinical Study Register. https://www.gsk-clinicalstudyregister.com/files2/gsk-101468-207-clinical-study-report-redact.pdf.
ClinCalc DrugStats Database version 17.0. http://clincalc.com/DrugStats/Drugs/RopiniroleHydrochloride.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflicts to report.
Rights and permissions
About this article
Cite this article
Kurin, M., Bielefeldt, K. & Levinthal, D.J. Prevalence of Nausea and Vomiting in Adults Using Ropinirole: A Systematic Review and Meta-Analysis. Dig Dis Sci 63, 687–693 (2018). https://doi.org/10.1007/s10620-018-4937-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-4937-3