Abstract
Background
Abnormality of PTEN gene and Wnt/β-catenin signaling have been strongly implicated in various malignant cancers. Recently, it has been noted that a functional interaction/cross-talk was found between the PTEN/PI3K/AKT and Wnt/β-catenin, which plays a key role in the development of cancers. However, few related studies on gastric cancer are available.
Aim
We examined the expression of PTEN and β-catenin in gastric cancer tissues and detected whether down-regulation of PTEN promotes the migration and invasion in gastric cancer cells along with its underlying mechanism.
Materials and Methods
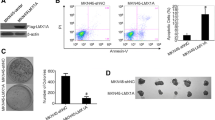
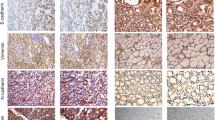
Immunocytochemistry, a wound healing assay, a Matrigel invasion assay, an immunofluorescence staining were performed to detect expression of PTEN and β-catenin in gastric cancer and adjacent normal tissues, cell migration, cell invasion, and the effects of PTEN knockdown on β-catenin in cells, respectively. Further, MMP-2 and MMP-9 activities were analyzed by zymography assay. The changes in related proteins were further quantified by western blotting.
Results
Low expression of PTEN was found in majority of gastric cancer tissues, which showed significant associations with differentiation grade in gastric cancer patients. Further, a negative correlation was revealed between PTEN and β-catenin protein expression in gastric cancer tissues (r = − 0.546, P < 0.01). Additionally, PTEN knockdown promoted the migration and invasion of cells and caused an obvious increase in p-AKT, p-GSK-3β, β-catenin, E-cadherin, MMP-7, MMP-2, and MMP-9 in gastric cancer cells.
Conclusion
Our results indicated PTEN gene might induce cell invasion and migration via regulating AKT/GSK-3β/β-catenin signaling pathway, playing a vital role in the progression of gastric cancer.






Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108.
Yang L, Parkin DM, Ferlay J, Li L, Chen Y. Estimates of cancer incidence in China for 2000 and projections for 2005. Cancer Epidemiol Biomarkers Prev. 2005;14:243–250.
Yang L. Incidence and mortality of gastric cancer in China. World J Gastroenterol. 2006;12:17–20.
Guo M, Yan W. Epigenetics of gastric cancer. Methods Mol Biol. 2015;1238:783–799.
Choi YJ, Kim N. Gastric cancer and family history. Korean J Intern Med. 2016;31:1042–1053.
Suzuki H, Iwasaki E, Hibi T. Helicobacter pylori and gastric cancer. Gastric Cancer. 2009;12:79–87.
Elistratova EP, Laktionov PP, Shelestiuk PI, Tuzikov SA, Vlasov VV, Rykova E. Immunochemical and molecular-genetic markers in gastric cancer diagnostics. Biomed Khim. 2009;55:15–31.
Li J, Yen C, Liaw D, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science. 1997;275:1943–1947.
Saal LH, Gruvberger-Saal SK, Persson C, Lovgren K, Jumppanen M, et al. Recurrent gross mutations of the PTEN tumor suppressor gene in breast cancers with deficient DSB repair. Nat Genet. 2008;40:102–107.
Yokomizo A, Tindall DJ, Drabkin H, Gemmill R, Franklin W, et al. PTEN/MMAC1 mutations identified in small cell, but not in non-small cell lung cancers. Oncogene. 1998;17:475–479.
Wang SI, Puc J, Li J, Bruce JN, Cairns P, et al. Somatic mutations of PTEN in glioblastoma multiforme. Cancer Res. 1997;57:4183–4186.
Hollander MC, Blumenthal GM, Dennis PA. PTEN loss in the continuum of common cancers, rare syndromes and mouse models. Nat Rev Cancer. 2011;11:289–301.
Dey N, Crosswell HE, De P, et al. The protein phosphatase activity of PTEN regulates SRC family kinases and controls glioma migration. Cancer Res. 2008;68:1862–1871.
Davidson L, Maccario H, Perera NM, et al. Suppression of cellular proliferation and invasion by the concerted lipid and protein phosphatase activities of PTEN. Oncogene. 2010;29:687–697.
Zhang F, Wang Y, Sun P, Wang ZQ, Wang DS, Zhang DS, Wang FH, Fu JH, Xu RH, Li YH. Fibrinogen promotes malignant biological tumor behavior involving epithelial-mesenchymal transition via the p-AKT/p-mTOR pathway in esophageal squamous cell carcinoma. J Cancer Res Clin Oncol. 2017. doi:10.1007/s00432-017-2493-4.
Su K, Huang L, Li W, Yan X, Li X, et al. TC-1 (c8orf4) enhances aggressive biologic behavior in lung cancer through the Wnt/beta-catenin pathway. J Surg Res. 2013;185:255–263.
Zhang H, Xue Y. Wnt pathway is involved in advanced gastric carcinoma. Hepatogastroenterology. 2008;55:1126–1130.
Tian X, Liu Z, Niu B, Zhang J, Tan TK, et al. E-cadherin/beta-catenin complex and the epithelial barrier. J Biomed Biotechnol. 2011;2011:567305.
Huang J, Xiao D, Li G, Ma J, Chen P, et al. EphA2 promotes epithelial-mesenchymal transition through the Wnt/beta-catenin pathway in gastric cancer cells. Oncogene. 2014;33:2737–2747.
Bonvini P, Hwang SG, El-Gamil M, et al. Nuclear beta-catenin displays GSK-3beta- and APC-independent proteasome sensitivity in melanoma cells. Biochim Biophys Acta. 2000;1495:308–318.
Liu C, Li Y, Semenov M, et al. Control of beta-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell. 2002;108:837–847.
Persad S, Troussard AA, McPhee TR, Mulholland DJ, Dedhar S. Tumor suppressor PTEN inhibits nuclear accumulation of beta-catenin and T cell/lymphoid enhancer factor 1-mediated transcriptional activation. J Cell Biol. 2001;153:1161–1174.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29.
Shen L, Shan YS, Hu HM, et al. Management of gastric cancer in Asia: resource-stratified guidelines. Lancet Oncol. 2013;14:e535–e547.
Steck PA, Pershouse MA, Jasser SA, et al. Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet. 1997;15:356–362.
Sato K, Tamura G, Tsuchiya T, et al. Analysis of genetic and epigenetic alterations of the PTEN gene in gastric cancer. Virchows Arch. 2002;440:160–165.
Fei G, Ebert MP, Mawrin C, et al. Reduced PTEN expression in gastric cancer and in the gastric mucosa of gastric cancer relatives. Eur J Gastroenterol Hepatol. 2002;14:297–303.
Hafsi S, Pezzino FM, Candido S, et al. Gene alterations in the PI3K/PTEN/AKT pathway as a mechanism of drug-resistance (review). Int J Oncol. 2012;40:639–644.
Horie Y, Suzuki A, Kataoka E, et al. Hepatocyte-specific Pten deficiency results in steatohepatitis and hepatocellular carcinomas. J Clin Invest. 2004;113:1774–1783.
Kim CS, Vasko VV, Kato Y, et al. AKT activation promotes metastasis in a mouse model of follicular thyroid carcinoma. Endocrinology. 2005;146:4456–4463.
Laurent PA, Severin S, Gratacap MP, Payrastre B. Class I PI 3-kinases signaling in platelet activation and thrombosis: PDK1/Akt/GSK3 axis and impact of PTEN and SHIP1. Adv Biol Regul. 2014;54:162–174.
Manning BD, Toker A. AKT/PKB signaling: Navigating the network. Cell. 2017;169:381–405.
Moore SF, van den Bosch MT, Hunter RW, Sakamoto K, Poole AW, Hers I. Dual regulation of glycogen synthase kinase 3 (GSK3)alpha/beta by protein kinase C (PKC)alpha and Akt promotes thrombin-mediated integrin alphaIIbbeta3 activation and granule secretion in platelets. J Biol Chem. 2013;288:3918–3928.
Wang Y, Fu D, Su J, et al. C1QBP suppresses cell adhesion and metastasis of renal carcinoma cells. Sci Rep. 2017;7:999.
Clevers H, Nusse R. Wnt/beta-catenin signaling and disease. CELL. 2012;149:1192–1205.
MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17:9–26.
Landman EB, Miclea RL, van Blitterswijk CA, Karperien M. Small molecule inhibitors of WNT/beta-catenin signaling block IL-1beta- and TNFalpha-induced cartilage degradation. Arthritis Res Ther. 2013;15:R93.
Acknowledgments
We are grateful to Mr. Hong Xia from the Key Laboratory of Hubei Province for Digestive System Disease for laboratory assistance, kindly guidance, help, advice, and support.
Funding
The study was supported by the research grants from the National Natural Science Foundation of China (Nos. 81372551 and 81572426).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Ethical approval
All procedures performed in studies involving human participants were approved by Clinical Research Ethics Committee of Renmin Hospital of Wuhan University, which were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consents were obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Ma, J., Guo, X., Zhang, J. et al. PTEN Gene Induces Cell Invasion and Migration via Regulating AKT/GSK-3β/β-Catenin Signaling Pathway in Human Gastric Cancer. Dig Dis Sci 62, 3415–3425 (2017). https://doi.org/10.1007/s10620-017-4764-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4764-y