Abstract
Background
To investigate the protective effects of helix B surface peptide (HBSP) on acute liver injury induced by carbon tetrachloride (CCl4).
Methods
HBSP (8 nmol/kg) was intraperitoneally injected into C57 BL/6 mice 2 h after CCl4 administration. Serum and liver tissue samples were collected 24 h after injury. Liver function and histological injuries were evaluated. Inflammatory cell infiltration and cytokines were examined and hepatocytes apoptosis was measured. The human liver cell line LO2 and murine primary hepatocytes were stimulated by CCl4 with and without HBSP treatment and glutathione peroxidase activity, cell survival, and apoptosis were evaluated. In addition, we examined the PI3K/Akt/mTORC1 pathway to elucidate the mechanism underlying HBSP-mediated protection in acute liver injury.
Results
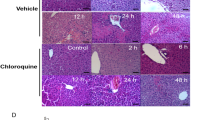
HBSP significantly decreased serum alanine aminotransferase, aspartate aminotransferase, lactate dehydrogenase, and pro-inflammatory cytokines in liver tissues after CCl4 injection compared with those in the control group. Immunohistochemical staining indicated that the number of CD3-, CD8-, and CD68-positive cells and the expression of cleaved caspase-3 were significantly decreased by HBSP treatment. Additionally, HBSP reduced apoptosis in vivo. In an in vitro study, the glutathione peroxidase activity and survival rate increased, while the total apoptotic rate was reduced in the HBSP-treated group compared with that in the control group after CCl4 treatment. HBSP activated the PI3K/Akt/mTORC1 pathway, which was confirmed by the PI3K inhibitor LY294002 both in vivo and in vitro. Furthermore, HBSP increased the survival of mice with acute liver injury, and this effect was abolished by LY294002.
Conclusions
HBSP is a potential therapeutic agent against acute liver injury induced by CCl4.






Similar content being viewed by others
Abbreviations
- CCl4 :
-
Carbon tetrachloride
- TNF-α:
-
Tumor necrosis factor-α
- IL-6:
-
Interleukin-6
- EPO:
-
Erythropoietin
- EPOR:
-
EPO receptor
- βcR:
-
β-Common receptor
- HBSP:
-
Helix B surface peptide
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- LDH:
-
Lactate dehydrogenase
- MDA:
-
Malondialdehyde
- MTT:
-
Methyl thiazolyl tetrazolium
- GSH:
-
Glutathione
- GSH-Px:
-
Glutathione peroxidase
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- DMEM:
-
Dulbecco’s modified Eagles medium
- SDS:
-
Sodium dodecyl sulfate
- SD:
-
Standard deviation
- SOD:
-
Superoxide dismutase
- ANOVA:
-
Analysis of variance
- IFN-γ:
-
Interferon-γ
References
Andrade RJ, Lucena MI, Fernandez MC, et al. Drug-induced liver injury: an analysis of 461 incidences submitted to the Spanish registry over a 10-year period. Gastroenterology. 2005;129:512–521.
Cederbaum AI, Lu Y, Wu D. Role of oxidative stress in alcohol-induced liver injury. Arch Toxicol. 2009;83:519–548.
Fujii T, Fuchs BC, Yamada S, et al. Mouse model of carbon tetrachloride induced liver fibrosis: histopathological changes and expression of CD133 and epidermal growth factor. BMC Gastroenterol. 2010;10:79.
Zhu R, Zeng G, Chen Y, et al. Oroxylin A accelerates liver regeneration in CCl(4)-induced acute liver injury mice. PLoS One. 2013;8:e71612.
Holt AP, Stamataki Z, Adams DH. Attenuated liver fibrosis in the absence of B cells. Hepatology. 2006;43:868–871.
Lin X, Huang R, Zhang S, et al. Methyl helicterate protects against CCl4-induced liver injury in rats by inhibiting oxidative stress, NF-kappaB activation, Fas/FasL pathway and cytochrome P4502E1 level. Food Chem Toxicol. 2012;50:3413–3420.
Brines M, Cerami A. Erythropoietin-mediated tissue protection: reducing collateral damage from the primary injury response. J Intern Med. 2008;264:405–432.
Ishii Y, Sawada T, Murakami T, et al. Renoprotective effect of erythropoietin against ischaemia–reperfusion injury in a non-human primate model. Nephrol Dial Transplant. 2011;26:1157–1162.
Hu L, Yang C, Zhao T, et al. Erythropoietin ameliorates renal ischemia and reperfusion injury via inhibiting tubulointerstitial inflammation. J Surg Res. 2012;176:260–266.
Wu Y, Zhang J, Liu F, et al. Protective effects of HBSP on ischemia reperfusion and cyclosporine a induced renal injury. Clin Dev Immunol. 2013;2013:758159.
Brines M, Grasso G, Fiordaliso F, et al. Erythropoietin mediates tissue protection through an erythropoietin and common beta-subunit heteroreceptor. Proc Natl Acad Sci USA. 2004;101:14907–14912.
Velly L, Pellegrini L, Guillet B, Bruder N, Pisano P. Erythropoietin 2nd cerebral protection after acute injuries: a double-edged sword? Pharmacol Ther. 2010;128:445–459.
Lippi G, Franchini M, Favaloro EJ. Thrombotic complications of erythropoiesis-stimulating agents. Semin Thromb Hemost. 2010;36:537–549.
Krapf R, Hulter HN. Arterial hypertension induced by erythropoietin and erythropoiesis-stimulating agents (ESA). Clin J Am Soc Nephrol. 2009;4:470–480.
Brines M, Patel NS, Villa P, et al. Nonerythropoietic, tissue-protective peptides derived from the tertiary structure of erythropoietin. Proc Natl Acad Sci USA. 2008;105:10925–10930.
Yang C, Zhao T, Lin M, et al. Helix B surface peptide administered after insult of ischemia reperfusion improved renal function, structure and apoptosis through beta common receptor/erythropoietin receptor and PI3K/Akt pathway in a murine model. Exp Biol Med (Maywood). 2013;238:111–119.
Robertson CS, Cherian L, Shah M, et al. Neuroprotection with an erythropoietin mimetic peptide (pHBSP) in a model of mild traumatic brain injury complicated by hemorrhagic shock. J Neurotrauma. 2012;29:1156–1166.
Collino M, Benetti E, Rogazzo M, et al. A non-erythropoietic peptide derivative of erythropoietin decreases susceptibility to diet-induced insulin resistance in mice. Br J Pharmacol. 2014;171:5802–5815.
Ueba H, Shiomi M, Brines M, et al. Suppression of coronary atherosclerosis by helix B surface Peptide, a nonerythropoietic, tissue-protective compound derived from erythropoietin. Mol Med. 2013;19:195–202.
Patel NS, Nandra KK, Brines M, et al. A nonerythropoietic peptide that mimics the 3D structure of erythropoietin reduces organ injury/dysfunction and inflammation in experimental hemorrhagic shock. Mol Med. 2011;17:883–892.
Yang C, Zhao T, Zhao Z, et al. Serum-stabilized naked caspase-3 siRNA protects autotransplant kidneys in a porcine model. Mol Ther. 2014;22:1817–1828.
Yang C, Li L, Xue Y, et al. Innate immunity activation involved in unprotected porcine auto-transplant kidneys preserved by naked caspase-3 siRNA. J Transl Med. 2013;11:210.
Yang C, Jia Y, Zhao T, et al. Naked caspase 3 small interfering RNA is effective in cold preservation but not in autotransplantation of porcine kidneys. J Surg Res. 2013;181:342–354.
Borkham-Kamphorst E, Huss S, Van de Leur E, Haas U, Weiskirchen R. Adenoviral CCN3/NOV gene transfer fails to mitigate liver fibrosis in an experimental bile duct ligation model because of hepatocyte apoptosis. Liver Int. 2012;32:1342–1353.
Zhao Z, Yang C, Wang L, et al. The regulatory T cell effector soluble fibrinogen-like protein 2 induces tubular epithelial cell apoptosis in renal transplantation. Exp Biol Med (Maywood). 2014;239:193–201.
Vesey DA, Cheung C, Pat B, Endre Z, Gobe G, Johnson DW. Erythropoietin protects against ischaemic acute renal injury. Nephrol Dial Transplant. 2004;19:348–355.
Burger DE, Xiang FL, Hammoud L, Jones DL, Feng Q. Erythropoietin protects the heart from ventricular arrhythmia during ischemia and reperfusion via neuronal nitric-oxide synthase. J Pharmacol Exp Ther. 2009;329:900–907.
Hochhauser E, Pappo O, Ribakovsky E, et al. Recombinant human erythropoietin attenuates hepatic injury induced by ischemia/reperfusion in an isolated mouse liver model. Apoptosis. 2008;13:77–86.
Gunnarson E, Song Y, Kowalewski JM, et al. Erythropoietin modulation of astrocyte water permeability as a component of neuroprotection. Proc Natl Acad Sci USA. 2009;106:1602–1607.
Ueba H, Brines M, Yamin M, et al. Cardioprotection by a nonerythropoietic, tissue-protective peptide mimicking the 3D structure of erythropoietin. Proc Natl Acad Sci USA. 2010;107:14357–14362.
Collino M, Thiemermann C, Cerami A, Brines M. Flipping the molecular switch for innate protection and repair of tissues: long-lasting effects of a non-erythropoietic small peptide engineered from erythropoietin. Pharmacol Ther. 2015;151:32–40.
Aldaba-Muruato LR, Moreno MG, Shibayama M, Tsutsumi V, Muriel P. Protective effects of allopurinol against acute liver damage and cirrhosis induced by carbon tetrachloride: modulation of NF-kappaB, cytokine production and oxidative stress. Biochim Biophys Acta. 2012;1820:65–75.
Tilg H, Kaser A, Moschen AR. How to modulate inflammatory cytokines in liver diseases. Liver Int. 2006;26:1029–1039.
Canbay A, Friedman S, Gores GJ. Apoptosis: the nexus of liver injury and fibrosis. Hepatology. 2004;39:273–278.
Neri LM, Borgatti P, Capitani S, Martelli AM. The nuclear phosphoinositide 3-kinase/AKT pathway: a new second messenger system. Biochim Biophys Acta. 2002;1584:73–80.
Cantley LC. The phosphoinositide 3-kinase pathway. Science. 2002;296:1655–1657.
Hsu JT, Kan WH, Hsieh CH, et al. Mechanism of estrogen-mediated attenuation of hepatic injury following trauma-hemorrhage: Akt-dependent HO-1 up-regulation. J Leukoc Biol. 2007;82:1019–1026.
Mazelin L, Panthu B, Nicot AS, et al. mTOR inactivation in myocardium from infant mice rapidly leads to dilated cardiomyopathy due to translation defects and p53/JNK-mediated apoptosis. J Mol Cell Cardiol. 2016;97:213–225.
Acknowledgments
The study was supported by the National Natural Science Foundation of China (Nos. 81301820, 81472673, 81400752, 81401877, 81500457, 81672720), and the National Clinical Key Special Subject of China.
Author information
Authors and Affiliations
Contributions
SDW, CY and NX designed the research, conducted the experiments, collected and analyzed the data, and wrote the manuscript. LYW and YL contributed to the data acquisition. JYW and XZS revised the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
All animal experiments were performed in accordance with the guidelines of the Care and Use of Laboratory Animals of the Laboratory Animal Ethical Commission of Fudan University with good surgical practices.
Availability of data and materials
The data and all outputs of the current study are available for testing by reviewers and scientists who wish to use them with kind full permission.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10620_2017_4553_MOESM2_ESM.jpg
Supplemental Figure 1. Cytotoxicity of CCl4 at different concentrations in LO2 cells in vitro. (A) Serum ALT, AST, and MTT were measured in the groups treated with different concentrations of CCl4 (40, 60 and 80%). (B-C) Apoptosis was detected by flow cytometry analysis in the groups treated with different concentrations of CCl4. Data are represented as the mean ± SD (n = 5). (JPEG 2641 kb)
10620_2017_4553_MOESM3_ESM.jpg
Supplemental Figure 2. The expression of EPOR/βcR and Cyp2E1 in the liver. The expression of EPOR and EPOR/βcR were detected in the normal murine liver tissue (A) and murine primary hepatocyte (B). The expression of Cyp2E1 in the liver was investigated in different treatment groups (C). (JPEG 2193 kb)
Rights and permissions
About this article
Cite this article
Wu, S., Yang, C., Xu, N. et al. The Protective Effects of Helix B Surface Peptide on Experimental Acute Liver Injury Induced by Carbon Tetrachloride. Dig Dis Sci 62, 1537–1549 (2017). https://doi.org/10.1007/s10620-017-4553-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4553-7