Abstract
Background
Orai1, which is involved in store-operated calcium entry, has recently been implicated in cancer progression. However, the role of Orai1 in colorectal cancer (CRC) progression remains unclear.
Methods
We used real-time PCR and western blot to measure Orai1 expression in four CRC cell lines, 60 tumor pairs, and corresponding non-tumor tissues from CRC patients. Immunohistochemistry was performed to examine Orai1 expression in CRC and corresponding non-tumor tissues. Statistical analyses were applied to evaluate the prognostic value and associations of Orai1 expression with clinical parameters. Furthermore, the Orai1 gene was overexpressed in HCT116 cell and silenced with siRNA in LOVO cell. Moreover, cell proliferation and apoptosis were measured using MTT assay and flow cytometry, and a molecular mechanism of Orai1 regulation by miR-519 was explored.
Results
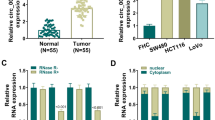
Orai1 expression was higher in CRC tissues than adjacent non-cancerous tissues, and this was positively correlated in CRC patients with distant metastasis and poor prognosis. Also, increased expression of Orai1 was observed in highly invasive CRC cell lines and ectopic expression of Orai1 enhanced cell proliferation and inhibited apoptosis; silencing Orai1 suppressed cell proliferation and induced apoptosis. The Akt/GSK3β pathway contributed to Orai1 effects in CRC cells, and Orai1 was a direct target of miR-519, a microRNA not previously reported to be involved in both CRC tissues and cell lines.
Conclusions
We identified a novel CRC regulatory circuit involving the miR-519-Orai1 axis, and dysfunction of this drives diverse aspects of CRC pathogenesis.




Similar content being viewed by others
References
Center MM, Jemal A, Smith RA, Ward E. Worldwide variations in colorectal cancer. CA Cancer J Clin. 2009;59:366–378.
Chibaudel B, Tournigand C, Andre T, de Gramont A. Therapeutic strategy in unresectable metastatic colorectal cancer. Ther Adv Med Oncol. 2012;4:75–89.
Zheng L, Zhang Y, Liu Y, et al. MiR-106b induces cell radioresistance via the PTEN/PI3K/AKT pathways and p21 in colorectal cancer. J Transl Med. 2015;13:252.
Prevarskaya N, Skryma R, Shuba Y. Calcium in tumour metastasis: new roles for known actors. Nat Rev Cancer. 2011;11:609–618.
Parkash J, Asotra K. Calcium wave signaling in cancer cells. Life Sci. 2010;87:587–595.
Roberts-Thomson SJ, Peters AA, Grice DM, Monteith GR. ORAI-mediated calcium entry: mechanism and roles, diseases and pharmacology. Pharmacol Ther. 2010;127:121–130.
Parekh AB. Store-operated CRAC channels: function in health and disease. Nat Rev Drug Discovery. 2010;9:399–410.
Song K, Zhong XG, Xia XM, et al. Orai1 forms a signal complex with SK3 channel in gallbladder smooth muscle. Biochem Biophys Res Commun. 2015;466:456–462.
Fedida-Metula S, Feldman B, Koshelev V, Levin-Gromiko U, Voronov E, Fishman D. Lipid rafts couple store-operated Ca2+ entry to constitutive activation of PKB/Akt in a Ca2+/calmodulin-, Src- and PP2A-mediated pathway and promote melanoma tumor growth. Carcinogenesis. 2012;33:740–750.
Davis FM, Azimi I, Faville RA, et al. Induction of epithelial-mesenchymal transition (EMT) in breast cancer cells is calcium signal dependent. Oncogene. 2014;33:2307–2316.
Yang S, Zhang JJ, Huang XY. Orai1 and STIM1 are critical for breast tumor cell migration and metastasis. Cancer Cell. 2009;15:124–134.
Chen YF, Chiu WT, Chen YT, et al. Calcium store sensor stromal-interaction molecule 1-dependent signaling plays an important role in cervical cancer growth, migration, and angiogenesis. Proc Natl Acad Sci USA. 2011;108:15225–15230.
Zhu H, Zhang H, Jin F, et al. Elevated Orai1 expression mediates tumor-promoting intracellular Ca2+ oscillations in human esophageal squamous cell carcinoma. Oncotarget. 2014;5:3455–3471.
Zhan ZY, Zhong LX, Feng M, Wang JF, Liu DB, Xiong JP. Over-expression of Orai1 mediates cell proliferation and associates with poor prognosis in human non-small cell lung carcinoma. Int J Clin Exp Pathol. 2015;8:5080–5088.
Abdelmohsen K, Kim MM, Srikantan S, et al. miR-519 suppresses tumor growth by reducing HuR levels. Cell Cycle (Georgetown, Tex.). 2010;9:1354–1359.
Abdelmohsen K, Srikantan S, Tominaga K, et al. Growth inhibition by miR-519 via multiple p21-inducing pathways. Mol Cell Biol. 2012;32:2530–2548.
Qin Y, Wang J, Gong W, et al. UHRF1 depletion suppresses growth of gallbladder cancer cells through induction of apoptosis and cell cycle arrest. Oncol Rep. 2014;31:2635–2643.
Cvejic S, Zhu Z, Felice SJ, Berman Y, Huang XY. The endogenous ligand Stunted of the GPCR Methuselah extends lifespan in Drosophila. Nat Cell Biol. 2004;6:540–546.
Tommasi S, Pinto R, Pilato B, Paradiso A. Molecular pathways and related target therapies in liver carcinoma. Curr Pharm Des. 2007;13:3279–3287.
Liu L, Dong Z, Liang J, et al. As an independent prognostic factor, FAT10 promotes hepatitis B virus-related hepatocellular carcinoma progression via Akt/GSK3beta pathway. Oncogene. 2014;33:909–920.
Garrett CR, Coppola D, Wenham RM, et al. Phase I pharmacokinetic and pharmacodynamic study of triciribine phosphate monohydrate, a small-molecule inhibitor of AKT phosphorylation, in adult subjects with solid tumors containing activated AKT. Invest New Drugs. 2011;29:1381–1389.
Zhang H, Li Y, Lai M. The microRNA network and tumor metastasis. Oncogene. 2010;29:937–948.
Berridge MJ, Bootman MD, Roderick HL. Calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol. 2003;4:517–529.
Chiu WT, Tang MJ, Jao HC, Shen MR. Soft substrate up-regulates the interaction of STIM1 with store-operated Ca2+ channels that lead to normal epithelial cell apoptosis. Mol Biol Cell. 2008;19:2220–2230.
Carnero A. The PKB/AKT pathway in cancer. Curr Pharm Des. 2010;16:34–44.
Jayadev S, Petranka JG, Cheran SK, Biermann JA, Barrett JC, Murphy E. Reduced capacitative calcium entry correlates with vesicle accumulation and apoptosis. J Biol Chem. 1999;274:8261–8268.
Parekh AB, Putney JW Jr. Store-operated calcium channels. Physiol Rev. 2005;85:757–810.
Zanotto-Filho A, Braganhol E, Schroder R, et al. NFkappaB inhibitors induce cell death in glioblastomas. Biochem Pharmacol. 2011;81:412–424.
Makeyev EV, Maniatis T. Multilevel regulation of gene expression by microRNAs. Science (New York, N.Y.). 2008;319:1789–1790.
Yanaihara N, Caplen N, Bowman E, et al. Unique microRNA molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell. 2006;9:189–198.
Alvarez-Garcia I, Miska EA. MicroRNA functions in animal development and human disease. Development (Cambridge, England). 2005;132:4653–4662.
Krutzfeldt J, Rajewsky N, Braich R, et al. Silencing of microRNAs in vivo with ‘antagomirs’. Nature. 2005;438:685–689.
Abdelmohsen K, Srikantan S, Kuwano Y, Gorospe M. miR-519 reduces cell proliferation by lowering RNA-binding protein HuR levels. Proc Natl Acad Sci USA. 2008;105:20297–20302.
Acknowledgments
We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript. This study was supported by grants from the National Natural Science Foundation of China (No. 51320154).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Deng, W., Wang, J., Zhang, J. et al. Orai1, a Direct Target of microRNA-519, Promotes Progression of Colorectal Cancer via Akt/GSK3β Signaling Pathway. Dig Dis Sci 61, 1553–1560 (2016). https://doi.org/10.1007/s10620-015-4029-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-4029-6