Abstract
Background
Doublecortin-like kinase 1 (DCLK1), a putative tumor stem cell marker has been shown to be highly expressed in the stromal and epithelial compartments in colon and pancreatic cancer as well as Barrett’s esophagus (BE) and esophageal adenocarcinoma (EAC).
Aim
To prospectively investigate whether the immunohistochemical expression of DCLK1 was associated with detectable DCLK1 plasma expression in patients with existing BE and EAC.
Methods
Immunohistochemistry was performed on paraffin-embedded sections using DCLK1 antibody and scored based on staining intensity and tissue involvement. Purified human plasma samples were subjected to Western blot and ELISA analysis.
Results
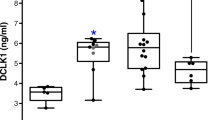
Forty (40) patients were enrolled: 10 controls (normal endoscopy) and 30 with BE/EAC (13 nondysplastic BE [NDBE], 9 dysplastic BE [DBE] and 8 EAC). Mean epithelial DCLK1 staining was as follows: controls = 0.11, NDBE = 3.83, DBE = 6.0, EAC = 7.17. Mean stromal DCLK1 staining was as follows: NDBE = 5.83, DBE = 5.375, EAC = 10.83. DCLK1 was detected by plasma Western blot in 1 control and in all patients with BE/EAC p < 0.0005. Plasma DCLK1 was elevated by ELISA in EAC compared to other groups, p < 0.05.
Conclusions
Increased expression of DCLK1 was observed in the epithelium, stroma and plasma of patients with BE/EAC. Furthermore, the presence of detectable DCLK1 in plasma of BE/EAC patients may provide a less invasive, detection tool in those patients as well as represent a novel molecular marker distinguishing between normal esophageal mucosa and BE or EAC.



Similar content being viewed by others
Abbreviations
- BE:
-
Barrett’s esophagus
- DBE:
-
BE with dysplasia
- TSCs:
-
Tumor stem cells
- DCLK1:
-
Doublecortin-like kinase 1
- EAC:
-
Esophageal adenocarcinoma
- GERD:
-
Gastroesophageal reflux disease
- HGD:
-
High-grade dysplasia
- LGD:
-
Low-grade dysplasia
- NDBE:
-
Nondysplastic BE
References
Locke GR 3rd, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ 3rd. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County. Minnesota Gastroenterol. 1997;112:1448–1456.
Spechler SJ. Clinical practice. Barrett’s Esophagus. N Engl J Med. 2002;346:836–842.
Reid BJ, Sanchez CA, Blount PL, Levine DS. Barrett’s esophagus: cell cycle abnormalities in advancing stages of neoplastic progression. Gastroenterology. 1993;105:119–129.
Pohl H, Welch HG. The role of overdiagnosis and reclassification in the marked increase of esophageal adenocarcinoma incidence. J Natl Cancer Inst. 2005;97:142–146.
Quante M, Bhagat G, Abrams JA, et al. Bile acid and inflammation activate gastric cardia stem cells in a mouse model of Barrett-like metaplasia. Cancer Cell. 2012;21:36–51.
Tu SP, Quante M, Bhagat G, et al. IFN-gamma inhibits gastric carcinogenesis by inducing epithelial cell autophagy and T-cell apoptosis. Cancer Res. 2011;71:4247–4259.
Sureban SM, May R, Lightfoot SA, et al. DCAMKL-1 regulates epithelial-mesenchymal transition in human pancreatic cells through a miR-200a-dependent mechanism. Cancer Res. 2011;71:2328–2338.
Sureban SM, May R, Mondalek FG, et al. Nanoparticle-based delivery of siDCAMKL-1 increases microRNA-144 and inhibits colorectal cancer tumor growth via a Notch-1 dependent mechanism. J Nanobiotechnol. 2011;9:40.
Sureban SM, May R, Ramalingam S, et al. Selective blockade of DCAMKL-1 results in tumor growth arrest by a Let-7a MicroRNA-dependent mechanism. Gastroenterology. 2009; 137:649–659, 659 e1-2.
Sureban SM, May R, Weygant N, et al. XMD8-92 inhibits pancreatic tumor xenograft growth via a DCLK1-dependent mechanism. Cancer Lett. 2014;351:151–161.
Sureban SM, May R, Qu D, et al. DCLK1 regulates pluripotency and angiogenic factors via microRNA-dependent mechanisms in pancreatic cancer. PLoS ONE. 2013;8:e73940.
Vega KJ, May R, Sureban SM, et al. Identification of the putative intestinal stem cell marker doublecortin and CaM kinase-like-1 in Barrett’s esophagus and esophageal adenocarcinoma. J Gastroenterol Hepatol. 2012;27:773–780.
Regitnig P, Reiner A, Dinges HP, et al. Quality assurance for detection of estrogen and progesterone receptors by immunohistochemistry in Austrian pathology laboratories. Virchows Arch. 2002;441:328–334.
Hur C, Miller M, Kong CY, et al. Trends in esophageal adenocarcinoma incidence and mortality. Cancer. 2013;119:1149–1158.
Wani S, Falk GW, Post J, et al. Risk factors for progression of low-grade dysplasia in patients with Barrett’s esophagus. Gastroenterology. 2011;141:1179–1186.
Bhat S, Coleman HG, Yousef F, et al. Risk of malignant progression in Barrett’s oesophagus patients: results from a large population based study. J Natl Cancer Inst. 2011;103:1049–1057.
Rugge M, Zaninotto G, Parente P, et al. Barrett’s esophagus and adenocarcinoma risk: the experience of the North-Eastern Italian registry (EBRA). Ann Surg. 2012;256:788–794.
Acknowledgments
CWH received grant support from VA Merit Award and OCAST-AR101-030.
Conflict of interest
CWH is a co-founder of COARE Biotechnology Inc; other authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Whorton, J., Sureban, S.M., May, R. et al. DCLK1 Is Detectable in Plasma of Patients with Barrett’s Esophagus and Esophageal Adenocarcinoma. Dig Dis Sci 60, 509–513 (2015). https://doi.org/10.1007/s10620-014-3347-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3347-4